Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-6636
Research Article(ISSN: 2637-6636) 
Periodontitis in the Developmental Age: Pathogenesis, Epidemiology, Differential Diagnosis and Treatment. A Narrative Review Volume 3 - Issue 5
Sergio Mazzoleni, Francesco Saverio Ludovichetti*, Christian Bacci, Andrea Zuccon, Antonio Gracco and Edoardo Stellini
- Department of Neurosciences, Dentistry section, University of Padova, Italy
Received: January 31, 2020; Published: February 13, 2020
*Corresponding author: Francesco Saverio Ludovichetti, Resident, Department of Neurosciences, Dentistry Section, University of Padova, Via Giustiniani 2, 35131 Padova, Italy
DOI: 10.32474/IPDOAJ.2020.03.000173
Abstract
Objective: The purpose of this study is to deepen the description of aggressive periodontitis in developmental age patients going through pathogenesis, epidemiology, diagnosis, treatment and differential diagnosis.
Methods: The database searching was performed on PubMed and Scopus using the following keywords: “prepubertal periodontitis, aggressive periodontitis, periodontitis in children, periodontal disease in children and adolescents, early-onset periodontitis”. Both clinical, laboratorial and review studies were taken into consideration.
Results: Aggressive periodontitis affects a low percentage of children, and in those patients. Actinomycetemcomitans is the main bacterium identified in affected sites. Moreover, it has been found that Genetics plays a fundamental role in development and progression, which is important to distinguish the various oral manifestations excluding the possibility that they are a consequence of systemic pathologies. Although it mainly affects young patients, the treatment does not differ from that applied in adult subjects and it consists of a causal therapy, a mechanic and a pharmacological one, in particular the antibiotics associated with professional hygiene has shown very satisfactory results.
Discussion: Given the great variability of oral manifestation symptoms, there is no specific criterion for defining a high-risk group at prepubertal age, further research is needed to identify a robust set of genetic, microbiological and host factors markers that may facilitate the diagnosis of the disease.
Keywords: Aggressive periodontitis; periodontal disease; developmental age; differential diagnosis
Introduction
Periodontal disease is one of the most widespread diseases in the world and is, as a prevalence, immediately after diseases such as diabetes and hypertension [1]. Its clinical aspects have long been analyzed since it constitutes a worldwide problem. The pathology affects subjects of every race, sex and age and also some risk factors are needed in addition to the individual susceptibility for it to develop. Even today there are not enough scientific certainties to establish the behavior of periodontal disease in the age group that affects children and young adults. In any case, we proceeded through a search in the literature with the attempt to deepen its clinical aspects, etiology, diagnosis, predisposing factors and treatment. Periodontal diseases, despite being widespread especially in the adult population, are not so rare even among young people [2]. For example, gingivitis affects over 70% of children over the age of seven [3]. Bimstein in 1991 underlined the importance of prevention, early diagnosis and treatment of periodontal diseases in children and adolescents because they have a high severity and prevalence [4] and furthermore, the oral-dental incipient pathologies on small subjects can develop into periodontal diseases in adults. However, the degree of extension and destruction of periodontitis also responds to a personal predisposition to the disease. The severity index of periodontal disease can also be mediated by the presence of some systemic diseases such as hypophosphatasia or leukocyte deposition deficiency [5].
According to Lamster IB and Pagan M, the metabolic syndrome (MetS) that is a spectrum of conditions that include dysglycemia, visceral obesity, atherogenic dyslipidemia (high triglycerides and low levels of high-density lipoprotein) and hypertension are associated with periodontal disease. They believe that this relationship is the result of systemic oxidative stress and an exuberant inflammatory response. Evidence suggests that periodontal therapy may reduce serum levels of inflammatory mediators so periodontitis treatment could become part of the metabolic syndrome therapy [6]. Among the various types of periodontitis, one of the less studied ones is aggressive periodontitis. The manifestations of aggressive periodontitis in young people have many controversial sides and consequently, the present study proposes to look for some clarifications regarding the aspects of the pathology
Methods
For this narrative review, the database searching was performed on PubMed and Scopus using the following keywords: “prepubertal periodontitis, aggressive periodontitis, periodontitis in children, periodontal disease in children and adolescents, early-onset periodontitis”, the investigation then focused on evaluating the specific aspects of aggressive periodontitis in children, consequently the following words have been introduced: “epidemiology, classification, progression, treatment, diagnosis”. Clinical and laboratorial studies were taken into consideration as well as literature reviews. The last database search was performed in September 2019.
Discussion
Aggressive periodontitis
Aggressive periodontitis can occur in several forms that is linked to a few dental sites or in a generalized sense. The first form usually affects smaller subjects and is connected to lesions of the first molars or incisors or both in the presence of little plaque and tartar, and the second form concerns post-puberty subjects with more permanent teeth. Very often, if left untreated, the localized forms evolve into general forms with the risk of a total compromise of the dental apparatus. Sometimes the signs of inflammation are not so easily detectable, which is why a child or teenager on the first visit should always be subjected to a more in-depth analysis by using probes to detect probing depth and radiological investigations. There are some mechanisms that regulate evolution in the various age groups, and the different anatomies and physiologies can modify the development of periodontitis. In particular, there are many structural inequalities between adults and children. The gingiva in the young is more vascularized, has less connective tissue around the deciduous teeth, the epithelium is thinner and less keratinized, characteristics that can expose to less defense to attacks bacterial. It can be said that a child, due to its thinness of tissues, is more exposed to risk and moreover a greater vascularization allows an easier transit of inflammation mediators and bacteria [7]. The typical signs that indicate the presence of a problem and that should alarm the parents are bleeding gums during home hygiene practices, swelling, halitosis accompanied by any recessions. Evidence shows that periodontal disease may increase during adolescence due to lack of motivation to practice oral hygiene but also due to changes related to puberty. Hormones such as progesterone, estrogen and testosterone cause greater blood circulation, greater sensitivity and greater response to any irritation, the gengiva are often red and swollen. Hormones are molecules with specific regulatory abilities and have powerful effects on the main determinants of development and on the integrity of the skeletal cavity including periodontal tissues [8].
Epidemiology
A 1987 study by Sweeney [9] evaluated alveolar bone loss
around primary teeth in a population of 2,264 children. Nineteen
patients (0.84%) showed periodontal bone destruction around
one or more primary teeth; in 2 of these patients, periodontal
disease was previously identified during clinical examinations.
The microbiological study also revealed a high prevalence of
Actinobacillus actinomycetemcomitans and Capnocytophaga.
Another study carried out by Bimstein [10] in 1994 verified
the prevalence of alveolar bone loss in a group of 317 5-yearold
New Zealand children. The results identified that there was
a questionable bone compromise in 8.5% of the children and a
defined bone loss of 2.1%. Darby et al in 2005 studied bone loss in
542 children aged between 5 and 12 years. Reading the patient’s
radiographs, each interdental site was evaluated as: no bone loss
and therefore distance from the amelite-cementitious junction
to the alveolar ridge of less than 2 mm, questionable bone loss
i.e. distance greater than 2 mm but less than 3 mm and bone loss
defined or distance greater than or equal to 3 mm.
The results showed that 61 (13%) children presented sites
with definite bone loss, 60 children had only a questionable bone
loss, 50 children had only a defined bone loss and 21 children had
both lesions. It was also found that children of Asian-Far Eastern
origin had a higher percentage of sites with bone loss than children
of Caucasian origin, 29.5% and 19.7%, respectively, but lower than
that of children of Middle Eastern origin (35.2%). In conclusion, the
present study showed that in the population studied, 26% had bone
loss but 13% had more severe and defined lesions [11]. The studies
described above thus show that the prevalence of periodontal
disease and in particular bone loss varies from 0.84% to 13%,
but in reality, the heterogeneity of such research and the lack of
standardization makes it clear how the results are discordant and
the prevalence remains mostly dubious.
Microbiology
There are some bacteria that mainly cause periodontal disease,
and these can be transmitted within the family where the contact
between subjects is very close; through the mother’s saliva, for
example, children may be exposed to risk. Much attention has been
paid to Actinomycetemcomitans as a species implicated in the
etiology of aggressive periodontitis. Its main virulence factor is a
leukotoxin capable of eliminating important cells of the immune
system. Genetic analyses have identified a population structure of
the clonal-type bacterium with evolutionary families corresponding
to serotypes. A particular highly leukototoxic clone (JP2) of
serotype b was discovered. Its characteristics are unique in fact that its increased leukototoxic activity is given by a deletion of 530
bases in the operon. The geographical mapping of the JP2 clone has
revealed that its colonization mainly concerns individuals of African
origin [12]. A study conducted by Burgess et al. in 2017 showed
the prevalence of the highly leukotoxic JP2 sequence compared to
the non-JP2 sequence of Aggregatibacter actinomycetemcomitans
within a group of 180 young African Americans aged between 5 and
25 years with and without localized aggressive periodontitis (LAP).
Subgingival plaque was collected from diseased sites, i.e. from
areas with probing depth greater than or equal to 5 mm that
presented bleeding and from healthy sites, i.e. from areas with
probing depth less than or equal to 3 mm that did not present
bleeding. Overall, 90 subjects (50%) tested positive for the JP2
sequence, 50 subjects (83.33%) with aggressive periodontitis
presented the sequence detected in 45 (75%) sick sites and 34
(56.67%) healthy sites [13]. Actinomycetemcomitans in general is
considered an opportunistic pathogen of the oral microbiome, in
fact many clonal types of the bacterium different from JP2 can be
isolated from healthy subjects, however, patients who present the
JP2 strain always show periodontal disease, so an etiological agent
is important for aggressive periodontitis in children, adolescents
and adults. In conclusion, there is a high risk for the development of
the disease in individuals colonized by the JP2 clone, furthermore
its transmission, as for other clonal types, occurs vertically by
close contact between people, indicating that subjects of the same
family may experience extrinsic routes of the subpopulation of the
bacterium [12].
Genetics
Genetics plays an important role in the appearance and severity of the disease, so if a person is diagnosed with aggressive periodontitis, it is also good to investigate the other members of the family in order to cure or prevent their appearance. A 1994 study by Mary L Marazita studied evidence of autosomal dominant inheritance and specific heterogeneity in aggressive periodontitis. Analyses were conducted on 100 families, and 104 subjects were diagnosed with aggressive periodontitis. Heterogeneity tests were used to compare the parameter estimates and the conclusions obtained in the black and non-black families. The results of the segregation analysis have verified that an autosomal dominant locus is sufficient to explain the patterns of disease transmission to the whole family. In conclusion, in the present study, we saw how the disease has a chance of appearing in the same branch of descent at 70% [13]. Family aggregation of aggressive periodontitis is not an unusual discovery. The conditions of development of this pathology can be more complex than simple Mendelian syndromes. Genetic studies indicate that there are several genetic variants expressing different forms of aggressive periodontitis, but currently it is not clear how many genes may be involved in these non-syndromic forms of disease [14]. It is important to remember that family models can also indicate exposure to common environmental factors within the same family. Therefore, the behavioral components shared by the same parental group must also be considered: education, socioeconomic status, oral hygiene, possible transmission of bacteria, diseases such as diabetes and environmental characteristics such as even passive smoking influence the susceptibility of the subject as risk factors. Another decisive reason for determining whether individuals develop periodontitis appears to be regulated by the way they respond to their microflora. Genetic factors also in this case modulate the way in which individuals interact with many environmental agents, including biofilm. The mutual influence of genetic and environmental factors, and not only of genes, determines the result, lifestyle factors open the way to the development of aggressive disease [15].
Treatment
When a child is diagnosed with aggressive periodontitis, prompt action must be taken to achieve maximum reduction of periodontal microorganisms with the aim of blocking the development of a more severe clinical picture. The treatment of aggressive disease in children and adolescents does not differ from the techniques applied to adults, in fact the etiology is always known to be bacterial regardless of age group. The treatment therefore should be based on the elimination of pathogens by professional hygiene, but, in reality, in these forms of periodontal disease given the high toxicity of the microorganisms, a systemic therapy with antibiotics must be associated to resolve the picture. The goal is to create a clinical condition that favors the maintenance of the greatest number of teeth for as long as possible. The initial phase of active treatment consists of mechanical cleaning, performed with or without the use of antimicrobial drugs.
The downsizing and smoothing of the roots have proved
effective in improving the clinical indices, but they do not always
guarantee long-term stability, which is why systemic antibiotics as
adjuvants for radicular treatment are to be administered during
therapy, and they are more effective than root resizing alone with
the additional application of local or antiseptic antibiotics [16].
A 2005 study by Guerrero et al. evaluated the systemic
administration of amoxicillin and metronidazole in non-surgical
therapy for the treatment of generalized aggressive periodontitis.
Forty-one systemically healthy subjects in whom the disease
was diagnosed were selected. Patients received non-surgical
treatment over a 24-hour period and one half received a course
of systemic antibiotic consisting of 500 mg of amoxicillin and 500
mg of metronidazole three times a day for 7 days while the other
group of subjects received placebo. After two and six months, they
were re-evaluated and the results were as follows: in patients on
antibiotic therapy in the 7 mm pockets there was a gain of 1.4 mm
and a recovery of bone equal to 1 mm in addition to the areas with
depths greater than or equal to 5 mm had a probing less than or
equal to 4 mm. Twenty five percent of sites in test patients had a
successful improvement in clinical attack, whereas for patients
treated only with placebo, the percentage of improved sites was
16% [17]. Another study by Kaner shows how the subgingival
application of chlorhexidine via a controlled release device
(CHX chip) does not improve the clinical outcome in generalized
aggressive periodontitis. The purpose of that study is to compare
whether the additional positioning of the CHX chip is as effective as the use of systemic antibiotics. A total of 36 patients were
diagnosed with aggressive periodontitis, one half was treated only
with slow-release chlorhexidine and the other half treated with
systemic antibiotic therapy. The subjects were re-evaluated 3 and
6 months after therapy, and it was shown that the level of clinical
attack, bleeding and probing depth had a significant improvement
in patients receiving amoxicillin and metronidaziol compared to
patients treated with the local application of antiseptics [18]. An
alternative method for the decontamination of periodontal sites
has been studied: photodynamic therapy. To reduce the excessive
use of antibiotics, new disinfection strategies have been sought.
Photodynamic therapy (PDT) or light-activated disinfection (LAD)
was first tested by Oscar Raab in the early 1900s.
For years it was abandoned due to the use of antibiotics, but it
has found a new application in the last decades both in the medical
field and in the dental field. The photodynamic reaction takes
advantage of the use of a photosensitizer (PS) and a light source
calibrated to specific wavelengths in the presence of oxygen. It
acts specifically against both Gram + and Gram- microorganisms
without causing any damage to the host cells. The toluidine blue is
very effective active against many bacteria including those involved
in periodontal disease. One example is Arweiler’s [19] research
in which the use of antibacterial photodynamic therapy (aPDT)
was studied in addition to mechanical scaling and root planning
therapy. The aim of that study was to evaluate the results following
non-surgical periodontal therapy and additional use of aPDT or
amoxicillin and metronidazole (AB) in patients with aggressive
periodontitis. Out of 36 patients treated with antibiotic therapy or
with two episodes of post-treatment photodynamic therapy, the
results after six months were the following: the probing depth was
found to be significantly reduced in both groups.
Despite this, the administration of amoxicillin and metronidazole
produced higher improvements than the existence of photodynamic
therapy, the number of pockets ≥7 mm was reduced from 141 to 3
after AB and from 137 to 45 after aPDT. Although both treatments
led to statistically significant clinical improvements, AB showed a
reduction in probing depth and a lower number of pockets ≥7 mm
compared to aPDT. In conclusion, photodynamic therapy associated
with the non-surgical periodontal therapy, despite giving favorable
results, cannot be considered a definitive alternative to the systemic
use of amoxicillin and metronidazole [20]. However, antibiotics
must be administered during or after mechanical therapy since
micro-organisms are particularly protected by biofilm in the
subgingival plaque. With regard to surgical treatment in patients
with aggressive periodontitis, it has been shown that it gives results
comparable to non-surgical treatment provided that correct oral
hygiene is maintained, that a rigorous maintenance program is
followed and that risk factors are kept under editable control [18].
Differential diagnosis
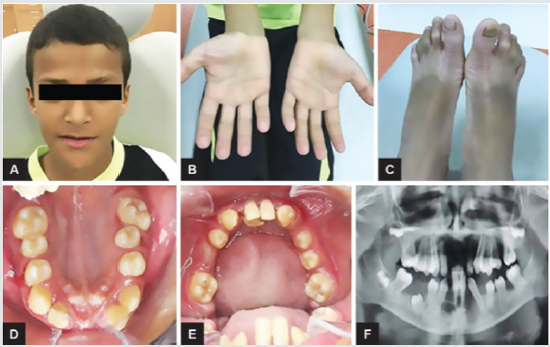
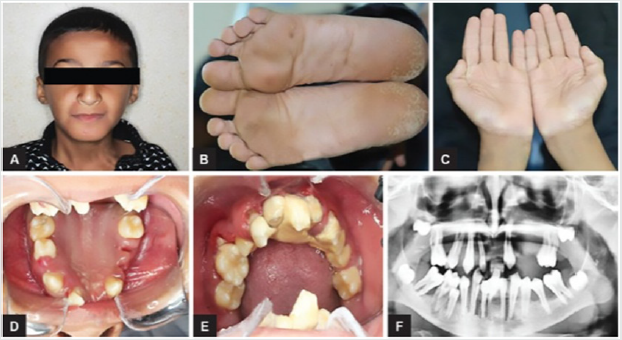
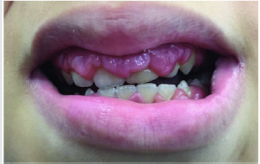
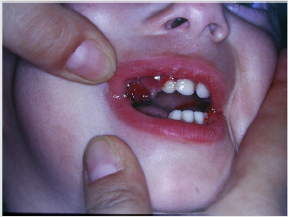
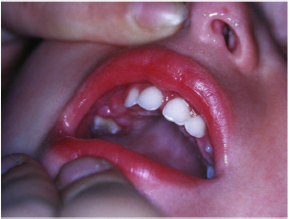
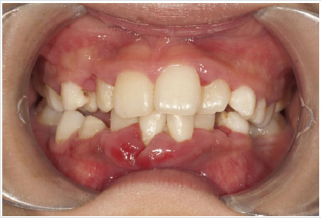
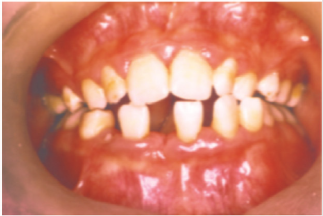
Periodontal disease is characterized by the imbalance between pathogens and host defenses leading to an inflammatory reaction around dental tissues. To diagnose aggressive periodontitis, it is necessary to investigate the health status of the subject and exclude the coexistence of systemic diseases. Some disorders can cause oral lesions clinically similar to those of aggressive periodontitis, but it is of fundamental importance to distinguish the various manifestations to make a correct diagnosis and intervene with the most appropriate treatment. Examples of significant conditions are AIDS, leukemia, diabetes or rare genetic disorders such as histiocytosis X and Papillon-Lefevre syndrome. The latter is a rare autosomal recessive disorder caused by mutations in the gene that codes for cathepsin C (dipeptidyl-peptidase I inhibitor). The syndrome is characterized by hyperkeratosis, destructive periodontitis that occurs from childhood, recurrent piogenic and systemic skin infections, susceptibility to bacterial infections and intra-cranial calcifications [21] (Figures 1&2). The prevalence is estimated to be between 1 / 250,000 and 1 / 1,000,000 subjects and is manifested in all ethnic groups. These dermatological features appear between the first year of life and 4 years and are accompanied by intraoral lesions that include gingival inflammation, mobility of the dental elements, even spontaneous bleeding and destruction of the periodontium. Patients with this syndrome show serious signs in the oral cavity until complete loss of deciduous bone, generating the normal appearance of the gum. However, with the eruption of permanent teeth, the form of aggressive periodontitis reappears. Any non-surgical but also surgical treatment is vain and almost always leads to partial or complete edentulism in the patient. The treatment is based on the intake of oral retinoids, which attenuate the palmoplantar keratoderma and slow down the lysis of the alveolar bone, and the skin lesions can also be treated with emollients in order to hydrate the affected area. Furthermore, good oral hygiene control, the use of mouthwashes and even antibiotics are recommended to slow the progression of periodontitis. Deciduous teeth or elements with excessive mobility must be extracted and eventually replaced by implants when the subject has completed growth.
The identification of the syndrome at an early age is something
multidisciplinary that can improve the patients’ prognosis [22].
Leukemia is a malignant neoplastic disease of white blood cells and
very often strikes in the pediatric age giving oral manifestations
prior to systemic onset [23]. Acute lymphoblastic leukemia (ALL or
ALL), specifically, develops when a cell destined to give rise to cells
of the immune system turns into a tumor and starts to multiply in
an uncontrolled way. Many studies show that acute lymphoblastic
leukemia is the most frequent tumor in children, representing
75% of all newly diagnosed leukemias and 25% of all childhood
malignancies [23]. The typical symptoms that characterize
lymphoblastic leukemia are fatigue, dyspnea, fever, pallor and
weight loss. Patients with this form of leukemia in the oral cavity
have pale mucous membranes and an important gingival bleeding
accompanied by lymphadenopathy in the head and neck region.
It has been shown that sometimes the initial sign of the disease
may correspond to a pericoronitis associated with a prolonged
contraction of the masticatory muscles. Numerous studies have
also reported a greater incidence of abnormalities in the oral cavity
such as the presence of large, irregularly shaped ulcers, halitosis
and a loose mucosa [24].
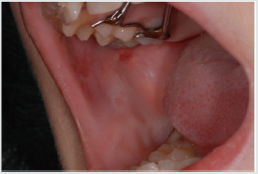
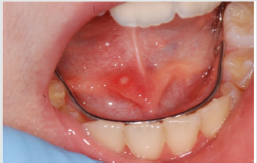
Acute myeloid leukemia (AML) is a disease that originates
from the bone marrow. The disease is more common in adults
over 60 years and infrequent before the age of 45. Patients with
AML have symptoms related to complications related to anemia,
neutropenia and thrombocytopenia, including weakness and
easy fatigue, infections of varying severity, gingival bleeding,
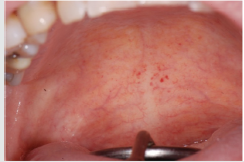
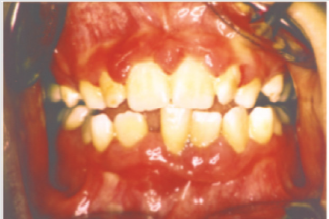
ecchymosis, epistaxis [25]. The oral examination can show pallor
of the mucosa (Figure 3), ulcerations (Figure 4), spontaneous
bleeding and bleeding (Figure 5) petechiae on gums, palate (Figure
6), tongue or lips, gingival hyperplasia (Figure 7) caused from
leukemic infiltration. Usually the lesions of the oral cavity are the
first manifestations of the disease, in particular, gingival swelling
represents 5% of the early complications [26]. In leukemic patients,
regardless of the form of the disease, oral manifestations occur as
initial evidence or of its recurrence. Symptoms mainly include gum
enlargement and bleeding, oral ulceration, petechiae, mucosal
pallor and oral infections. These lesions may be the result of
direct infiltration of leukemic cells or altered granulocyte function
[26]. With regard to oral hygiene, patients must undergo periodic
checkups and deplaquing or scaling sessions to reduce the level
of inflammation of the mucous membranes, local antiseptics or
antibiotics can be combined with active infections. Chemotherapy,
often used in the treatment of leukemia, also has consequences on
the subject that also affect the oral cavity. Often patients in therapy
are predisposed to the appearance of ulcers, lesions, infections and
are prone to have a partial xerostomia favoring plaque buildup.
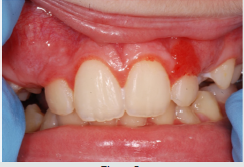
Scurvy is another disease that manifests itself with abundant gingival bleeding and which can be confused with aggressive periodontitis. The primary cause of scurvy is the insufficient intake of vitamin C mainly due to dietary imbalances. Childhood scurvy generally appears between the sixth and twelfth year of life. The child is easily irritated without appetite and fatigued. Sufferers of this disease develop anemia, weakness, fatigue, edema in some parts of the body, muscle pain in the lower limbs and ulceration of the gums (Figure 8) [27]. It occurs later as follicular hyperkeratosis and haemorrhage of the lower limbs, as well as bleeding in other areas such as the gingiva and joints [28]. If the disease is not treated it is potentially lethal due to important bleeding that can occur in the intracranial area or, due to the poor ability of the subject to heal due to open wound infections. At the oral level, the disease manifests itself in widespread hypertrophic areas of the violet-colored mucosa, a tendency for bleeding and the formation of hematomas. In small subjects, the symptoms that appear first are the pain and swelling of the joints accompanied by gingival hypertrophy [29].
Patients suffering from this pathology are administered quantities of vitamin C orally or through injections and, in a short time , all symptoms disappear [28] (Figure 9). Diabetes mellitus is a disease that can present in the oral cavity as aggressive periodontitis and be confused with this. It includes a group of chronic metabolic disorders that turn out to be an altered glucose tolerance or an altered metabolism of lipids and carbohydrates [30]. It has been shown by numerous researches that in diabetic children with poor metabolic control, there is a greater tendency for gingivitis [30]. In fact, the high levels of glucose in the blood cause changes in microcirculation, promote bacterial proliferation and interact with the response of the host. Hyperglycemia caused by diabetes mellitus alters the immune system and the increased availability of glucose in the oral cavity environment increases the proliferation of periodontopathic bacteria and causes marked oral inflammation (Figure 10). In patients with diabetes, a microangiopathy occurs and this change in the periodontium reduces the functions of the polymorphonuclear cells, the chemotaxis, the adherence, the phagocytosis, the use of oxygen and the elimination of antigens, thus favoring the progression of periodontal disease. Hyperglycemia also reduces the solubility of collagen, reduces the production of fibroblasts and causes an increase in the levels of pro-inflammatory mediators responsible for the destruction of connective tissues. Changes to collagen metabolism result in accelerated degradation of both non-mineralized connective tissue and mineralized bone.
Even the saliva undergoes both qualitative and quantitative changes. Often in subjects with diabetes, the salivary flow is reduced leading to a further development of the bacterial species [31]. In diabetic patients, periodontal disease develops at a younger age than the healthy population and periodontal impairment usually occurs in adolescence but sometimes earlier in children with diabetes [32]. In the oral cavity, there is therefore an edematous and very inflamed gingiva, bleeding when the probe passes and bone resorption can occur especially in cases of poor metabolic control (Figure 11) [33], but in patients with a good diet with good glycemic supervision and good oral hygiene, do not show evident alteration in the mucosa (Figures 12&13). There is then a relationship between higher levels of plaque and a higher incidence of gingivitis in children with diabetes, moreover, the differences in oral microflora and the impact of metabolic control of diabetes on periodontal health have indicated a higher risk of periodontitis in children with type 1 diabetes [34]. In conclusion, when you are confronted with a child who has an oral situation of persistent inflammation, you need to perform more specific tests to understand if it is periodontitis as a manifestation of systemic pathology or aggressive periodontitis itself. The dentist or hygienist is usually the first to diagnose some diseases due to the involvement of the periodontium, and it is important to have multidisciplinary management to try to minimize the physical, psychological and social effects of the patient at an early age.
Conclusion
In conclusion, the review of the literature shows that periodontal disease does not only affect adults but, even if less frequently, it also involves children and adolescents. The forms of aggressive disease show a family aggregation, cause an important and rapid destruction even in the absence of local irritative factors and occur in systemically healthy subjects. It has been seen that particularly virulent bacteria trigger the process of bone destruction in the disease, the main micro-organism involved is Actinomycetemcomitans, which produces powerful leucotoxins that can also severely damage the subject’s immune system cells. Some clonal types of the bacterium are very pathogenic, and JP2 is always isolated from subjects suffering from aggressive periodontitis indicating that it is an important etiological agent. Regarding the epidemiological aspects, the statistics show very variable data also depending on the country, and so far the prevalence of aggressive periodontitis is not known exactly but it can be said that it occurs mainly in subjects of African descent and in individuals who are predisposed from the genetic point of view.
To date there is no specific criterion for defining a high-risk group for prepubertal pathology, and further research is needed to identify a robust set of genetic, microbiological risk markers and host factors that favor a diagnosis of the disease in association with young people and adolescents. The identification of aggressive periodontitis can be implemented through periodontal screening associated with radiographs, and the routine use of BPE can be helpful for early diagnosis. The treatment consists of the same methods applied also to adult patients, that is to say a causal therapy, a mechanic and a pharmacological one, in particular the studies have shown that antibiotics associated with professional hygiene in patients with aggressive periodontitis give very satisfactory results. If, despite careful treatment and hygiene, a child continues to have periodontal problems, it is necessary to investigate the general health with more specific examinations. A form of periodontal injury in a young person can also be a symptom of systemic diseases that are extraneous to the parent’s awareness and early diagnosis can become vitally important. Aggressive periodontitis, although infrequent, is not to be underestimated and it is important to take children to regular checkups and scaling sessions. Preventing and diagnosing problems early is the key to success.
References
- Cortellini P, Carrassi A, de Sanctis M, Tonetti M (2006) Sidp Period-medicine project: periodontal diseases and systemic diseases. Italian Society of Periodontology.
- Tae Ju Oh, Robert Eber, Hom Lay Wang (2008) Periodontal diseases in the child and adolescent. Journal of Clinical Periodontology 29(5): 400-410.
- Page RC, Schroeder HE (1982) Prevalence, severity, and progression in periodontitis in man and other animals. A comparative review.
- Bimstein E, Delaney JJ, Sweeney EA (1988) Radiographic assessment of the alveolar bone in children and adolescents. Pediatr Dent 10(3): 199-204.
- Albandar JM, Rams TE (2002) Risk factor four periodontitis in children and young person. Periodontology 29: 207-222.
- Lamster IB, Pagan M (2017) Periodontal disease and the metabolic syndrome. Int Dent J 67(2): 67-77.
- Haubek D (2010) The highly leukotoxic JP2 clone of Aggregatibacter actinomycetemcomitans: evolutionary aspects, epidemiology and etiological role in aggressive periodontitis. APMIS Suppl 113(130): 1-53.
- Ainamo J, Nordblad A, Kallio P (1984) Use of the CPITN in populations under 20 years of age. Int Dent J 34(4): 285-291.
- EA Sweeney, GAP Alcoforado, S Nyman, J Slots (1987) Prevalence and microbiology of localized prepubertal periodontitis. Oral Microbiology and Immunology 2(2): 65-70.
- E Bimstein, ET Treasure, SM Williams, JG Dever (1994) Alveolar bone loss in 5‐year‐old New Zealand children: its prevalence and relationship to caries prevalence, socio‐economic status and ethnic origin. J Clin Period 21(7): 447-450.
- Ivan B, Darby Janet Lu Hanny Calache (2009) Radiographic study of the prevalence of periodontal bone loss in Australian school‐aged children attending the Royal Dental Hospital of Melbourne. J Clin Periodontol 32(9): 959-965.
- Burgess D, Huang H, Harrison P, Aukhil I, Shaddox L (2017) Aggregatibacter actinomycetemcomitans in African Americans with Localized Aggressive Periodontitis. JDR Clin Trans Res 2(3): 249-257.
- Mary L, Marazita John A, Burmeister John C, Gunsolley Thomas E, Koertge Kim Lake, et al. (1994) Evidence for Autosomal Dominant Inheritance and Race‐Specific Heterogeneity in Early‐Onset Periodontitis. J of Periodontol 65(6): 623-630.
- Mario Taba, Sergio Luis Scombatti de Souza, Viviane Casagrande Mariguela (2012) Periodontal disease: a genetic perspective. Braz Oral res 26(1): 32-38.
- De Carvalho FM, Tinoco EM, Govil M, Marazita ML, Vieira AR (2009) Aggressive periodontitis is likely influenced by a few small effect genes. J Clin Periodontol 36(6): 468-473.
- Teughels W, Dhondt R, Dekeyser C, Quirynen M (2014) Treatment of aggressive periodontitis. Periodontol 2000 65(1): 107-133.
- Guerrero A, Griffiths GS, Nibali L, Suvan J, Moles DR, et al. (2005) Adjunctive benefits of systemic amoxicillin and metronidazole in non-surgical treatment of generalized aggressive periodontitis: a randomized placebo-controlled clinical trial. J Clin Periodontol 38(1): 43-49.
- Kaner D, Bernimoulin JP , Hopfenmüller W , Kleber BM , Friedmann A (2007) Controlled-delivery chlorhexidine chip versus amoxicillin/metronidazole as adjunctive antimicrobial therapy for generalized aggressive periodontitis: a randomized controlled clinical trial. J Clin Periodontol 34(10): 880-891.
- Arweiler NB, Pietruska M, Pietruski J, Skurska A, Dolińska E, et al. (2014) Six-month results following treatment of aggressive periodontitis with antimicrobial photodynamic therapy or amoxicillin and metronidazole. Clin Oral Investig 18(9): 129-135.
- Güncü GN, Tözüm TF, Caglayan F (2005) Effects of endogenous sex hormones on the periodontium-review of literature. Aust Dent J 50(3): 138-145.
- Fageeh HN (2018) Papillon Lefèvre Syndrome: A Rare Case Report of Two Brothers and Review of the Literature. Int J Clin Pediatr Dent 11(4): 352-355.
- Fawad Javed, Achint Utreja, Fernanda O, Bello Correa, Mansour Al Askar, et al. (2012) Oral health status in children with acute lymphoblastic leukemia. Critical Reviews in Oncology/Hematology 83(3): 303-309.
- Kholoud A Lowal, Nader Ahmed Alaizari, Bassel Tarakji, Waleed Petro, Khaja Amjad Hussain, et al. (2015) Dental considaration for leukemic pediatric patients: an updated review for general dental practitioner. Mater Socio med 27(5): 359.
- Anirudhan D, Bakhshi S, Xess I (2008) Etiology and outcome of oral mucosal lesions in children on chemotherapy for acute lymphoblastic leukemia. Indian Pediatr 45(1): 47-51.
- Francisconi CF, Caldas RJ, Oliveira Martins LJ, Fischer Rubira CM, da Silva Santos PS (2016) Leukemic Oral Manifestations and their Management. Asian Pac J Cancer Prev 17(3): 911-915.
- Parveen Akhtar, Kiran Mushtaq Toor, Sumaira Khalil, Huma Saleem Khan, Saima Shabbi (2016) Scurvy“in the Land of Citrus Fruits”. Journal of Islamabad Medical & Dental College (JIMDC) 5(3).
- Mintsoulis D, Milman N, Fahim S (2016) A Case of Scurvy-Uncommon Disease-Presenting as Panniculitis, Purpura, and Oligoarthritis. J Cutan Med Surg 20(6): 592-595.
- Alqanatish JT, Alqahtani F, Alsewairi WM, Al Kenaizan S (2015) Childhood scurvy: an unusual cause of refusal to walk in a child. Pediatric Rheumatology 13:23.
- (2005) American Academy of Periodontology. Diabetes and periodontal diseases J Periodontol 2000.
- Firatli E, Yilmaz O, Onan U (1996) The relationship betweenclinical attachment loss and the duration of insulin-dependent diabetes mellitus (IDDM) in children and adolescents. J Clin Periodontol 23(4): 362-366.
- Salvi GE, Carollo Bittel B, Lang NP (2008) Effects of diabetes mellitus on periodontal and peri-implant conditions: update on associations and risks. Journal of Clinical Periodontology 35(8 Suppl): 398-409.
- Gülnur Emingil, Sükran Darcan, Ahmet Keskinoglu, Necil Kütükçüler, Gül Atilla (2001) Localized Aggressive Periodontitis in a Patient with Type 1 Diabetes Mellitus: A Case Report. Journal of Periodontology 72(9): 1265-1270.
- Marta Novotna, Stepan Podzimek, Zdenek Broukal, Erika Lencova, Jana Duskova (2015) Periodontal Diseases and Dental Caries in Children with Type 1 Diabetes Mellitus. Mediators Inflamm 2015(51): 379626.
- Jenkins WMM, Papapanou PN (2001) Epidemiology of periodontal disease in children and adolescents. Periodontol 2000 26: 16-32.
Editorial Manager:
Email:
pediatricdentistry@lupinepublishers.com

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...