
Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-6679
Review Article(ISSN: 2637-6679) 
Nanoemulgel Innovative Approach for Topical Gel Based Formulation Volume 1 - Issue 2
Malay N Jivani, Chandresh P Patel and Bhupendra G Prajapati*
- Department of Pharmaceutical Technology, India
Received: February 5,2018; Published: February 15, 2018
*Corresponding author: Bhupendra G Prajapati, Associate Professor, Shree S.K.Patel College of Pharmaceutical Education and Research, Ganpat University, Ganpat Vidyanagar, Mehsana, Gujarat, India
DOI: 10.32474/RRHOAJ.2018.01.000107
Abstract
The lipophilic nature of majority of newly developed drugs has resulting in poor bioavailability, and pharmacokinetic variations. The goal of this evaluation is to evaluate and report the current potential and future scope of nanoemulgel formulation for becoming an effective delivery system for poorly water-soluble drugs. Nanoemulgel consist two different systems in which drug containing nanoemulsionis incorporated into a gel base. The fusion of these two systems makes this formulation advantageous in several ways. Lipophilic drugs can be easily incorporated and the skin permeability of the incorporated drugs can be enhanced in several folds due to the finely distributed oil droplets in gel phase. Simultaneously, it can be targeted more specifically to the site of action and can avoid first pass metabolism and relieve the user from gastric/systemic incompatibilities. The nanoemulgel drug delivery system is a formulation related intervention to improve drug absorption and therapeutic profile of lipophilic drugs. An increasing trend in nanoemulgel use in recent years has been noticed because of the better acceptability of the preparation to the patients due to their non-greasy, convenience spreadability, and easy applicability and good therapeutic and safety profile. Despite having few limitations, nanoemulgel formulation can be considered as potential and promising candidates for topical delivery of lipophilic drugs in the future.
Keywords: Nanoemulgel; Topical Formulation; Drug Delivery through skin
Introduction
Emulgel
Emulgel is a combination of gel and emulsion where emulsion used can be both type W/O and O/W as a vehicle for purpose to deliver selected drug to the skin. Water Phase containing the gelling agent converts a classic emulsion in emulgel [1]. Dermatological use of Emulgel has many favourable properties like easy spreadable, greaseless, being thixotropic, water-soluble, easy removal, longer shelf life, non-staining, and bio-friendly.
Drugs can penetrate into the skin structure:
a. through thick stratum corneum, (SC)
b. Sebaceous follicle.
c. sweat ducts of skin,
Stratum corneum covers more than 99% of skin available for drugs to be absorbed. Passing through this is the rate limiting step for drug percutaneous absorption. Establishment of a concentration gradient thought to be major steps involved in percutaneous absorption, which provides force necessary for drug adsorption across the skin [2].
Nanoemulgel
Formation containing Nanoemulsion in gel base are called nanoemulgel, is the addition of Nanoemulsion system intergraded into gel matrix which influences a better skin permeation [3]. This mixture of nanomulgel acts as drug reservoirs, influencing the release of drug from inner phase to outer phase and further. Nanoemulgel on intact with skin release the oil droplets from the gel and this oil droplets penetrate into the SC of the skin and deliver the drug to intended site. Nanoemulsion-gel have a good adhesion property and high solubilising of drug in oil phase leads to larger concentration gradient towards the skin that further increase skin penetration of drug. Also patient compliance is improved due to increased spared ability compare to creams and ointments and decreased stickiness.
Rational
Topical dosage forms like cream, lotion, ointment have many disadvantages. One of which is stickiness, causing trouble to patients in application and having low spreading properties and rubbing requirement are also considered as disadvantages. Also stability of formulation for hydrophilic medication indicated some problems. Due to these drawbacks with the major group of semisolid preparations, the use of gelled formulation has expanded both in pharmaceutical preparations and in cosmetics. Gel is colloid containing 99 % part of liquid where macromolecular network of fibres built from a gelling substance and liquids are immobilized by surface tension between them. Despite of advantages a major problem is to delivery of hydrophobic natured drugs. Emulsion based approach can be used to incorporate lipophilic therapeutic moiety in gel based system to overcome this problem4.
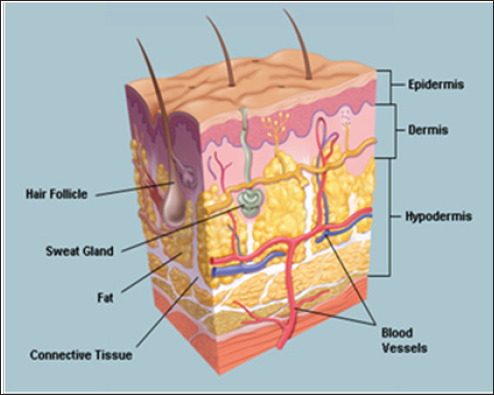
Skin Structure
The skin of most adult human body has average surface area of 1.8 m2, which contain average 200-300 sweat ducts and 40-70 hair follicles on sq. cm of the skin. Skin pH varies from 4 to 5.6 which depends of secretion form the sweat glands. Cutaneouse membrane is divides into 3 parts,
a. Epidermis
b. Dermis
c. Hypodermis
a.Epidermis
Epidermis is the outer most arena of the skin, containing stratified squamous epithelium which has 2 layers, stratum basale - deepest layer and stratum corneum - superficial layer. Stratum corneum, which is the outermost layer of skin, becomes hard due to the process of keratinisation and act as physical barrier to most drugs, SC composed of 10 to 20 cells layers. Cell of SC are flate and plate-like with 2500 μm wide, 34-44μm long, 0.5 to 0.20μm thick. Lipid content of SC includes glycosphingolipid, a neutral lipid, phospholipids, cholesterol sulphate and Keratin as protein. Stratum Basale lies between the SC and thedermis having thickness of 50-100μm. Cells Structure of this layer are similar to other living tissues. The water content of this layer is around 90% mainly contain melanin and langerhans cells [4,5].
b. Dermis/Corium
Deeper and thicker (2000 to 3000μm) region beneath the epidermis is called dermis, mostly contracted with structural fibrin and very few cells same as found in normal tissue. Dermal papillae are outer most layer having signalized projection. It also contains oil-secreting glands, sweat glands, nerve ending and hair follicles. Elastic and collagenous fibers within dermis provide starchability and tone to the skin. The density of its fibre meshwork, and therefore its physical properties, varies within an area, in different parts of the body, and with age and sex [6].
c. Hypodermis/Subcutaneous layer
The hypodermis, composed of loose connective tissue varies in thickness, merges with the deep lower part of the dermis. Apart of this it also composed of secretary sweat glands, fibrous tissue containing blood vessels and lymph vessels, and cutaneous nerves. Many consider that drug get absorbed into the systemic circulation without passing through this layer by means of blood vesicles and sweat glands [5].
Figure 1: Different Layers of the skin [23].
Drug Delivery Through Skin
Stratum Corneum of the skin acts as a powerful water barrier and protects deep inner structures and blood vesicles which are distributed below the skin. Especially continuous venous plexus which is supplied by flow of blood from the skin's blood capillaries is more important. More exposed areas like feet and hands; blood is also supplied directly through small arteries from highly muscular arteriovenous anastomoses. The skin prevents absorption along with loss of electrolytes and water. Passing from this barrier and getting absorbed in to skin layers, drug has to go through one of these channels; intercellular, transcellular, and follicular [7,8]. But follicular route also know as shunt pathway found controversial in many study so mainly other two are considered as main mechanism of drug absorption. Intercellular route thought to be predominating for most drugs compare to transcellular where drug has to absorb through several of lipophilic and hydrophilic differential skin layers [9] Figure 1.
Factors Affecting Topical Absorption of Drug
a. Physiological factors [10,11]
i. Lipid content of the skin - act as barrier for drug absorption and lowering this barrier property leads to increased penetration.
ii. Thickness of different skin layers -Greater the thickness lower the penetration rate, like palm and sole shows higher diffusion rate compare to other surfaces.
iii. Hair follicles density - large storage, about 10-12 times than SC.
iv. Skin pH.
v. Hydration of skin.
vi. Sweat gland density.
vii. Inflammation of skin disrupted stratum corneum has higher permeability.
viii. Blood flow.
ix. Skin temperature
b. Physicochemical factors [12]
i. Partition coefficient - higher log p value give rise to absorption.
ii. Effect of vehicles - hydro alcoholic gel provides the most efficient absorption through skin.
iii. Degree of ionisation.
iv. Molecular Weight. (less than 400 Dalton)
Advantage Of Nanoemulgel [13]
a. Stability of Nanoemulsion is enhanced due to distribution of oil droplets in Gel base; where affinity of the drug toward oil determines stability.
b. Also good adhesion on the skin with high solubilising power leads to high concentration gradient that increase penetration of drug as it moves down.
c. Moreover, these types of formulation give support to delivery of lipophilic and poorly water soluble drugs and also improve patient compliance.
d. Nanoemulgel also helps in controlled release of drugs having the shorter half-life.
e. Provide higher Spread-ability of the formulation than creams.
f. Nanoemulgel are Non toxic and non irritant.
g. Better loading of drug compare to other formulation.
h. Increase skin permeability and drug deposition.
Important Component of Nanoemulgel
a. Oils: Oils used in Nanoemulsion are generally mineral oils used as the vehicle for drugs E.g. castor oils and various fixed oils (cottonseed oil, maize oils, arachisoil ) Olive Oil, Coconut Oil, eucalyptus oil, rose oil, clove oil etc.[14]
b. Aqueous Phase: Commonly distilled water is used as a aqueous phase for the preparation of Nanomulsion and hydrogel.
c. Surfactant and Co-Surfactant: urfactants are used both to give emulsification at the time of formulation and control day to day stability during shelf life of prepared Nanoemulsion. General selection of surfactant depends on the type of emulsion [15]. (O/W or W/O) E.g. Span 80 (Sorbitanmonooleate), Acrysol K 140, Polyethylene-glycol-40-stearate, Acrysol, Labrasol, Stearic acid, PlurolOleique, Tween 80 (Polyoxyethylene- sorbitan-monooleate), Labrafil, Sodium stearate, Where agents like Transcutol ,Captex, Cammul, Migyol, etc. can be use as cosurfactant or co-solvents.
d. Gelling Agent: Polymers essential to give the structural network for the preparation of gels are known as gelling agents E.g. Natural - Agar, Tragacanth, Guar gum, Xanthan Gum, Semisynthetic and Synthetic Carbapol, Poloxamer, HPMC (cellulose derivatives)
e. Permeation Enhancers: They interact with different skin constituents to produce a reversible temporary increase in permeability. They can act by one or more mechanisms like,
i. Disrupting the highly compact structure of SC.
ii. Improving partition of drug or solvent or co-enhancer into the SC.
iii. Interacting intercellular protein.
Causing conformational changes in protein or solvent swelling is the key for alternating polar path. Some enhancers improve the fluidity of protein in SC, where some act on both pathways by disrupting multilaminate pathway. They can increase the diffusion of drug through skin proteins. Type of enhancer has a significant impact product designing [16] E.g. Eucalyptus oil, Linoleic acid, Lecithin, Oleic acid, Chenopodium oil, Isopropyl myristate, Urea.
Methods Of Formulation
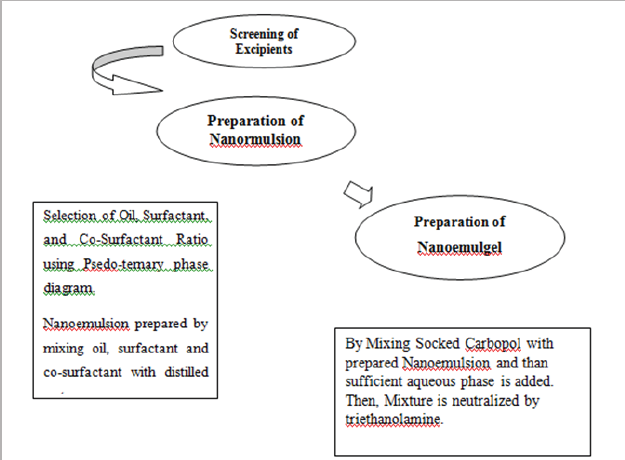
Formulation of Nanoemulsion-gel can be summarized in to following steps,
a. Screening of components
b. Preparation of Nanoemulsion
c. Preparation of Nanoemulgel.
a.Screening of components
Drug Solubility was determined in different oils by excess addition of drug into different components followed by continuously stirred 72 hours to achieve equilibrium. After that samples centrifuged and supernatant was taken and solubility was determined by appropriate analytical methods. Then, excipients in each category with the highest solubility of drug are selected for further studies [17].
i. Psedoternary phase diagram: Surfactant and cosurfactant (Nmix) were mixed in different ratios (2:1, 3:1 and 5:1). Each ratio chosen in increasing amount of surfactant respect to co surfactant for a study on the phase diagrams. Here aqueous phase (Distilled water) used as dilution media. Oil and Nmix was mixed at different ratios from 9:1 to 1:9 in different vials for each Nmix. Main objective for this is to cover for the study to decide boundaries of phases formed in the diagrams. It was developed using titration method with help of water as aqueous media. Slow titration of oil and Nmix is performed and visual observations are made for transparency of Nanoemulsion. The state of Nanoemulsion is marked on one axis of aqueous phase, the second one of oil and the third one of Nmix (surfactant and co-surfactant) [18].
b. Preparation of Nanoemulsion: The drug is then solubilized in oil and oil is addend in to Nmix, this mixture is diluted with water to form of Nanoemulsion of given drug.
c. Preparation of Nanoemulgel: Gel base is prepared using 1g of the Carbopol in a required quantity of water. After complete swelling and dispersion of Carbopol solution during 24 hours period, prepared Nanoemulsion is slowly added under continues stirring. Addition of Triethanolamine gives homogeneous gel dispersion. Finally required remaining part is adjusted with distilled water Figure 2.
Figure 2: Flowchart for Preparation of Nanomeulgel.
Optimization And Evaluation
a. Measurement of pH : Various Topical formulations have pH in range of 5-6 measured by using pH meter. For testing, 1g of gel is dissolve in 10ml water. PH of each formulation is done on triplicate to avoid error [18].
b. Size of globules: To determine this parameter 1.0 gm of gel was dissolved in water and stirred to get dispersion and then sample was injected into the photocell of Malvern zetasizerr.
c. Swelling Index: 1 gm of prepared topical nanoemulgel is taken on porous aluminium foil which is then placed on 10 ml of 0.1 N NaOH solutions. Sample removed time to time and weight is noted till no further change in weight:
Swelling Index (SW) % = [[Wt-Wo]/Wo] 100
Where, (SW) % = Percentage swelling,
Wo = Original weight of nanoemulgel
Wt = Weight of swollen nanoemulgel at time t
Measurement of Bio adhesive strength
One glass slide separated from two other glassed plates on each arm of apparatus. Single plate is for adding weight. Accurately 1 gm of nanoemulgel is placed between slides containing rate skin pieces (hairless0. Putting weight on single glass slide create some pressure to removed sandwich of two slides. Addition of extra weight is followed as 200 mg/ min to until the detachment of the skin surface. Required weight to detach the nanoemulgel from skin gave bio adhesive strength1. It is calculated by using following equation:
Bio adhesive Strength = W / A
Where, W= Weight required (in gms) and A=Area (cm2)
Determination of Rheological properties
20gm of Nanoemulsion-gel filled in 25ml beaker was used to measure viscosity by using Spindle number S64 by Brookfield viscometer [19].
Accelerated stability studies
As given in ICH guidelines. The formulations are stored in oven at 37±2°C, 45±2°C and 60±2°C differently for 3 months. Drug content is analysed every two week by suitable analytical method. Stability measurement is based on change in pH of gel or drug degradation [20].
a. Determination of % drug content: 1 g of Nanoemulgel is mixed with 25 ml of methanol. This solution is sonicated for 30 min. Drug content was calculated using the suitable analytical method from this solution.
Spreadability of Gellifed Nanoemulgel:
It can be measured by using Slip and Drag basis, as suggested by Mutimer, Here 2gm od Nanoemulgel is palced on lower ground slide which is fixed with wooden block and sandwiched is prepared by other glass slide having similar size which is attached with hook having 500mg weight placed. After 5 min extra weight was placed on pan connected with second slide. Time to cover 5cm distance for upper slide was noted and used to calculate spreadability by using following equation:
Spreadability (S) = M*L / T
Where, M = Weight tied to upper slide,
L = Length of glass slides
T = Time taken to cover distance by upper slide
a. Skin irritation test
0.25 gm Nanoemulgel is applied to each different site (two sites/rabbit). After 24 hr of application rabbit skin site are wiped and cleaned, Change in colour of skin or undesirable change in morphology is noted and checked.
b. In-vitro Diffusion studies
Franz diffusion cell is used to perform diffusion study of prepared nanomeulgel. A cellophane membrane is used for study and 0.5g of sample applied on membrane and diffusion is carried out for 8 hr at 37±1°C using phosphate buffer (pH 7.4). At time interval of 1 hr, 1 ml pg sample is collected and replaced with new buffer solution. Collected samples are analyzed by using suitable analytical method [21].
Assessment of Skin Permeation
The changes like chemical and structural in epidermal layer are determined by using differential scanning calorimetry (DSC). In order to assess mechanism of permeation, thermal transitions in desiccated SC membranes of rats is investigated by means of DSC. Both treated and untreated skin samples were previously hydrated on 27% Sodium-Br solution for at least 48 h to ensure lowering hydration to 20%. The skin samples are stored over silica gel, for 3 days in desiccators before analysis. The skin sheets is cut into pieces and 4 mg weighted pieces is sealed in 10|iL aluminium pans and kept in the differential scanning calorimetry unit along with empty pan as a reference. Flow of Nitrogen is adjusted to 20ml/ min which is used as purge gas. Samples are heated continuously at 10°C/min rate for the range of 30-400°C and fluctuation in DSC Graph is noted and studied [22].
Conclusion
Topical Nanoemulgels have proven as better option for effective and convenient drug delivery system. Gel and non-greasy like properties are giving more patient compliance and lacks of oily as a base provides better drug release compared to other formulations. Incorporation of Nanoemulsion into gel matrix makes formulation dually control released system, Problems like creaming and phase separation which is associated with classical emulsion gets resolved with improved sparedability. Nanoemulsion loaded gel gives higher effectiveness in some topical disorders. Future of Nanomulsion-Gel based formulations may provide a better and reliable solution for delivery of hydrophobic drugs. A considerable lot of medications utilized as a part of treatment of skin infection are hydrophobic natured and such medications can be conveyed successfully as Nanoemulgel where drug is incorporated into oil phase of Nanoemulsion and then merged with gel base. In spite of having couples of impediments, nanoemulgel has the likelihood to possess the focal place for topical conveyance for lipophilic drugs in future
References
- Panwar, N Upadhyay, M Bairagi, S Gujar, G Darwhekar (2011) Emulgel: A Review. Asian Journal of Pharmacy and Life Science 1(3): 333-343.
- S Yadav, M Mishra, A Tiwari, Ashutosh Shukla (2017) Emulgel: A New Approach for Enhanced Topical Drug Delivery. International Journal of Current Pharmaceutical Research 9(1): 15-19.
- 3. R Sigh (2014) Emulgel: A Recent Approach for Topical Drug Delivery System. Asian Journal of Pharmaceutical Research and Development 2(2): 13-15.
- S Pant, A Badola, S Baluni, W Pant (2015) A Review on Emulgel Novel Approach for Topical Drug Delivery System. World Journal of Pharmacy and Pharmaceutical Sciences 4(10): 1728-1743.
- S Mader (2004) Understanding Human Anatomy & Physiology. The McGraw-Hill Companies 5th edn. p. 70-72.
- K Van De Graaff (2001) Human Anatomy. The McGraw-Hill Companies 6th edn. pp. 106-112.
- M Rieger, L Lachman, H Lieberman, J Kanig (1986) the Theory and Practice of Industrial Pharmacy. PA Lea and Febiger, Philadelphia 3rd edn. Pp. 502-533.
- Waugh A, Grant A (2004) Ross and Wilson Anatomy and Pharmacology in Health and Illness. Churchill living stone pp. 361-364.
- Maibach, R Feldmann, T Milby, serat WF (1971) Regional Variation in Percutaneous Penetration in Man. Archives of Environmental Health, An International Journal 23(3): 208-211.
- Y Kalia, R Guy (2001) Modeling Transdermal Drug Release. Advanced Drug Delivery Reviews 48: 159-172.
- Ayub AC, Gomes AD, MV Lima, C Soares, L Ferreira, et al. (2007) Topical Delivery of Fluconazole In-Vitro Skin Penetration and Permeation using Emulsions as Dosage Forms. Drug Development and Industrial Pharmacy 33: 273-280.
- Ajazuddin, Alexander A, Khichariya A, S Gupta, R Patel, et al. (2013) Recent Expansions in an Emergent Novel Drug Delivery Technology, Emulgel. Journal of Control Release 171: 122-132.
- Joshi Baibhav, G Shing, S Saini, V Singla (2011) Emulgel : A Comprehensive Review on the Recent Advances In Topical Drug Delivery. International Research Journal of Pharmacy 2(11): 66-70.
- Montenegro L, C Carbone, G Condorelli, R Drago, G Puglisi, et al. (2006) Effect of Oil Phase Lipophilicity on In Vitro Drug Release from O/W Micro emulsions with Low Surfactant Content. Drug Development and Industrial Pharmacy 32: 539-548.
- S Savale (2015) A Review - Self Nanoemulsifying Drug Delivery System (SNEDDS). International Journal of Research in Pharmaceutical and Nano Sciences 4(6): 385-397.
- S Mortazavi, R Aboofazeli (2003) an Investigation into the Effect of Various Penetration Enhancers on Percutaneous Absorption of Piroxicam. Iranian Journal of Pharmaceutical Research 2: 135-140.
- R Shankar, V Tiwari, C Mishra, C Singh, D Sharma, et al. (2015) Formulation and Evaluation of Ketoconazole Nanoemulsion Gel for Topical Delivery. American Journal of Pharmtech Reseatch 5(5): 446-462.
- J Modi, J Patel (2011) Nanoemulsion-Based Gel Formulation of Aceclofenac for Topical Delivery. International Jourmacy and Pharmaceutical Science Research 1(1): 6-12.
- G Bonacucina, M Cespi, G Palmieri (2009) Characterization and Stability of Emulsion Gels Based on Acrylamide/Sodium AcryloyldimethylTau- rate Copolymer. American Association of Pharmaceutical Science Pharm SciTech 10(2): 368-375.
- H Masmoudi, P Piccerelle, Yveline Le D, Jacky kister (2006) A Rheolog- ical Method To Evaluate The Physical Stability of Highly Viscous Pharmaceutical Oil-In-Water Emulsions. Pharmaceutical Research 23(8): 1937-1947.
- Y Tanwar, A Jain (2012) Formulation And Evaluation of Topical Diclofenac Sodium Gel Using Different Gelling Agent. Asian Journal of Pharmaceutical Research and Health Care 4(1): 1-6.
- F Shakeel, S Baboota, A Ahuja, J Ali, Shafiq S, et al. (2008) Skin Permeation Mechanism of Aceclofenac Using Novel Nanoemulsion Formulation. Pharmacies 63: 580-584.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...

.png)











