Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-6921
Research Article(ISSN: 2641-6921) 
Post-Breast Cancer Chronic Wounds with Solid Calcifications Treated with Cu/SiO2-TiO2 Nano Biocatalyst Volume 3 - Issue 3
Tessy López*1,2, Verónica Sánchez de la Barquera3, Francisco J Padilla-Godínez1, Paola Ramírez1 and Esteban Gómez-López4
- 1Department of Nanotechnology and Nanomedicine, Autonomous Metropolitan University Xochimilco, Coyoacán, Mexico
- 2Department of Neurology and Neurosurgery, Children’s Hospital of Mexico “Federico Gómez, Cuauhtémoc, Mexico
- 3VitaBene Clinic, Ciudad Satélite, Estado de México 52994, Mexico
- 4AG Nanooptik, Humboldt-Universität zu Berlin, Germany
Received: October 03, 2020; Published:October 22, 2020
*Corresponding author: Tessy López, Department of Nanotechnology and Nanomedicine, Autonomous Metropolitan University Xochimilco, Coyoacán, Mexico City 04960, Mexico
DOI: 10.32474/MAMS.2020.03.000163
Abstract
Objective: To describe patient outcomes with respect to the use of Cu/SiO2-TiO2 nanoparticles embedded in a polymeric gel as treatment for post-breast cancer chronic wounds with solid calcifications.
Methods: The nanoparticles were synthesized by the sol-gel method. The compound was incorporated into a polymeric gel matrix. SEM and TEM microscopy studies were carried out to evaluate grain size, morphology, and texture of the pure nanoparticles. The chronic wounds were treated by applying zinc oxide as emollient, Triticum vulgare (Italdermol®) and the Nano biocatalyst. The wound was covered with Solvaline® N dressings for the absorption of the exudate.
Results: The Cu/SiO2-TiO2 nanobiocatalyst displayed average particle size between 5 and 10 nm with high particle dispersion. Ordered arrangement of the nanoparticles was observed in TEM micrographs suggesting anatase crystalline structure. Microcalcifications extracted from the wound were suspected to be calcium carbonates and calcium oxalates, exhibiting sizes of the order of 20 μm. The administration of the nanoparticles with Triticum vulgare enhanced granulation tissue formation and revascularization by limiting bacterial infection. After 8 months of the first application the wound had completely healed in an atrophic scar, with no sign of inflammation or infection. No adverse effects were observed in the patient.
Conclusions: Post-breast cancer chronic wounds were successfully treated with Cu/SiO2-TiO2 nanoparticles. Good outcomes were observed in terms of tissue regeneration, wound healing, and infection hindering. Calcifications were eliminated. The nanoparticles can be used as a primary apposite to stimulate the autolytic debridement of injures due to their excellent local absorption and bactericide action.
Declaration of Interest: The study’s sponsors were not involved in the study design, the collection, analysis and interpretation of the data, the writing of this manuscript and the decision to submit this article for publication. The views express in this article are those of the authors.
Keywords: Cu/SiO2-TiO2, Chronic wounds, Breast cancer, Microcalcification, Tissue regeneration
Key Points:
a. A nanobiocatalyst based on mixed-oxides impregnated with transition metals was synthesized by the sol-gel method for the
treatment of a post-breast cancer chronic wound.
b. The nanobiocatalyst presented displayed average particle size between 5 and 10 nm with high particle dispersion and
ordered particle arrangement suggesting anatase crystalline structure as observed by SEM and TEM.
c. The post-breast cancer wound exhibited the formation of microcalcifications (20 μm) composed of calcium carbonates and
calcium oxalates.
d. The administration of the nanobiocatalyst enhanced the formation of granulation tissue and revascularization by limiting
bacterial infection.
e. The 30-years-old wound completely healed in an 8-months period, with no adverse effects detected.
f. The microcalcification formation was eliminated after tissue regeneration.
Introduction
Despite recent advances in the detection and treatment of metastatic breast cancer, mortality related to this disease remains as one of the highest due to the emergence of therapy-resistant cancer cells [1-5]. In 2018, an estimated 2 million new global cases were diagnosed representing 23% of all cancers, incidence rates varying from 19.3 per 100,000 women in Eastern Africa to 89.7 per 100,000 women in Western Europe [6]. By 2030, the number of deaths in Latin America is expected to achieve 74,000 per year [7]. The Globocan 2018 estimated that in Mexico the incidence for breast cancer was 39.5 cases per 100,000 habitants, with a total of 27,283 new cases and 6,884 deaths [8,9]. Common treatments for nonmetastatic breast cancer include endocrine therapy and cytotoxic chemotherapy [10]. The main surgical approach for cancer of the breast in all stages, early or late, is a radical and total mastectomy, breast-conserving surgery, and breast reconstruction, all followed by irradiation [11]. Possible side effects of surgery include seroma, hematoma, lymphoedema, cording, restricted range of movement in the shoulder, nerve pain, tissue necrosis, microcalcification formation, and surgical site infection [12-15]. The incidence of wound complications after breast cancer surgery are substantial, ranging from 6 to 30% [16-19]. There are several factors related to increased risk of complications in post-surgical patients, including patient risk factors (e.g., diabetes, obesity, and smoking), [20,21] surgical technique, [22] and type of surgery. [23] In fact, it has been suggested in previous studies that radiation increases the risk of complications, both in patients receiving breast implants, [24-29] and in those treated with autologous reconstructions. Murthy et al. [30-32] proposed that delayed wound healing is associated with an increased rate of systemic recurrence after primary breast cancer excisional surgery, [33] therefore one main point of attention in postoperative care must be the control of wound-closure-hindering infections.
In past studies, we have evaluated the healing properties of copper nanoparticles stabilized in a titania-silica mixed oxide matrix (Cu/SiO2-TiO2) as a treatment for chronic diabetic foot ulcers [34,35]. The nanoparticles prepared by the sol-gel process allowed to avoid a programmed supracondylar amputation in the patient, with favorable clinic evolution of the chronic ulcer by enhancing tissue regeneration and wound healing.
Aim of the Study
The aim of this study was to determine the wound healing properties of the Cu/SiO2-TiO2 nanoparticles for the management of breast-cancer-related chronic wounds in combination with Triticum vulgare (used as a treatment of oral mucositis by radiotherapy and chemotherapy in cancer patients) [36] and Solvaline® N (wound dressing).
Materials and Methods
Ethanol (99.8%), copper acetylacetone (97%), sulfuric acid (98%), phosphoric acid (98%), tetraethyl orthosilicate (98%) and titanium butoxide (98%) were purchased from Sigma Aldrich Company (Saint-Louis, MO, USA). Solvaline® dressings were purchased from Lohmann & Rauscher (Milwaukee, WI, USA), and aqueous extract of Triticum vulgare (Italdermol®) was obtained from Italmex (Mexico City, CDMX, Mexico).
Preparation of Cu/SiO2-TiO2 Nanoparticles
The nanoparticles were synthesized following the procedure of secrecy No. 101/100/014/13. A flask with deionized water and ethanol was kept at room temperature and constant stirring, the adequate amount of copper acetylacetonate was added to this mixture, then the pH was adjusted to pH 1 with sulfuric and phosphoric acids. During a period of 4h, tetraethyl orthosilicate and titanium butoxide were added dropwise to the mixture under stirring. Once the alkoxides were completely added, the temperature was increased while stirring until gelation. The nanoparticles were obtained when the gel dried. The compound was incorporated into a polymeric gel matrix.
Scanning and transmission electron microscopy studies
The grain size, morphology, and texture of the pure nanoparticles with no polymeric gel matrix were characterized by scanning electron microscopy (SEM) in a JEOL JSM-6010LV microscope equipped with an energy dispersive spectroscopic (EDS) microanalysis system (OXFORD). The nanoparticles’ size was determined by transmission electron microscopy (TEM) in a JEOL JEM-2100F, operated at 120 kV voltage. The images were obtained using a CCD Mega Vision (III) camera.
Clinical Study
Patient Clinical History
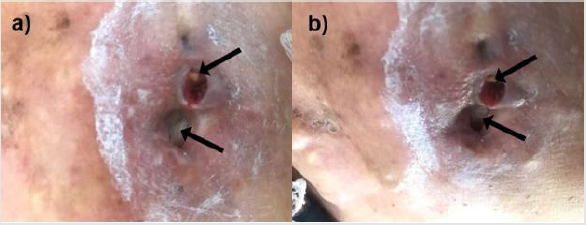
A 78-years-old female patient from Mexico City presents a family history of diabetes mellitus and hypertension, as well as cases of chronic venous insufficiency and deep vein thrombosis, from both mother and father lineages. She presents type-2 diabetes mellitus for 15 years treated with biguanide and insulin 30-0-20, with medication non-adherence problems. The patient refers previous history of mixed dyslipidemia and hypertension. Furthermore, the patient was diagnosed with Invasive Ductal Carcinoma (IDC) in the left breast thirty years ago, which conducted to total mastectomy and irradiation. The breast cancer exhibited total remission for 30 years. The patient reported to medical consultation for presenting small wounds in the chest and secondary calcifications that were growing inside and outside the injury since the beginning of irradiation therapy. During first physical exploration blood pressure was 140/90 mmHg, heart frequency had a rate of 76 beats per minute, respiratory rate of 18 breath per minute, temperature was 36.9°C. The patient denies fever, general discomfort, asthenia, and adynamia. The size of the wound located in the left thorax is 2.0 x 1.0 x 1.5 cm with macerated borders (Figure 1a&1b), the perilesional skin showed edema, erythema, and hyperpigmentation, with yellow exudate and foul odorous suppuration, visible calcifications were identified inside the wound. She exhibits a scar due to the previous total mastectomy of the left breast. The patient refers moderate pain at palpation and during wound cleaning.
Figure 1: Post-breast cancer chronic wound with solid calcifications. The size of the wound is 2.0 x 1.0 x 1.5 cm with macerated borders. Calcifications are identified with black arrows.
Clinical Diagnosis
Chronic radiation-induced ulcer in the chest wall after surgery in breast cancer. Risk factors for systemic recurrence: female, total mastectomy, chronic untreated ulcers, type-2 diabetes mellitus, hypertension.
Treatment
Advanced wound care was initiated by applying zinc oxide (ZnO) on the macerated skin due to its epithelialization and infection-control properties [37]. The Cu/SiO2-TiO2 nanoparticles embedded in a polymeric matrix were applied in combination with Triticum vulgare inside the wounds to generate granulation tissue and limit bacterial infection. The wound was covered with Solvaline® N dressings for the absorption of the exudate. Additional management included prophylaxis antibiotics on one occasion and analgesics each 12 hours when pain was presented.
Results
Electronic Microscopy Studies Of The Nanoparticles
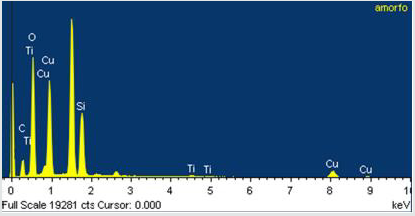
SEM images showed Cu/SiO2-TiO2 nanoparticles formed conglomerates with visible crystalline structure and average particle size between 0.1 and 1 μm, as shown in (Figure 2a). The conglomerates exhibit a flake morphology with a flustered arrangement of the nanoparticles. A deeper analysis increasing magnification to 37,000x (Figure 2b), 70,000x (Figure 2c), and 100,000x (Figure 2d) allowed to observe individual nanoparticles with sizes ~ 10 nm grouped in 100-nm conglomerates. A further study of TEM (Figure 3) allowed to corroborate individual particle size to be of the order of 5 nm. Particle arrangement observed in TEM images suggest the presence of crystalline structure, possibly microcrystalline anatase with grain sizes of 10-30 nm as observed in previous XRD studies regarding the SiO2-TiO2 mixed oxide matrix [38-40] (the diffractograms are not shown in this paper for the sake of brevity). EDS measurements in the conglomerates showed homogeneous distribution of the elements present in the compound with atomic percentages as expected (Figure 4). The atomic composition is shown in Table 1.
Figure 2: SEM micrographs of the Cu/SiO2-TiO2 nanoparticles at 15,000 x (a), 37,000 x (b), 70,000 x (c), and 100,000 x (d). Nanoparticle conglomerate formation with sizes 0.1–1.0 μm is observed. Individual nanoparticles exhibit average particle size of 5-10 nm.

Figure 3: TEM micrographs of the Cu/SiO2-TiO2 nanoparticles with dimensions 50 nm (a, b) and 10 nm (c, d). Individual particle size was identified to be < 10 nm. The nanoparticles exhibit crystalline structures with grain sizes of 10-30 nm. Particle arrangement suggests an anatase crystalline structure.
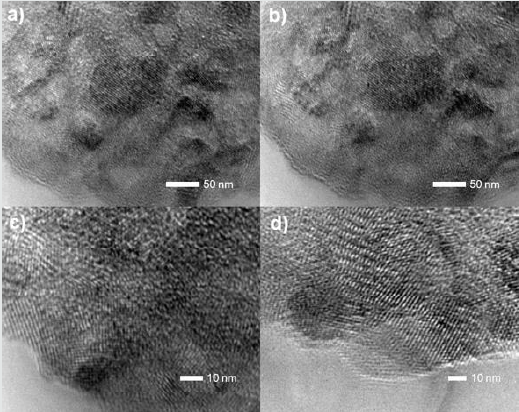
Figure 4: EDS measurement in the conglomerates with the presence of Ti, Si, O, and Cu, elements that compose the Cu/SiO2-TiO2 nanoparticles. Carbon peaks correspond to the carbon tape in which the sample was placed.
Microcalcification Analysis
Microcalcification accompanies several benign and malignant alterations in breast parenchyma [41,42]. Most commonly the calcifications (5 μm to 100 μm) are composed of hydroxyapatite (Ca10(PO4)6(OH)2), which forms concentric concretions and amorphous masses, [43] exhibiting grey-white, opaque, and ovoid form or fusiform shapes with irregular surfaces [44]. Less often, calcium oxalate dihydrate (Weddellite) is deposited, forming single crystals up to 1 mm long, [44,45] which are amber in color, partially transparent and form pyramidal structures with relatively planar surfaces [46]. Before treatment with Cu/SiO2-TiO2 nanoparticles and Triticum vulgare, the wound was cleaned, and intern calcifications were removed. After a few applications, internal calcifications emerged from the wound, which captured our attention. For a better understanding of the properties of the calcifications obtained, the crystals were analyzed by SEM and EDS techniques to identify morphology, texture, and composition. In (Figure 5) a sample of epithelial tissue is observed with the presence of individual calcifications with sizes of the order of 20 μm. The masses exhibit amorphous structures with no visible presence of crystal twinning. According to bibliography revised, [15,43-45] our hypothesis is that the calcifications are composed of calcium carbonate (CaCO3), calcium oxalate (Ca(CO2)), or hydroxyapatite (Ca10(PO4)6(OH)2); the aforementioned is intended to be corroborated in a further EDS study. Calcium oxalate has been shown to be associated with benign lesions of the breast or at most non-invasive lobular carcinoma in situ [47]. Calcium carbonate microcalcifications identified with breast tissues grow in calcite and aragonite crystal structures [48].
Figure 5: SEM micrographs of the calcifications observed in the wound-tissue sample. Individual calcifications with sizes of the order of 20 μm are observed surrounded by erythrocytes deposited in the surface of the epithelial tissue.

Clinical Study
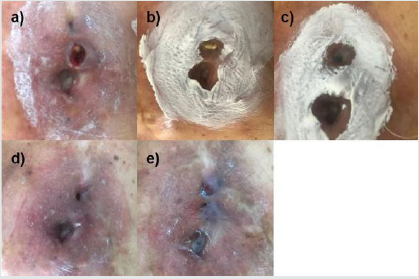
Administration of Cu/SiO2-TiO2 nanoparticles with Triticum vulgare enhanced granulation tissue formation by limiting bacterial infection as early as two months after the first application. The wound was dry and clean, with sharp edges and edema reduction. After four months of treatment, the size and depth of the chronic wound had reduced significantly, presenting an erythematous clean bed and a decrease in calcification formation. At the 6-months mark, granulation tissue was observed with optimal reepithelization and no fibrin due to revascularization and fibroblast production. After 8 months of the first application, the wound has completely healed in an atrophic scar. No inflammation was observed. The healing process is observed in (Figure 6).
Figure 6: Evolution of the chronic wound with Cu/SiO2-TiO2 + Triticum vulgare treatment at time 0 (a), after 2 months (b), after 4 months (c), after 6 months (d), and after 8 months (e). The white film observed corresponds to the zinc oxide applied as emollient.
Discussion
The clinical case presented several risk factors that seriously complicated the wound healing process and represented a possible systemic recurrence: chronic untreated ulcers, hypertension, and type-2 diabetes mellitus. In healthy patients, the healing process involves coordinated interactions between diverse immunological and biological systems, which are divided into four time-dependent steps: (i) coagulation and hemostasis, (ii) inflammation, (iii) proliferation, and (iv) wound remodeling [49-55]. The four steps are essentially dependent on a proper blood irrigation to the wound for the transport of the cells (endothelial, thrombocytes, neutrophils, etc.), nutrients, and coagulation and inflammatory factors that will intervene in the process [56-59]. Diseases that lead to compromised microcirculation, such as diabetes, inhibit the delivery of oxygen and nutrients to tissue injuries, impairing wound healing [60] and causing the development of chronic wounds on account of impaired growth factor production, angiogenic response, collagen accumulation, fibrosis, and abnormal blood pressure [61]. Furthermore, due to chemotaxis and impaired leukocyte function and inadequate migration of neutrophils and macrophages to the wound, [62-64] diabetic patients develop a preponderance for infections such conditions demand for wound healing treatments that enhance reepithelization while reducing infection progression [65,66].
Titania-silica mixed oxide nanoparticles with high specific surface areas (up to 645 m2/g) have been studied regarding their catalytic properties for a broad variety of applications, [38,40,67- 69] including absorption of organic pollutants, [70] drug delivery, [71,72] and catalysis [73-75]. The incorporation of transition metals (such as platinum and copper) in the mixed matrix has been reported to enhance the antibacterial properties of the compound. [34,35] For the treatment of the chronic wounds the patient exhibited, we synthesized Cu/SiO2-TiO2 nanoparticles with catalytic and antibacterial properties. The nanoparticles were obtained by the sol-gel process through a hydrolysis catalyzer to achieve acid conditions in the gelation process, and thus, to obtain acid nanomaterials. The development of acidic properties in binary mixed metal oxides in terms of Lewis and Brønsted acidic sites has a direct impact on the catalytic properties of the nanoparticles. [38,76,77] The nanoparticles’ size observed in SEM and TEM studies (5-10 nm) makes them capable to traverse the plasmatic membrane of the bacteria through passive diffusion [78]. Once inside the bacteria, due to synthesis method used and their functionalized surface, the nanoparticles are able to catalyze the breakage of C-C and C-N bonds present in DNA and RNA nitrogenous bases hence destabilizing the structure of the molecule and inhibiting the bacterial reproduction [79]. This biocatalytic process is believed to generate molecular oxygen which enhances the proliferation of fibroblast activity, [80] collagen synthesis, [81] and inflammation, [82] hence improving the generation of granulation tissue and reepithelization, and significative decreasing the time of recovery.
The patient’s wounds treated with the Cu/SiO2-TiO2 nanoparticles exhibited total recuperation after 8 months since the first application. Through the healing process, internal solid calcifications emerged from the wound. They were removed and analyzed by SEM technique. The presence of calcium compounds in the breast is related to benign lesions, as the chronic wounds the patient exhibited. After the application of the nanoparticles, the calcification formation was reduced and eliminated as a sign of lesion recovery [41,83]. The successful outcome achieved with the treatment meant a significant improvement in the patient’s quality of life, eliminating the possibility of a systemic recurrence of breast cancer [18].
Conclusion
Post-breast cancer chronic wounds were successfully treated with Cu/SiO2-TiO2 nanoparticles in a patient with the menace of systemic recurrence due to risk factors such as hypertension and type-2 diabetes mellitus. Good outcomes were observed in terms of tissue regeneration and wound healing. The therapy with the nanoparticles embedded in a polymeric gel hindered infection formation allowing wound reepithelization and healing with a time of recuperation significantly reduced (8 months in a 30-years-old chronic wound). Furthermore, calcifications present in the wound were removed and the recurrent formation was eliminated after the treatment. No adverse effects were observed in the patient. Scientific evidence supporting the efficacy and safety of the Cu/ SiO2-TiO2 therapy is hereby shown. The nanoparticles can be used as a primary apposite to stimulate the autolytic debridement of injures, due to their physical-pharmaceutical properties: excellent local absorption because of its nanoparticulate composition and bactericide action that helps control local infections. Yet, future studies must be carried out to further confirm the efficacy of the nanoparticles.
Reflective questions
a. Microcalcification formation elimination is related directly with the catalytic effect of the nanoparticles or with the wound healing process?
b. Do we need to assess the effectiveness the nanobiocatalyst
in the real world and not simply rely on clinical trial data?
c. What can be done to improve wound healing rates?
d. How can the efficiency of health-care delivery be
improved?
Author Contribution
All authors contributed to the study conception and design. The design and synthesis of the nanoparticles were executed by TL, PR and FJPG. The characterization techniques were made by EGL. The patient’s treatment was carried out by VSB. The first draft of the manuscript was written by FJPG. All authors commented on previous versions of the manuscript and critically revised it. FJPG was responsible for compiling the final draft. All authors read and approved the final draft.
Funding
This study was funded by the Autonomous Metropolitan University – Xochimilco and NANOTUTT S.A. de C.V.
Compliance With Ethical Standards
Conflict of Interest: The authors declare that they have no conflict of interest.
Ethics Approval: This study was performed in line with the principles of the Declaration of Helsinki. Necessary ethical clearance was obtained from the ethical committee office code number JUDI- 01/07 CDMX under the protocol: [“Security and efficacy of the local application of Pt/SiO2-TiO2 nanomaterials in patients with diabetic foot ulcers” Education and Investigation Direction], and the study was conducted in accordance with the ethical Good Clinical Practice.
Declarations
Consent to participate. Informed consent was compulsory for contribution in the study. The staff notified the contributor with the objectives, dates, drugs, diet, possible risks, and general activities through the clinical study.
Consent to Publish: No personal data of the patients was mentioned in the study.
ORCID ID
1. Tessy López: https://orcid.org/0000-0002-6048-0419
2. Francisco J. Padilla-Godínez: https://orcid.org/0000-
0002-9253-2463
References
- Al-Hajj M, Wicha MS, Benito-Hernández A, et al. (2003) Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A 100(7): 3983-3988.
- Stockler M, Wilcken NRC, Ghersi D, Simes RJ (2000) Systematic reviews of chemotherapy and endocrine therapy in metastatic breast cancer. Cancer Treat Rev 26(3): 151-168.
- Schultz LB, Weber BL (1999) Recent advances in breast cancer biology. Curr Opin Oncol 11(6): 429-434.
- Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, CA Cancer J Clin 66(1): 7-30.
- Siegel RL, Miller KD, Fedewa SA, Stacey A, Lynn F, et al. (2017) Colorectal cancer statistics, CA Cancer J Clin 67(3): 177-193.
- Zaidi Z, Dib HA (2019) The worldwide female breast cancer incidence and survival, 2018 [abstract]. Cancer Res 79 Abstract nr 4191.
- World Health Organization (2008) The Global Burden of Disease: 2004 Update. WHO Press Geneva, Switzerland.
- Global Cancer Observatory. Globocan 2019.
- National Institute of Statistics and Geography (2015) Informe 2014: Actividades y Resultados [Inform 2014: Activities and Results].
- Waks AG, Winer EP (2019) Breast Cancer Treatment: A Review. JAMA-J Am Med Assoc 2019; 321(3): 288-300.
- Fisher B, Jeong J-H, Anderson S, John Bryant, Edwin R Fisher, et al. (2002) Twenty-five-year follow-up of a randomized trial comparing radical mastectomy, total mastectomy, and total mastectomy followed by irradiation. N Engl J Med 347(8): 567-575.
- Gieni M, Avram R, Dickson L, F Farrokhyar, P Lovrics, et al. (2012) Local Breast Cancer Recurrence After Mastectomy and Immediate Breast Reconstruction for Invasive Cancer : A Meta-Analysis. Breast 21(3): 230-236.
- Henry NL, Shah PD, Haider I (2020) Cancer of the breast. In: Niederhuber JE, Armitage JO, Doroshow JH, et al. (eds). Abeloff’s Clinical Oncology (6th edn) Elsevier.
- Gallagher M, Jones DJ, Bell-Syer SV (2019) Prophylactic antibiotics to prevent surgical site infection after breast cancer surgery. Cochrane Database of Syst Rev 9(9): CD005360.
- Morgan MP, Cooke MM, McCarthy GM (2005) Microcalcifications associated with breast cancer: an epiphenomenon or biologically significant feature of selected tumors? J Mammary Gland Biol Neoplasia 10(2): 181-187.
- Sørensen LT, Hørby J, Friis E, B Pilsgaard, T Jørgensen (2002) Smoking as a risk factor for wound healing and infection in breast cancer surgery. Eur J Surg Oncol 28(8): 815-820.
- Tran CL, Langer S, Broderick-Villa G, DiFronzo LA (2003) Does reoperation predispose to postoperative wound infection in women undergoing operation for breast cancer. Am J Surg 69(10): 852-856.
- Hall JC, Hall JL (2004) The measurement of wound infection after breast surgery. Breast J 10(5): 412-415.
- Alderman AK, Wilkins EG, Kim HM, Lowery JC (2002) Complications in post-mastectomy breast reconstruction: two-year results of the Michigan breast reconstruction outcome study. Plast Reconstr Surg 109(7): 2265-2274.
- Haley RW, Culver DH, Meade Morgan W, Hooton TM, et al. (1985) Identifying patients at high risk of surgical wound infection. Am J Epidemiol 121(2): 206-215.
- Mangram AJ, Horan TC, Pearson ML (1999) Guideline for the prevention of surgical site infection. Infect Control Hosp Epidemiol 27(2): 97-132.
- Ritter MA, Marmion P (1988) The exogenous sources and controls of microorganisms in the operating room. Orthop Nurs 7(4): 23-28.
- Gruendemann BJ, Mangum SS (2001) Infection prevention in the surgical setting (1st Edn) WB Saunders. pp.387.
- Kronowitz SJ (2012) Current status of implant-based breast reconstruction in patients receiving postmastectomy radiation therapy. Plast Reconstr Surg 130(4): 513e-523e.
- Whitfield GA, Horan G, Irwin MS, Malata CM, Wishart GC, et al. (2009) Incidence of Severe Capsular Contracture Following Implant-Based Immediate Breast Reconstruction with or without Postoperative Chest Wall Radiotherapy Using 40 Gray in 15 Fractions. Radiother Oncol 90(1): 141-147.
- Jhaveri JD, Rush SC, Kostroff K (2008) Clinical outcomes of postmastectomy radiation therapy after immediate breast reconstruction. Int J Radiat Oncol Biol Phys 72(3): 859-865.
- Krueger EA, Wilkins EG, Strawderman M, P Cederna, S Goldfarb, et al. (2001) Complications and patient satisfaction following expander/implant breast reconstruction with and without radiotherapy. Int J Radiat Oncol Biol Phys 49(3): 713-721.
- Contant CME, van Geel AN, van der Holt B, C Griep, R Tjong Joe Wai, et al. (2000) Morbidity of immediate breast reconstruction (IBR) after mastectomy by a subpectorally placed silicone prosthesis: the adverse effect of radiotherapy. Eur J Surg Oncol 26(4): 344-350.
- Tallet AV, Salem N, Moutardier V, Pascal Ananian, Anne-Chantal Braud, et al. (2003) Radiotherapy and immediate two-stage breast reconstruction with a tissue expander and implant : complications and esthetic results. Int J Radiat Oncol Biol Phys 57(1): 136-142.
- Kronowitz SJ (2012) Current status of autologous tissue-based breast reconstruction in patients receiving postmastectomy radiation therapy. Plast Reconstr Surg 130(2): 282-292.
- Williams KJ, Carlson GW, Bostwick J 3rd, J T Bried, G Mackay (1997) The effects of radiation treatment after TRAM flap breast reconstruction. Plast Reconstr Surg 100(5): 1153-1160.
- Rogers NE, Allen RJ (2002) Radiation effects on breast reconstruction with the deep inferior epigastric perforator flap. Plast Reconstr Surg 109(6): 1919-1924.
- Murthy BL, Thomson CS, Dodwell D, Shenoy H, Mikeljevic JS, et al. (2007) Postoperative wound complications and systemic recurrence in breast cancer. Br J Cancer 97(9): 1211-1217.
- López-Goerne T, Arévalo A, Ramírez P, Larraza P (2017) Copper nanoparticles as treatment of diabetic foot ulcers: A case report. Glo Adv Res J Med Med Sci 6(10): 267-270.
- López-Goerne T, Rodríguez-Pérez CE, Álvarez-Cordero R (2015) Uso de la nanopartícula de SiO2-TiO2 en el tratamiento de úlceras en pie diabético: comunicación preliminar. Rev Fac Med UNAM 58(3): 5-12.
- Barbosa-Zamora A, Ponce-Olivera RM, Arellano-Mendoza MI (2017) Triticum vulgare como tratamiento de mucositis oral por radioterapia y quimioterapia en pacientes oncoló Dermatol Rev Mex 61(5): 379-385.
- Kogan S, Sood A, Garnick MS (2017) Zinc and wound healing: a review of zinc physiology and clinical applications. Wounds 29(4): 102-106.
- López T, Bosch P, Tzompantzi F, Navarrete J, López-Salinas E, et al. (2000) Effect of sulfation methods on TiO2-SiO2 sol-gel catalyst acidity. Appl Catal A-Gen 197(1): 107-117.
- López T, Ortiz E, Meza D, Basaldella E, Bokhim X, et al. (2011) Controlled release of phenytoin for epilepsy treatment. Mater Chem Phys 126(3): 922-929.
- López-Goerne T, Ramírez P, Álvarez D, Francisco Rodríguez-Reinoso, Ana M Silvestre-Albero, et al. (2018) Physicochemical properties and in vivo evaluation of Pt/SiO2-SiO2 Nanomedicine (Lond) 13(17): 2170-2185.
- Going JJ, Anderson TJ, Crocker PR, Levison DD (2007) Weddellite calcification in the breast: eighteen cases with implications for breast cancer screening. Histopathology 16(2): 119-124.
- Holme TC, Reis MM, Thompson A, Robertson A, Parham D, et al. (1993) Is mammographic microcalcification of biological significance? Eur J Surg Oncol 19(3): 250-253.
- Chin DD, Wang J, Mel de Fontenay M, Anastasia Plotkin 2, Gregory A Magee, et al. (2019) Hydroxyapatite-binding micelles for the detection of vascular calcification in atherosclerosis. J Mater Chem B 7(41): 6449-6457.
- Frappart L, Boudeulle M, Boumendil J, Lin HC, Martinon I, et al. (1984) Structure and composition of microcalcifications in benign and malignant lesions of the breast : study by light microscopy, transmission and scanning electron microscopy, microprobe analysis, and X-ray diffraction. Hum Pathol 15(9): 880-889.
- Büsing CM, Keppler U, Menges V (1981) Differences in microcalcification in breast tumors. Virchows Arch [Pathol Anat] 393(3): 307-331.
- Cox RF, Hernandez-Santana A, Ramdass S, McMahon G, Harmey JH, et al. (2012) Microcalcifications in breast cancer: novel insights into the molecular mechanism and functional consequence of mammary mineralization. Br J Cancer 106(3): 525-537.
- Haka AS, Shafer-Peltier KE, Fitzmaurice M, Joseph Crowe, Ramachandra R, et al. (2002) Identifying microcalcifications in benign and malignant breast lesions by probing differences in their chemical composition using Raman spectroscopy. Cancer Res 62(18): 5375-5480.
- Rodgers K, Lewis R (2002) The nature of breast tissue calcifications. In: Evans A, Pinder S, Wilson R, Ellis I. (eds) Breast Calcifications: A Diagnostic Manual (1st Edn). Greenwich Medical Media.
- Vanwijck R (2001) Surgical biology of wound healing. Bull Mem Acad R Med Belg 115(3-4): 175-184. French.
- Degreef HJ (1998) How to heal a wound fast. Dermatol Clin 16(2): 365-375.
- Attinger CE, Janis JE, Steinberg J, Jaime Schwartz, Ali Al-Attar, et al. (2006) Clinical approach to wounds: debridement and wound be preparation including the use of dressings and wound-healing adjuvants. Plast Reconstr Surg 117(7 Suppl): 72S-109S.
- Broughton G 2nd, Janis JE, Attinger CE (2006) Wound healing: an overview. Plast Reconstr Surg 117(7 Suppl): 1e-S-32e-S.
- Hunt TK, Hopf H, Hussain Z (2000) Physiology of wound healing. Adv Skin Wound Care 13(2 Suppl): 6-11.
- Glat PM, Longaker MT (1997) Wound healing. In: Aston SJ, Beasley RW, Thorne CH (eds) Grabb and Smith’s Plastic Surgery (5th Edn). Lippincott-Raven.
- Velnar T, Bailey T, Smrkolj V (2009) The wound healing process: an overview of the cellular and molecular mechanisms. J Int Med Res 37(5): 1528-1542.
- Edlich RF, Rodeheaver GT, Morgan RF, Berman DE, Thacker JG (1988) Principles of emergency wound management. Ann Emerg Med 17(12): 1284-1302.
- Strecker-McGraw MK, Jones TR, Baer DG (2007) Soft tissue wound and principles of healing. Emerg Med Clin North Am 25(1): 1-22.
- Broughton G 2nd, Janis JE, Attinger CE (2006) The basic science of wound healing. Plast Reconstr Surg 117(7 Suppl): 12S-34S.
- Jespersen J (1988) Pathophysiology and clinical aspects of fibrinolysis and inhibition of coagulation. Experimental and clinical studies with special reference to women on oral contraceptives and selected groups of thrombosis prone patients. Dan Med Bul 35(1): 1-33.
- Saaristo A, Tammela T, Fārkkilā A, Marika Kärkkäinen, Erkki Suominen, et al. (2006) Vascular endothelial growth factor-C accelerates diabetic wound healing. Am J Pathol 169(3): 1080-1087.
- Gianino E, Miller C, Gilmore J (2018) Smart wound dressings for diabetic chronic wounds. Bioengineering (Basel) 5(3): 51.
- Delamaire M, Maugendre D, Moreno M, H Allannic, B Genetet (1997) Impaired leucocyte functions in diabetic patients. Diabet Med 14(1): 29-34.
- Wysocki J, Wierusz-Wysocka B, Wykretowicz A, Wysocki H (1992) The influence of thymus extracts on the chemotaxis of polymorphonuclear neutrophils (PMN) from patients with insulin-dependent diabetes mellitus (IDD). Thymus 20(1): 63-67.
- Hirsch T, Spielmann M, Zuhaili B, Till Koehler, Magdalena Fossum, et al. (2008) Enhanced susceptibility to infections in a diabetic wound healing model. BMC Surg 8(1).
- Ferringer T, Miller OF 3rd (2002) Cutaneous manifestations of diabetes mellitus. Dermatol Clin 20(3): 483-492.
- Greenhalgh DG (2003) Wound healing and diabetes mellitus. Clin Plastic Surg 30(1): 37-45.
- López T, Ortiz E, Guevara P, et al. (2014) Cu/TiO2-SiO2 nanostructured materials for brain cancer treatment. TechConnect Briefs 2(1):371-374.
- López T, Jardon G, Gómez E, Gracia A, Hamdan A, et al. (2015) Ag/TiO2-SiO2 sol gel nanoparticles to use in hospital-acquired infections (HAI). Mater Sci Eng C 4(6): 1-6.
- López T, Figueras F, Manjarrez J, Bustos J, Alvarez M, et al. (2010) Catalytic nanomedicine: a new field in antitumor treatment using supported platinum nanoparticles: in vitro DNA degradation and in vivo tests with C6 animal model on Wistar rats. Eur J Med Chem 45(5): 1982-1990.
- Li Y, Kim S-J (2005) Synthesis and characterization of nano titania particles embedded in mesoporous silica with both high photocatalytic activity and adsorption capability. J Phys Chem B 109(25): 12309-12315.
- Farooq A, Shukur A, Astley C, Lubomira Tosheva, Peter Kelly, et al. (2018) Titania coating of mesoporous silica nanoparticles for improved biocompatibility and drug release within blood vessels. Acta Biomater 76: 208-216.
- Georgescu D, Brezoiu AM, Mitran RA, Daniela B, Cristian M, et al. (2017) Mesostructured silica-titania composites for improved oxytetracycline delivery systems. C R Chim 20(11-12): 1017-1025.
- De Clercq R, Dusselier M, Poleunis C, Claude Poleunis, Damien P, et al. (2018) Titania-silica catalysts for lactide production from renewable alkyl lactates : structure-activity relations. ACS Catal 8(9): 8130-8139.
- Gao X, Wachs IE (1999) Titania-silica as catalysts: molecular structural characteristics and physicochemical properties. Catal Today 51(2): 233-254.
- Hu S, Willey RJ, Notari B (2003) An investigation on the catalytic properties of titania-silica materials. J Catal 220(1): 240-248.
- Tanabe K, Misono M, Ono Y, Hattori H (1990) New Solid Acids and Bases: Studies in Surface Science and Catalysis, Vol 51 Elsevier Science Publishers.
- Navarrete J, López T, Gómez R, Figueras F (1996) Surface acidity of sulfated TiO2-SiO2 sol-gels. Langmuir 12(18): 4385-4390.
- Behzadi S, Serpooshan V, Tao W, Alkawareek MY, Dreaden EC, et al. (2018) Cellular uptake of nanoparticles: journey inside the cell. Chem Soc Rev 46(14): 4218-4244.
- López-Goerne TM (2013) Nanomedicina Catalítica: Ciencia y Cáncer. Arkhé Ediciones.
- Hunt TK, Niinikoski J, Zederfeldt B (1972) Role of oxygen in repair processes. Acta Chir Scand 138(2): 109-110.
- Siddiqui A, Galiano RD, Connors D, Gruskin E, Wu L, et al. (1996) Differential effects of oxygen on human dermal fibroblasts : acute versus chronic hypoxia. Wound Repair Regen 4(2): 211-218.
- Rabkin JM, Hunt TK (1998) Infection and oxygen. In: Davis JC, Hunt TK. (Edn) Problem Wounds: The Role of Oxygen, Elsevier.
- Frouge C, Meunier M, Guinebretière JM, Gilles R, Vanel D, et al. (1993) Polyhedral microcalcifications at mammography : histological correlation with calcium oxalate. Radiology 186(3): 681-684.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...