
Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-6636
Mini Review(ISSN: 2637-6636) 
Electric Pulp Testing in Children During Permanent Teeth Apexes Formation Volume 4 - Issue 4
PetrikasA*and Letunovskaya SA
- Department of Therapeutic Stomatology, Tver State Medical University, Russia
Received: August 13, 2020; Published: August 20, 2020
*Corresponding author: Petrikas A, Department of Therapeutic Stomatology, Tver State Medical University, Russia
DOI: 10.32474/IPDOAJ.2020.04.000194
Mini Review
Pain sensation is evolutionarily the primary basis of sensitive human activity, and it is quite stable. The stability of electrodontometry (EOM) indicators is demonstrated by a narrow zone of the pain threshold of 2-6 mkA of healthy front teeth, which was first evaluated by Rubin L. R. in 1953. This value was confirmed by many domestic researchers [1,2]. Rubin tried to extend it to all teeth, but the indicators of the lateral teeth, although stable, are 5-15 mkA higher [3-5]. The most important feature of the sensitivity of the dental pulp is that it is represented by an exceptional pain [6,7]. Its sensitivity is very high -2-6 mkA- and sharply differs from the sensitivity of the skin and all surrounding tissues, including the gums and periapical tissues. It is located at the level of 100-200 mkA. The pulp, perceiving tactile, thermal and electrical irritation, translates it into a painful sensation. In the human tooth, the circulatory and nervous systems of the pulp are separated as a thin soft string and are protected from external influences by a thick tube of enamel and dentin. The nerves in the pulp are 20.5% by weight.The pulp is represented by two main pain points that determine pain sensitivity. In the crown pulp, these are odontoblasts and dentine tubes with 1-2 mm of nerve branches of A-Delta fibers entering them. At the root it is a narrow thin apical part of the canal, where the pulpar nerves of the pulp core from C- fibers are tightly placed together with the blood vessels. Secondary cement is also involved in creating the apical opening, preventing myelination of the nerve tissue there[8]. The presence of myelin A-Delta fibers in the apex has not been proven. Pain fibers A-Delta are slightly myelinated, located in the crown, have a higher rate of conduction (dentin pain). They respond to the pulse current as an acute pain sensation. C-fibers transmit late, dull and not always clear pulp pain. It is interesting that all foreign authors use A-Delta fibers [6], to consider themechanisms of electrodontometry (EOM) from the standpoint of the theory of Branstrom, with an emphasis
on EPT (Electrical pulpal test) as dentin stimulation. Foreign researchers exclude the reaction of pain by C-fibers, which make up the bulk of the nerve structures of the tooth.
Interestingly, EPT is rated as a low-value test for primary or permanent teeth during apex formation [9].Many children with normal teeth do not respond even to high stimulation currents [9].We believe that for the reaction of all neuroreceptors in single teeth, a more universal AC stimulator with a broad total effect, mainly on C-fibers, is needed.The aim of our investigation was: to evaluate tooth maturation in children aged 6.5 - 15 years in dynamics in the study of pain caused by electrotesting in three directions:
a) Reduction of the pain threshold of the central incisors,
b) x-ray width of the apex, and
c) The nature of the pain: pain or not pain.
Material and Methods
280 teeth were examined in 280 children from 6 to 15 years (central incisors). Patients were divided by age into 5 groups:1 - 6,5 – 7 years; 2 – 7-8 years; 3 – 9-10 years; 4 11-12 years; 5 – 13-15 years.After receiving informed consent, the oral cavity was examined, the child was instructed, and the central incisors were isolated from saliva. The threshold of pain sensitivity of the dental pulp was determined using the IVN-01 Pulpotest-Pro electrodontodiagnostic device. Measurements were made on the central upper incisor. The pulp tester used created a series of alternating current pulses, devoid of polarization, with a frequency of 50 Hz with a gradually increasing current strength on a scale from 0 to 200 mkA until the first sensation: pain/non-pain[10-15]. The active electrode was located in a PVC tube into which the contact gel was inserted. The diameter of the active electrode was 4 mm. The passive metal electrode was in the hand of the subject. When pain occurred, the subject released the button, thus stopping the current supply, and the value of the EOM was recorded on the display. Indications of the pain threshold after a two-time (one- time) measurement were recorded.In addition to the digital indicator of EOM, the response to electrical stimulation was determined. The main criterion for pain was to divide it into pain / non-pain. All patients according to indications, as a rule, aesthetic, carried out radiography. Intraoral and x-ray images were used to measure the diameter of the tooth apex in mm[16-18].
Results
The results are shown in Table 1. Figure 1 shows a descending curve similar for all three parameters under study: EOM, apex, and pain.The highest EOM index was observed in the first age group (140.86 mkA), exceeding that in the second age group by more than 1.5 times (91.95 mkA). At the same time, the average diameter of the apex decreased from 2.55 to 1.96 mm. For both parameters, the differences between the first and second age groups were statistically significant (t-criteria 5.70 and 6.8, respectively). Between the second and third age groups, the difference in EOM indicators was sharper— 91.95 mkA to 39.75 mkA-with almost the same smooth decrease in the diameter of the apical opening (1.96 to 1.39 mm). For both parameters, the differences between the second and third age groups were statistically significant (criterion t = 7.48 and 8.90). Indicators of EOM in the third and fourth age groups differed a little, although the average value of EOM in the fourth group is slightly lower: 35.83 mkA compared to 39.75 mkA (the differences were not statistically significant, the criterion t = 0.70, and the diameter of the apex was significantly different: 0.82 mm compared to 1.39 mm (criterion t = 11.40). In the fourth and fifth age groups, the EOM as well as the apex diameter differed significantly, but without a sharp decrease in these indicators: from 35.83 to 24.35 mkA and from 0.82 to 0.72 mm (criterion t = 2.21 and 2.36, respectively). A sharp drop in EOM indicators occurred at the age of 6.5 to 11 years with a furthermore gradual decline (Figure 1).In the first age group, 13 subjects had no pain at all, and 6 (38%) subjects had a pre-pain sensation that was close to tactile. In the second group, 4 children had no pain, and 5 had afeeling of pre-pain in the form of cold or heat with spreading in the gum above the tooth (14.5%). In the group of 9-10 years, 4 subjects had a feeling of pre-pain (7.14%), in 11-12 years in 5%, in 13-15 years in 1.92%(Figure 2).
Table 1: Electrodontometry of teeth, apex diameter and sensations (pain, non-pain) in 280 children of different ages.
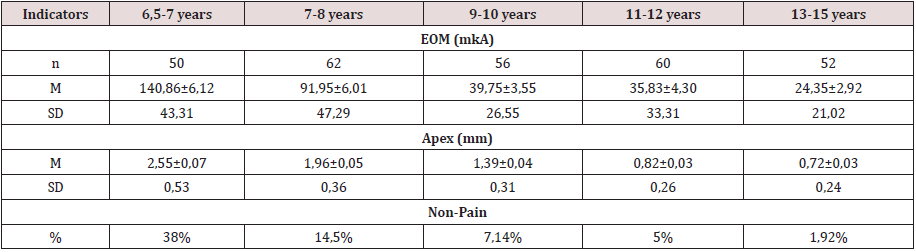
Figure 1: The Value of the pain threshold (A) and the value of the apical opening (B) of the central incisors in children of different ages:1 - 6,5 – 7 years; 2–7-8 years; 3– 9-10 years; 4 11-12years; 5–13-15 years.
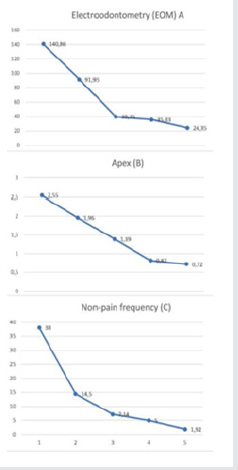
Figure 2: Central incisors of a 6.5-year-old girl. EOM 199 and 195 mkA (apex 3 mm; there is no pain when measuring).
Discussion of Results
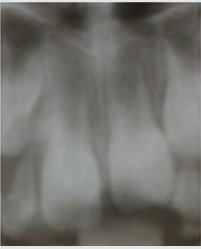
Figure 3: Central incisors of a 12-year-old girl, EOM 13 and 15 mkA (apex 0.8 mm; pain felt during measurement).
Pain is one of the main feelings. With age, the senses improve. The difference in pain assessment is visible on the graph (Figure 1). The youngest ones have the greatest feeling of “non- pain”. Children replaced real pain with feelings of cold, heat, etc. We have combined these feelings into the concept of pre-pain.Pain sensitivity of a permanent tooth is the main pain criterion of a person, in which, unlike animals, there is contact with the researcher. The phenomenon of a sharp decrease in the sensitivity of permanent teeth that are in the process of eruption has been established. A close relationship has been established between the reaction of A-Delta fibers and pulsed diagnostic current. The role of C-fibers – the basis of the tooth’s nervous system-is underestimated. The electric current due to its strength and the large electrode covers the entire pulp, both coronal and apical (Figure 3). In our literature, there is a tendency to prohibit devices that operate from alternating sinusoidal current with a frequency of 50 Hz [1,2]. The regularities between the degree of tooth eruption and the reduction of its pain threshold to a minimum level, while reducing the apical narrowing, and clarifying the pain response in a young subject were determined. The formation of the apex consists in its gradual narrowing, including ingrowth of cement into the apical part of the root canal. It is dominated by C-fibers. Pulse pulp testers do not “feel” the pulp in the erupting teeth.It should be noted that when the final apex was formed, the threshold value did not always correspond to the previously accepted norm of 2-6 mkA. In one 15-year-old patient, the initial pain threshold was 125 mkA.
Conclusions
The phenomenon of reducing the pain sensitivity of permanent teeth during eruption due to the reaction of C-fibers has been established. Pulse pulp testers do not work in teeth during apex formation.
References
- Makeeva IM et al. (2018) Efficiency of electro-dental diagnostics using various types of current. Dentistry 6: 34-37.
- Nikolaev AI, E V Petrova (2014) Electrodonto diagnostics Textbook. Med press- inform 6: 34-37.
- Rubin LR (1976) Elektroodontodiagnostika M p. 136.
- Mukhin ON (1969) Age-related features of electrical excitability of intact permanent teeth. In the book: Clinic and treatment of congenital and acquired defects of the maxillofacial region p. 179-181.
- Lubomirski GB (2010) Clinical and electrometric indicators of dentin caries. Cand Diss Perm.
- Bjorn H (1953) Electric stimulation threshold of teeth with regard to sensitivity in operations involving dentin and pulp. Sven TandlakTidskr 46: 314.
- Cohen (2007) Pathways of the Pulp 9th ed, Copyright © 2006 Mosby, An Imprint of Elsevier p. 1-18.
- Harty F (1976) J Endodontic in clinical practice. Bristol p. 235.
- Camp JH, Barrett EJ, Pulver J (2002) Pediatric endodontic: endodontic treatment for the primary and young permanent dentition. Cohen S RC (Eds.), Burn Pathway of the pulp 8th 2002: 804-807.
- Brännström M (1986) The hydrodynamic theory of dentinal pain: Sensation in preparations, caries, and the dentinal crack syndrome. Journal of Endodontics 12(10): 453-457.
- Chen E, Abbott PV (2009) Dental Pulp Testing: A Review. International Journal of Dentistry (1687-8728): 365785.
- Trombritge H, Kim S (2002) Structure and function Pulp Complex. Sohep S, Burn RC (Eds.), Pahtways the pulp Mosby p. 430-431.
- Närhi M, Jyväsjärvi E, Virtanen A, Huopaniemi T, Ngassapa D, et al. (1992) Role intradental A- and C-type nerve fibresindent a lpainmechanisms. Proc Finn Dent Soc 88(Suppl 1): 507-516.
- Reader A, Nusstein J, Drum M (2017) Successful Local Anesthesia for Restorative Dentistry and Endodontics Quintessence Publishing Co Inc.
- Moroz VT, Ignatov YUD, Kalinin VI (1989) Threshold of electric excitability of the pulp of various groups of teeth and their changes under the influence of analgin, amidopirin and diazepam. Stomatology 68(4): 30-32.
- Zyuzkov DI (2004) The condition of the tooth pulp in inflammatory periodontal diseases. Cand Diss Tver.
- Van TH, JT McSpadden, HE Goodis (2007) Instruments, Materials, and Devices. Cohen (Eds.), Pathways of the Pulp. 9th Ed p. 1-20.
- Mumford JM, Björn H (1962) Problems in electrical pulp-testing and dental algesimetry. Int Dent J 12: 161.
Editorial Manager:
Email:
pediatricdentistry@lupinepublishers.com

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...




