
Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-6679
Review Article(ISSN: 2637-6679) 
"Crystallo Co Agglomerations" The Novel Approach For Micro particulation Volume 1 - Issue 3
Chandresh P Patel, MalayN Jivani and Bhupendra G Prajapati*
- Department of Pharmaceutical Technology, Ganpat University, India
Received: March 14, 2018; Published: March 20, 2018
*Corresponding author: Bhupendra G Prajapati, Associate Professor, Department of Pharmaceutical Technology, Ganpat University, Ganpat Vidhyanagar, Mehsana, Gujarat-384012, India
DOI: 10.32474/RRHOAJ.2018.01.000115
Abstract
Coordinate pressure is the favoured technique for tablet producing because of the effortlessness in preparing and its cost adequacy. None the less, to apply coordinate pressure in tablet fabricating, the medication utilized ought to have great stream properties and compaction qualities. Numerous medications are inadequate with regards to these properties thus it isn't conceivable to pack them specifically into tablets. Crystallo co agglomeration (CCA) is an imaginative procedure created with plans to furnish the medications with great micrometrics and mechanical qualities. The procedure of CCA includes crystallization took after by concurrent agglomeration of the medication with the guide of a good solvent and/or a connecting (bridging liquid) fluid and an awful dissolvable. It is likewise conceivable to consolidate different medications (to get a blend tablet), excipients (e.g. disintigrants for quick dissolving tablets) and diverse polymer blends (to change the medication discharge properties). In the current years, attempts were made to deliver Crystallo co agglomerates of different medications, which were quickly talked about in this article. Despite the fact that extensive scale utilizations of CCA isn't yet made conceivable, this method gives another line of chances to the tablet fabricating process, guaranteeing ease, single ventured generation of particles with great micromeritics and mechanical characters which can be specifically packed.
Keywords: Crystallo co agglomeration; Good solvent; Bridging liquid; Micromeritics
Abbreviations: CCA: Crystallo Co Agglomeration; GIT: Gastro-Intestinal Tract; CS: Crushing Strength; Tc: Crystallization Temperature; PVP: Polyvinyl Pyrrolidone; HPMC: Hydroxy Propyl Methylcellulose; PEG: Poly Ethylene Glycol; EC: Ethyl Cellulose; PVP: Poly Vinyl pyruvate
Introduction
The oral course is the most favoured alternative to direct medications for foundational and in addition neighbourhood impacts in the human body. Strong measurement structures, for example, tablets and cases are the favoured class of items controlled through the oral course. There are a few favourable circumstances accessible for strong dose structures, for example, great restorative impacts, cost, simplicity of pressing and shipment, item ID through monogram or etching, strength, adjustment in discharge properties and patient consistence. Tablets might have slight favourable position above case as a favoured strong dose shape for oral organization. The goal behind the plan and advancement of packed tablet is to convey orally the right dosage of medication in appropriate frame, at legitimate time and in the coveted area with the capacity to secure the concoction respectability. The plan and assembling of tablets comprise of basic advances, for example, granulation and pressure.
The properties of powder material associated with the plan assume a basic part in compelling granulation and after that onwards in pressure into tablets. The tablets might be defined by wet granulation, pressure granulation and direct pressure. The procedure, for example, wet granulation and pressure granulation includes more advances and time to wrap up. The materials which are not steady under warmth and dampness may not be reasonable to process through more prevalent system of wet granulation. Until the late 1950's, the majority of tablets were produced in the world by means of granulation techniques and subsequent compression [1,2]. The essential target of granulation is to deliver free streaming and exceptionally compressible blend of fixings. The excipients should indicate great stream, union and greasing up properties under strain to make such compacts.
The accessibility of new types of excipients and altered hardware and additionally advances enabled the conceivable outcomes to create tablets by coordinate pressure. The direct compression was long used to for the compression of single crystalline compound into a compact without the addition of other substances. The immediate pressure process has points of interest, for example, lesser preparing time and steps, diminished work, lesser process approval, less synthetic solidness issues and so forth. The reason, why CCA has increased extraordinary place in oral medication conveyance framework lies in its straight forwardness and capacity to create round agglomerates in a solitary advance [3,4]. The circular agglomerates acquired can be utilized as spansules or specifically compressible agglomerates. They offer points of interest like magnificent stream characters, uniform size dissemination, and reproducible pressing/filling. Substantial surface territory offered by circles brings about uniform dispersion all through gastro-intestinal tract (GIT) prompting lessening in the restricted lethality [5-7].
In addition, this uniform dissemination may enhance assimilation and bioavailability of medication/s. In light of the low surface zone to-volume proportion contrasted with powder or granules, they can be considered as a brilliant coating substrate [8,9]. Circles indicate change in the remedial characteristics of dose frame because of good dosing and dealing with properties [10]. They are less helpless to dosage dumping [11], and disappointment of a couple of units may not be as important as disappointment of a solitary unit framework. Another critical preferred standpoint of circles lies in that they get minimum influenced by the typical gastric discharging time and consequently medicate conveyance utilizing same is less inclined to physiological factors [12]. It has been accounted for that pellets littler than around 2.4 mm breadth are free from stomach related capacity of the stomach and the end arrangement of the pyloric sphincter to be discharged from the stomach. A greatest pellet breadth of 1.5 mm has been prescribed for an ideal various unit definition [13,14].
A few discoveries have referred to limit measure beneath 1 mm. The impact of both thickness and size of the pellet influences the gastrointestinal travel time [15,16]. The higher thickness of the pellets has delayed the gastric travel time, while the bigger size marginally drawn out the little gut travel time however not the gastric travel time[17].
Advantages[18]
a. Great stream properties.
b. Uniform size dissemination.
c. The procedure is extremely straightforward
d. Less handling cost which makes the generation financial.
e. Unit tasks are minimal.
f. Single step age of agglomerates.
g. The procedure requires less work - one individual required for whole task.
h. The straightforwardness in the process helps in empowering the maker to go along effectively with CGMP.
i. Crystallo co agglomerates can be utilized as tablet intermediates and for the plan of MUPS.
j. Large surface territory that empowers uniform dispersion of medication through gastro-intestinal tract. This thus helps in diminished poisonous quality, enhanced assimilation and along these lines sufficient bioavailability.
k. The low surface region to volume proportion makes them incredible holding up substrates
l. They have great remedial characteristics because of enhanced dosing and taking care of properties.
m. They are minimum influenced by gastric discharging.
n. Their medication conveyance is less inclined to physiological factors.
o. They indicate less measurements dumping.
p. If the pellet measure is under 2.4 mm distance across, at that point they are free from gastric stomach related capacity and shutting arrangement of pyloric sphincter
Kinetics for spherical crystallization & crystallo-co- agglomeration
a. Process
Initial works reported that spherical crystallization follows first order or second order kinetics, but detailed work on mechanism of agglomeration have shown that agglomeration process follows first order kinetics.[8] This behaviour is explained by the restricted movement of particles in space due to particle interaction, such as layering agglomerates of fine particles on coarse ones.
Spherical crystallization process has been described by a selective coalescence mechanism. The kinetic equation is,
Log d = C log t + C' (λ) (1.1)
Where, d = diameter of agglomerates (mm)
t = agglomeration time (min)
C' (λ) = function of coalescence time
C = constant
During crystallo-co-agglomeration process, agglomerates were spheronized and compacted. The compaction process of agglomerates was represented by the changes in porosity of agglomerates with agglomeration time. The agglomerates were more easily compacted by increase in agitation speed and amount of bridging liquid, because they increase the sheer force applied to agglomerates as well as enhance the plasticity.
Process Design Studies
In CCA, synchronous crystallization and agglomeration of particles are done in a solitary advance and circular agglomerates are acquired. The framework outline for CCA prescribes utilization of good solvent to solubilise drug, non-solvent to cause precipitation/crystallization of drug/s and the crossing over fluid which basically must be immiscible with non- to shape the fluid extensions between solidified particles and insoluble solids amid the procedure of agglomeration. Once in a while crossing over fluid goes about as a decent soluble also [19]. Till date, two strategies have been created for CCA. Solvent change technique includes synchronous crystallization and agglomeration of at least two medications from a decent soluble and spanning fluid by expansion of a non-dissolvable. The second strategy includes crystallization of medication from a framework containing a decent soluble and crossing over fluid and its synchronous agglomeration with insoluble medication/diluents by expansion of a non-soluble.
Determination of both of these techniques requires information of the physicochemical properties of medication and dissolvable framework. Once the technique has been chosen, at that point its preparing should be possible in a vessel portrayed by Morishima et al. [13,20] for SC. The controlled tumult of substance in Morishima vessel creates round agglomerates. The endpoint of the agglomeration procedure can be judged by the span of agglomerates, clearness of supernatant and vaporization of natural solvent/s from the agglomeration framework. Successful outline of the CCA procedure relies upon various variables influencing the procedure of crystallization and agglomeration. Or maybe, it is an exceptionally complex procedure to be examined, getting affected by various definition and process factors. Different components influencing CCA have been described ahead.
Formulation Factors/Variables
a. Diluent selection
The utilization of diluent has been proposed in CCA for size enlargement of low measurements drugs. Diluent chose must be physico-chemically and physiologically inert, and cheap. Also, it ought to be insoluble in the watery stage to keep away from the misfortunes through the constant/outside phase. Considering wanted qualities, powder has been utilized as a diluent in the improvement of the CCA procedure [21]. By utilizing powder, placebo beads have been set up by Limzerwala. Along these lines, Gadekar and Jadhav have built up the procedure for size enlargement of low dosage bromhexine hydrochloride (BXH) utilizing Talk as diluents [21]. On similar lines, utilization of powder has been made by Pawar in the agglomeration of ibuprofen, a high dose drug [22,21]. No reports on the gastrointestinal disorders caused by Talk upon oral ingestion have been showed up [23]. Adsorption considers have indicated minimum adsorption of cimitedine [24] and bromhexinehydrochloride [25] on Talk. Along these lines, it can be inferred that, claim of Talk as an excipient/diluent in dab/ pellet making gets fortified further. As of late, starch and Na-starch glycolate has been utilized as a part of arrangement of rapidly disintegrating agglomerates of naproxen by the CCA procedure.
b. Solvent System
The solvent system choice for the CCA procedure relies upon solubility and stability of medication/s. Since, dominant parts of medications are soluble in organic solvents and inadequately soluble in water. Utilization of organic solvent (generally nontoxic) has been suggested as a good solvent and additionally bridging fluid and water as an outside/preparing stage (non-solvent). This kind of solvent choice has been recommended because of rare prerequisite of organic solvent [21]. The bridging fluid should complete particular wetting of crystals/solids and frame fluid extensions amid the procedure of agglomeration, and at the same time, it ought to be immiscible with a non-solvent. On the off chance that bridging fluid is utilized as a good solvent, it implies, it performs double part of a good solvent and bridging fluid. The good solvent utilized ought to be unpredictable and immiscible with nonsolvent to maintain a strategic distance from tranquilize misfortune because of co-dissolvability [21,22].
c. Dispersion of internal phase
The internal phase made out of medication solution/suspension with or without diluent and bridging liquid ought to be effectively emulsified/scattered in the outer phase. The procedure can be helped by determination of different appropriating operators/ dispersants. Different hydrophilic polymers and surfactants, for example, polysorbates, polyvinyl pyrrolidone (PVP), and polyvinyl liquor (PVA) have been accounted for to encourage scattering in ideal fixations [19].
d. Polymers
It was discovered that the Crystallo co agglomerates unadulterated drugs have poor compressibility and handling characteristics. This will keep the utilization of direct compressing in tablet making and accordingly falls flat the reason. So different polymers like hydroxy propyl methylcellulose (HPMC), poly ethylene glycol (PEG), ethyl cellulose (EC) and poly vinyl pyruvate (PVP) were used. This enhances the micromeritics mechanical and drug discharge properties of the agglomerates [19].
e. Drug Loading
The degree of drug loading in agglomerate changes the necessity of bridging liquid, good solvent, and non-solvent in CCA. It has been watched that the drug loading pronouncedly affects the general nature of agglomerates. An expansion in drug loading has indicated expanded drug misfortune through the outside stage. On the off chance that the framework has insoluble diluent/excipient, solidified drug gets kept on its surface and creates the miniscular type of drug. The impact of drug content on tablet ability and drug discharge attributes of bromhexineHCl-powder agglomerates arranged by Crystallo-co-agglomeration has been contemplated by Jadhav [26,27]. It has been accounted for that in spite of known poor cohesively of BXH, its part in enhancing rigidity has been set up at higher drug stack in agglomerates. The impact of elasticity in accomplishing broadened drug discharge has likewise been underlined. At long last, it was inferred that the drug content deciding elasticity of smaller is in charge of the accomplishment of expanded drug discharge from minimized.
f. Drug loss to supernatant
The drug loss to supernatant determines the drug entrapment and the overall efficiency of the CCA process. During the agitation process, maximum crystallization and agglomeration of drug/s should be ensured. Attempts have been made to reduce the drug loss by processing the contents at low temperature, pH adjustments, and addition of solubility suppressants to the external phase [19].
g.Yield of the process
The process yield depends on the amount of crystallisation occurred from the good solvent as well as the extend ofagglomeration from the bridging liquid. Thus the selection of solvent system holds an important role in the process yield of Crystallo co agglomeration. The solubilisation of drug is determined by the good solvent and the crystallisation is done by the non solvent. The bridging is an interparticular interaction. Hence for obtaining desirable yield proper selection of solvent system is recommended [28].
Process Variables
a. Agitation
The primary capacity of agitation is emulsification or scattering. The size, shape, sphericity and quality of the agglomerates were influenced by agitation. Rapid fomentation may bring about expanded sphericity and diminished quality of the agglomerates. It was additionally discovered that with the expansion in speed of agitation, it might diminish the time required for the procedure and it decreases the agglomeration [4].
b. Time required for batch processing
The season of agitation chooses the fulfilment of agglomeration. Fragmented agitation prompts deficient blending of different ingredients, in this manner inadequate development of agglomerates. This likewise reduces the evaporation of organic solvents from the response vessel, while overabundance agitation results in fine arrangement [29]. The end purpose of agglomeration assurance is basic in CCA. It can be discovered by judging the clarity of the supernatant, leftover natural solvent and achievement of appropriate agglomerate size [4].
c. Evaluation Techniques Used for the Crystallo co Agglomerates
i. Surface Topography [30]
In surface topography studies, the agglomerates were captured utilizing an optical magnifying lens with camera at its unique amplification. The zone (An) and edge (P) of the agglomerates were gotten from tracings of augmented photomicrographs. This can be utilized to ascertain shape factor (S).
S = P2 actual/ (4nA actual).
Twenty granules per batch can be evaluated.
ii. Differential scanning calorimetry
Differential scanning calorimetry incorporates the estimation of changes that happen when heat to the example while they are subjected to controlled temperature programming [31].
DSC thinks about the thermo tropic conduct of particles. The procedure like crystallization can be watched utilizing DSC. At the point when temperature of an example is expanded step by step the consistency of undefined solids will diminish. At a specific point the atoms may achieve adequate vitality in order to mastermind themselves into gems. This temperature is crystallization temperature (Tc). This procedure of change of a shapeless strong into a crystalline strong is an exothermic procedure and is shown in the thermogram (diagram acquired) as a pinnacle. This guideline is utilized as a part of the investigation of crystallo co agglomerates. Thermograms of medications, polymers and agglomerates are performed utilizing a differential scanning calorimetry. The DSC temperature ought to be adjusted. Precisely measured examples are fixed in an aluminium cauldron. The framework ought to be cleansed with nitrogen gas.
iii.Micromeritics Properties
With a specific end goal to get consistency in tablet weight, the agglomerates must stream and pack easily into the die cavity of the tabletting punching machine.. Along these lines, micromeritics properties are assessed for molecule plan of agglomerates for guide pressure to enhance the stream and pressing properties of pharmaceutical powders. Agglomerates are assessed for flow ability by Angle of repose utilizing the settled fixed funnel free standing cone method. Molecule estimate circulation is considered by sifter investigation. In this agglomerates held on strainers are weighed and the subsequent information is utilized to acquire the mean geometric diameter by plotting the aggregate rate undersize versus the normal size on log average particle size on log probability paper. Qualities for angle of repose = 30 in. dictate free streaming material while angle of repose = 40 demonstrate poor streaming material [3].
iv. Sphericity Determination
Sphericity of the agglomerates is the most imperative attributes and diverse procedures have been utilized to decide it. For satisfactory nature of agglomerates the shape factor ought to be between 1 and 1.2 while 0.6 estimation of shape factor describes great sphericity of agglomerates. The shape factor is dictated by assessing the sum by which the anticipated picture of particles go amiss from a circle and ascertained by methods for the anticipated zone of the agglomerates and its periphery Photomicrographs got by optical magnifying instrument are utilized to figure the region (An) and edge (P) of agglomerates. The molecule shape of both groups is estimated by estimating the shape factor, circularity factor and length-to-width proportion [32].
Shape Factor (P) = P1/2/P'
Where P = 2n (A/7) 1 /2
Circularity Factor (5) = (P) 2 / (12.56*A)
v. Crushing Strength
Crushing strength is assessed to decide mechanical strength since it specifically mirrors the mechanical strength of smaller or tablet. Agglomerates ought to have great mechanical strength on account of expanded in. Trap particle constrain within the agglomerated crystals [33]. Crushing strength of agglomerates is controlled by mercury load cell method. Agglomerates of various bunches are arbitrarily tested and subjected to crushing strength assurance and normal was taken. The logarithmic relationship was set up between crushing strength (CS) and agglomerate size as appeared by the accompanying equation [34]:
log CS=m log D+C
Where m is slope, D is agglomerate diameter and C is intercept calculated by regression analysis of the log D vs. log CS.
vi. Dissolution Studies
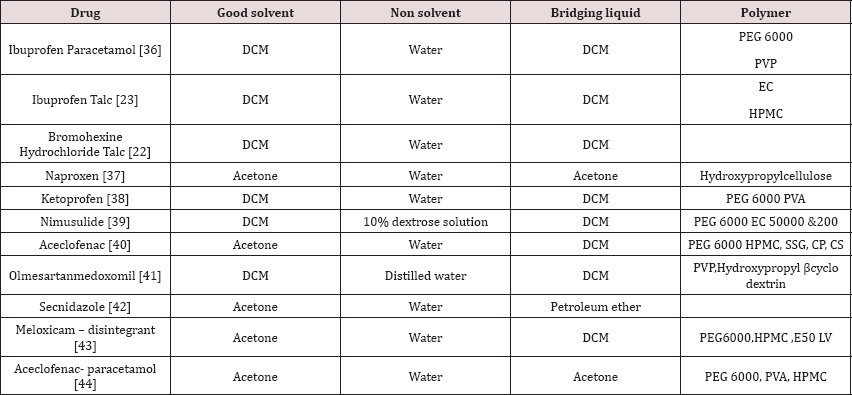
The agglomerate framed by CCA is then subjected to dissolution studies keeping in mind the end goal to understand the pharmacokinetics and in this way bioavailability of the acquired item. U S P suggests dissolution studies for the dissolution studies, which incorporate rotating basket type apparatus, rotating paddle apparatus, reciprocating cylinder apparatus, flow through cell apparatus, and paddle over cell apparatus, cylinder apparatus, and reciprocating plate apparatus. Any of the above gadgets can be utilized for dissolution studies relying on the type of tablet assessedi. e. regardless of whether traditional, controlled release and so on (Table 1)[35-44].
Table 1: Works Held On Cca till this Date.
Conclusion
Crystallo-co-agglomeration method has been created to overcome the limitations of spherical crystallization and utilized for estimate amplification of all, low dose, high dose, ineffectively compressible drugs and combination of drug with or without diluent. It includes combination of crystallization and agglomeration utilizing bridging liquid and crystallization medium. This system is straightforward completed in a solitary advance and more beneficial because of less number of unit tasks and economic as far as handling cost. Legitimate choice of solvent, bridging liquid and diluent can expand the drug discharge and enhance disintegration, absorption and bioavailability of drug and lessen localized toxicity. From late investigations, it can be concluded that this method speaks to a proficient method for creating spherical agglomerates with enhanced micromeritics, mechanical and compressional properties in outline of numerous unit particulate drug conveyance frameworks. There is a wide scope of research around there and it is continuously drawing in the in the rest of specialists everywhere throughout the world.
References
- Leom L, Herbert L (2013) The Theory and Practice of Industrial Pharmacy. 4th edn. New Delhi: CBS Publishers and Distributers, India.
- Shangraw RF (1989) Compressed Tablets by Direct Compression Granulation Pharmaceutical Dosage Forms: Tablets 1 (2nd edn). USA: Marcel Dekker.
- Ankita C, Pramod Kumar S, Mayank B (2011) A review on recent advancement in crystallo-co-agglomeration. Advances in biological research 5(6): 273-281.
- Christy MJ, Lekshmi P, Licey C, Bijin (2013) Crystallo-Co-Agglomeration: An Innovative Technique for Size Enlargement and Improved Flow Properties of Powders 1(2).
- Tapia C, Buckton G, Newton J (1993) Factors influencing the mechanism of release from sustained release matrix pellets produced by extrusion- spheronization. Int J Pharm 92: 211-218.
- Neau SH, Chow MY, Durrani MJ (1996) Fabrication and characterization of extruded and spheronized beads containing carbopol 974P NF resin. Int J Pharm 131: 47-55.
- Goskonda SR, Hileman GA, Upadrashta SM (1994) Controlled release pellets by extrusion-spheronization. Int J Pharm 111: 89-97.
- Jorgensen K, Christensen FN, Jacobsen L (1997) Dissolution stability of multiparticulate controlled release tablets. Int J Pharm 153: 1-11.
- Umprayn K, Chitropas P, Amarekajom S (1999) Development of terbutalinesulfate sustained-release coated pellets. Drug DevInd Pharm 25(4): 477-491.
- Kojima M, Nakagami H (2002) Development of controlled release matrix pellets by annealing with micronized water-insoluble or enteric polymers. J Control Release 82: 335-343.
- Bechgaard H, Hegermann NG (1978) Controlled-release multiple-units and single-unit doses: a literature review. Drug DevInd Pharm 4: 53-67.
- Tang SK, Chan LW, Heng WS (2005) Coating of multiparticulates for sustained release. Am J Drug Deliv 3(1): 17-28.
- Freely LC, Khosla R, Davis SS (1987) Investigating the gastric emptying of different sizes of non- disintegrating tablets in humans using gamma scintigraphy. J Pharm Pharmacol 39: 31-37.
- Bechgaard H (1982) Critical factors influencing gastrointestinal absorption: What is the role of pellets? Acta Pharm Technol 28: 149-157.
- Devereux JE, Newton JM, Short MB (1990) The influence of density on the gastrointestinal transit of pellets. J Pharm Pharmacol 42: (7) 500501.
- Clarke GM, Newton MJ, Short MD (1993) gastrointestinal transit of pellets of differing size and density. Int J Pharm 100: 81-92.
- Tuleu C, Andrieux C, Boy P, Chaumeil JC (1999) Gastrointestinal transit of pellets in rats: Effect of size and density. Int J Pharm 180: 123-131.
- Chaudhari A, Sharma PK, Bansal M (2011) A Review on Recent Advancements in Crystallo Co Agglomeration. Advances In Biological Researches 1: 273-281.
- Paradkar AR, Pawar AP, Jadhav NR (2010) Crystallo-co-agglomeration: A novel particle engineering technique. Asian Journal of Pharmaceutics 4(1).
- Kawashima Y, Okumura M, Takenaka H (1982) Spherical crystallization: Direct spherical agglomeration of salicylic acid during crystallization. Science 216(4550): 1127-1128.
- Limzerwala RB (1996) Optimization of the wet agglomeration of talc [Mastersthesis], University of Pune, India.
- Jadhav NR (1998) Studies on agglomerates of bromhexine hydrochloride loaded talc prepared by crystallo-co-agglomeration [master's thesis], University of Pune, India.
- Pawar AP, Paradkar AR, Kadam SS, Mahadik KR (2004) Agglomeration of ibuprofen with talc by novel crystallo-co-agglomeration technique. AAPS PharmSciTech 5: 30-35.
- Ganjian F, Cutie AJ, Jochsaberger T (1980) In vitro adsorption studies of cimetidine. J Pharm Sci 69: 352-353.
- Jadhav NR (2009) Studies on applications of crystallo-co-agglomeration technique in pharmaceutical formulations [doctoral thesis], BharatiVidyapeeth University, Pune, India.
- Sanghavi NM, Sivanand R, Kotwaney HN (1979) Dissolution pattern of miniscularsulfisoxazole. Ind J Pharm Sci 41: 116-117.
- Alsaidan SM, Abdulhakeem AA, Eshra AG (1998) Improved dissolution rate of indomethacin by adsorbents. Drug DevInd Pharm 24: 389-394.
- Pawar AP (1999) Studies on spherical crystallisation of various pharmaceuticals [Doctoral thesis], University of Pune, India.
- Pawar AP, Paradkar AR, Kadam SS, Mahadik KR (2007) Effect of Polymers on Crystallo-Co-Agglomeration of Ibuprofen-Paracetamol: Factorial Design. Indian J Pharm Sci 69(5): 658-664.
- Parida R (2010) Evaluation Parameters for Spherical Agglomerates Formed by Spherical Crystallization Technique. Int J Pharm Biol Sci 1(3): 1-10.
- Adhikrao Yadhav, Venkat Yadhav (2010) Directly Compressible RoxithromycinRecrystallised Agglomerates By Solvent Change Method. Der Pharmacia Lett 2(1): 25-40.
- Siddaramaiah RN (2010) Design and evaluation of controlled release of piroxicam from pellets of microcrystalline cellulose and HPMC blends .International J Pharm Tech Res 2: 1465-1473.
- Goyal K, N Sharma, PK Sharma (2010) Spherical crystallization: A method for improving powder and tablet characteristics. Der Pharmacia Lett 2: 246-254.
- Jarosz PJ, EL Parrott (1983) Comparison of granule strength and tablet tensile strength. S Pharmaceutical Sci 72(5): 530-535.
- Bhrahmankar DM, Jaiswal SB (2009) Biopharmaceutics And Pharmacokinetics-A Treatise, VallabhPrakashan, (2 nd edn) pp. 327-331.
- Pritish K, Sunil D, Inayat BP, Harsha S (2012) Preparation of spherical crystal agglomerates via crystallo-co- agglomeration technique. Digest J Nanomate Biostr 7(3): 1123-1236.
- Maghsoodi M, Taghizadeh O, Martin GP, Nokhodchi A (2008) Particle Design Of Naproxen- Disintegrant Agglomerates For Direct Compression By Crystallo Co Agglomeration Technique. Int J Pharm 351: 45-54.
- Vikash C, Rajesh M (2008) Tailoring Of Ketoprofen Particle Morphology Via Novel Crystallo Co Agglomeration Technique To Obtain Directly Compressible Material. Asian J Pharm 2: 61-67.
- 39. Bhattacharya SP, Bhattacharya I, Patro N (2010) Standardisation And Optimization Of Micromeritic Properties of Nimesulide For ProcessingInto Tablet Dosage Form By Crystallo Co Agglomeration Technology. ASJP 4: 24-27.
- Md S, Khan KAA, Doddayya H, Reddy SR, Udupi RH (2011) Particle Design Of Aceclofenac- Disintegrant Agglomerates For Direct Compression By Crystallo Co Agglomeration Technique. Asian J Pharm Technol 1: 40-48.
- Yadav AA, Yadav DS, Karekar PS, Pore YV, Pankaj G (2012) Enhanced Solubility And Dissolution Rate Of OlmesartanMedoxomil Using Crystallo Co Agglomeration Technique. Der Pharmacia Sinica 3: 160169.
- Raval MK, Sorathiya KR, Chauhan NP, Patel JM, Parikh RK, et al. (2013) Influence of polymers/excipients on development of agglomerated crystals of secnidazole by crystallo-co-agglomeration technique to improve processability. Drug DevInd Pharm 39(3): 437-446.
- Shashank A, Prakash GS, Sanjay J (2012) Particle Design Of Meloxicam Disintegrant Agglomerates For Fast Dissolution And Direct Compressibility By Crystallo Co Agglomeration Technique. Novel SciInt J Pharm Sci 1: 289-297.
- Prathish K, Dewani S, Pathana IB, Sonaye H (2012) Preparation Of Spherical Crystal Agglomerates Via Crystallo Co Agglomeration Technique. Digest J NanomatBiostr 7: 1223-1236.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...

.png)








