Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2638-6070
Review Artcle(ISSN: 2638-6070) 
The Combined Effects of IgA-Mediated Immunity and Rice Consumption in Suppressing COVID-19 Infections
Volume 3 - Issue 2Shaw Watanabe1* and Kazumoto Inuma2
- 1Asia Pacific Clinical Nutrition Society, Japan
- 2Ricetech Corporation, Japan
Received: July 25, 2020; Published: August 05, 2020
*Corresponding author: Shaw Watanabe, Life science Promoting Association, Asia Pacific Clinical Nutrition Society, Japan
DOI: 10.32474/SJFN.2020.03.000158
Abstract
The number of COVID-19 cases in 19 major countries on 5 continents revealed a strong negative correlation with rice consumption per capita. As previously reported, the overall frequency of IgA-deficiency is inversely correlated with COVID-19 infection. We therefore studied the correlation between rice consumption and IgA-deficiency in a subset of 8 countries for which enough data were available. The results revealed a negative correlation with a coefficient of determination (R2) of 0.55. To further confirm the relationship between rice consumption and COVID-19 cases, we selected 9 Asian countries. The result was the same as described, and countries with the rice consumption more than 80-100kg/capita/year showed the lower incidence. In Japan, the specific COVID-19 infection rate by age groups was investigated, in relation with average rice consumption by age categories. The crude correlation between rice consumption and COVID-19 incidence was -0.59, but the age-adjusted correlation became -0.81. These suggested that SARS-CoV-2 infection is strongly affected by staple foods, and that rice dieters have a stronger innate immunity toward COVID-19. Improvement of agricultural policies and eating habits, especially the promotion of brown rice food and rice bran, are expected to be useful strategies against to counter the coronavirus pandemic.
Keywords: COVID-19; Innate immunity; Rice consumption; IgA deficiency; Statistics
Introduction
We have previously reported that brown rice eaters are less likely to be obese and in healthy state [1,2]. They showed a daily bowel movement and good stool figures. Their intestinal microbiota showed a predominance of Firmicutes phyla, including Blautia, Faecalibacterium, Ruminococcus generi. These are known to contribute to the balance of innate immunity. We considered this microbiota pattern to be a possible factor to explain large differences in COVID-19 infection rates between Japan and Western countries. Statistical analysis based on data from 19 major countries on 5 continents revealed a strong negative correlation (coefficient of determination = 0.59) between rice consumption and the incidence of COVID-19 cases [3]. By contrast, wheat consumption showed a positive correlation. The cumulative number of COVID-19 patients per 100,000 population varies between countries [4-6] Spain and Italy have more than 30, many European countries have between 10 and 20. And East Asian countries have 0.1-0.3. Various host factors can influence mortality, but innate immunity prevails at the initial stage of infection. IgA is a major factor of mucosal immunity, but genetic deficiency of IgA is common in Caucasians [7-10]. We found that COVID-19 infection was paralleled with IgA deficiency [11]. Diarrhea and other gastrointestinal symptoms occur in about 10% of people with SARS-CoV-2 infection. The infected pharyngeal epithelium may fall into the digestive tract along with saliva, and infection may be established when the intestinal immunity is weak. IgA has a secretory form, and IgA deficiency is common in Europe and the United-States, compared people of East-Asian origin. This difference may also explain variations in COVID-19 mortality. So, we felt it was necessary to further investigate the relationship between the frequency of IgA deficiency and rice consumption by comparing countries. Another question was how much is the effective dose of rice consumption? To analyze more deeply, we selected rice-eating countries in Asia, and analyzed the relationship between rice consumption and COVID-19 incidence. Rice consumption habits have decreased by half during the last 60 years in Japan. So, we watched the relationship between rice consumption and COVID-19 to know whether or not the decreasing rice consumption effected on the incidence by age subpopulation.
Statistical Analysis
The coefficient of determination was calculated by the least
square method and regression analysis of two variables, using the
IBM-SPSS version 24 software [12]. Data on per capita consumption
of rice by country (kg/year, 1998) and the incidence of new corona
cases are based on the literature [13,14]. Original data on rice
consumption come from the USDA (United States Department of
Agriculture) in which rice consumption was defined as the amount
of polished rice ingested per individual per year. In a previous
report, we used data on rice consumption in 1998. This time for
Asian countries we adjusted the 60-year average to secular changes
observed from 1960 to 2019 and re-calculated the correlation.
Regarding IgA-deficiency, data are available for a limited number of
countries: Saudi Arabia, Nigeria, Italy, Spain, the United Kingdom,
the United States, China and Japan [6-8].
Trends in rice consumption in 9 Asian countries (Vietnam,
Myanmar, Indonesia, Thailand, Philippines, China, India, South
Korea and Japan), were obtained from the USDA [15]. The cumulative
number of COVID-19 infected persons in Japan (as of June 6, 2020)
relative to 1 million people was used as the dependent variable. In
Japan, the age-related distribution of COVID-19 patients could be
obtained, so rice consumption was estimated using the same age
categories. Rice consumption by age categories was calculated by
using lifetime rice consumption as the denominator. For example,
if people were in their 50s, the cumulative total for the previous 55
years was divided by 55 to obtain the annual consumption.
Correlation between rice consumption and IgA deficiency
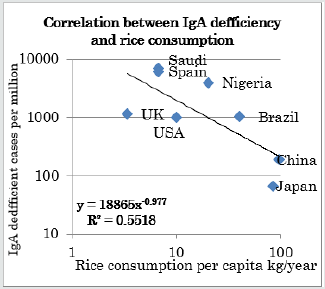
In Figure 1, the horizontal axis shows rice consumption per capita (kg/year) and the vertical axis shows the number of IgA deficient people per million. Both axes use logarithmic scales. The coefficient of determination is 0.55, confirming a negative correlation.
Rice consumption and COVID-19 incidence in Asian countries
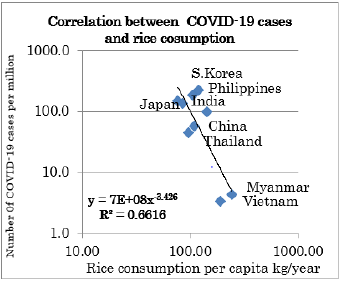
The next survey was conducted on the number of infected people in nine Asian countries. A negative correlation was also confirmed by coefficient of determination is 0.55 in Asian countries (Figure 2).
We investigated the transition of rice consumption in nine Asian countries for 60 years, according to world food statistics. Rice consumption increased in Vietnam and Myanmar, where the number of infected people is low, while rice consumption was halved in Japan and South Korea, where the number of infected people was high. There was little change in rice consumption in the other five countries (Figure 3).
Number of infected people and rice consumption in Japan by age categories
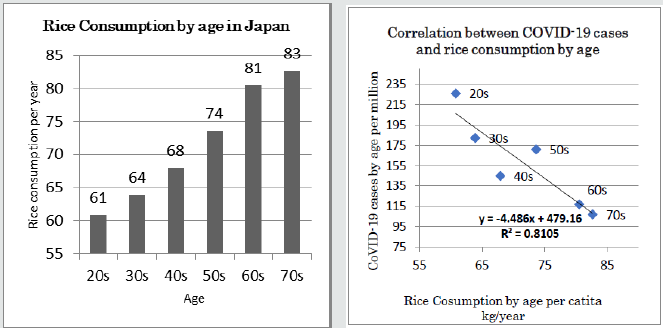
The annual per capita rice consumption of Japanese people has halved in 50 years due to the Westernization of food, as shown in Figure 3. On the other hand, according to the Nikkei Corona Statistics of 2020/6/10, the age distribution of people infected with SARS-CoV-2 shows a higher incidence in younger age groups [16]. Figure 4 shows the correlation between the number of infected people (per 100,000 population) in Japan and rice consumption by age group (20s to 70s). The coefficient of determination is very high at 0.82. Infection resistance seemed to decrease as rice is no longer the main staple food.
Figure 3: Changes of rice consumption in Asian countries High consumption was kept in Myanmar and Vietnam. Most countries kept more than 80kg/year, but Korea and Japan decreased below the 60kg/year.
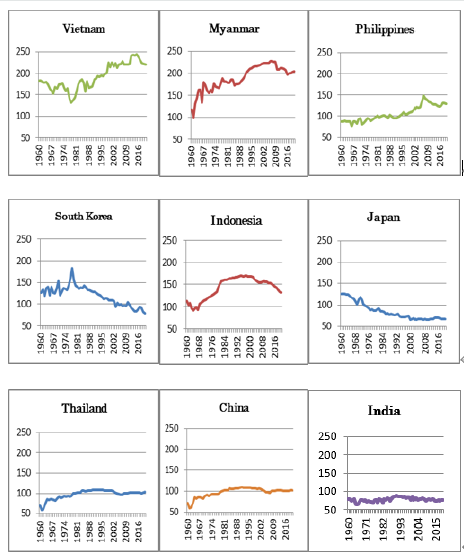
Figure 4: Estimated rice consumption in age category in Japan and age-related COVID-19 incidence. Younger generations who left rice eating habit showed a higher incidence.
Discussion and Conclusions
Our data suggested that infection with SARS-CoV-2 coronavirus
is strongly affected by the nature of staple foods [3]. The reason
would be that rice dieters have a favorable intestinal microbiota
profile, which strengthens their innate immunity to suppress
COVID-19 infection [2]. This is more clearly shown by the lower
incidence of COVID-19 observed in East-Asia, typically in Vietnam
and Myanmar. In Japan and South Korea, rice consumption has
been decreasing almost by half, and younger people showed a
higher incidence of COVID-19. An exception is Taiwan, where the
COVID-19 incidence rate has remained low despite a markedly
decreased consumption of rice. Dietary factors other than rice may
be involved, like vegetables [17,18]. However, Italy and Spain, where
Medeteranean diet is common have not shown signs of suppression
of corona pandemic [4-19]. In Taiwan another factor would be the
strong leadership of public health specialists, who took measures
based on their experience of SARS. This confirms the importance of
political leadership for the success of public health measures. The
epidemiological situation of the new coronavirus pandemic varies
greatly from country to country. Why has Japan been less affected?
Professor Shinya Yamanaka of Kyoto University has called upon
the world to broaden investigations of an unknown factors X. The
authors think that “X = rice”, and that rice eaters have better innate
immunity. This hypothesis is supported by a statistically significant
negative correlation between the prevalence of IgA-deficient
people and the magnitude of local rice consumption. The coefficient
of determination is 0.75 for a total population of 4.5 billion people
in 19 major countries (58% of the world population). Furthermore,
in the Japanese subpopulation the negative correlation is even
stronger (R2 = -0.82) after adjustment for age. Based on the above,
the authors believe that rice eaters have higher immunocompetence
and thus suppress more easily SARS-CoV-2 infection.
It is expected that the pandemic due to virus infection will
hit again. Among future measures, it is important to scale up the
capacity of health systems, but it is difficult to predict if vaccines will
be available, and there are economic constraints to their deployment
in developing countries. As measures against virus infection
from a medium to long-term perspective, it is recommended to
incorporate rice in the diet, promote the use of brown rice and rice
bran, and review the capacity of rice farming. It would be the best
way to keep the advantage of intestinal innate immunity [20-22].
Improvements in agricultural policies and eating habits, especially
the promotion of brown rice and rice bran, are expected to be
effective strategies to counter the COVID-19 pandemic [23,24]. The
main functional nutrients contained in rice are vitamins, minerals,
and antioxidants, with decreasing relative contents as one moves
from rice bran, brown rice, polished rice and flour. The mechanisms
whereby rice strengthens the intestinal innate immunity relate to
dietary fibers and short-chain fatty acids produced by particular
intestinal bacteria which stimulate regulatory T cell proliferation
[25,26]. The suppression of cytokine storm seemed to be effective
to prevent severe COVID-19 infections. We regard brown rice as
“Medical Rice” and we are working to promote its spread [27,28].
It should be noted that with the advance of food in Europe and the
United States, rice consumption in Korea, Taiwan, as well as Japan,
has halved in 50 years. If this tendency is left unchecked, there is a
risk that resistance to infection will fall to similar levels seen in the
West.
Acknowledgement
The authors deeply appreciate Dr. Philippe Calain for his sincere discussion and editing of this manuscript.
COI
The authors declare that there are no conflicts of interest.
References
Pneumonia. Int J Fam Med Prim Care 1(3): 1011-1014.
- Hirakawa A, Aoe S, Watanabe S, Hisada T, Mocizuki J, et al. (2019) The nested study on the intestinal microbiota in GENKI Study with special reference to the effect of brown rice eating. J Obes Chronic Dis 3(1): 11-13.
- Watanabe S, Inuma K (2020) Low COVID-19 Infection and Mortality in Rice Eating Countries Scholarly Journal of Food and Nutrition 3(1): 326-328.
- The Visual and Data Journalism Team. Coronavirus pandemic: Tracking the global outbreak (2020).
- Russell TW, Hellewell J, Jarvis CI, van Zandvoort K, Abbott S, et al. (2020) Estimating the infection and case fatality ratio for coronavirus disease (COVID-19) using age-adjusted data from the outbreak on the Diamond Princess cruise ship, February2020. Euro Surveill 25(12): 2000256.
- Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, et al. (2020) Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area. JAMA 323(20): 2052-2059.
- Yazdani R, Azizi G, Abolhassani H, Aghamohammadi A (2017) Selective IgA deficiency: Epidemiology, pathogenesis, clinical phenotype, daiagnosis, prognosis and management. Scand J Immunol 85(1): 3-12.
- Yel L (2010) Selective IgA deficiency. J Clin Immumol 30(1): 10-16.
- Cunningham-Rundles C (2001) Physiology of IgA and IgA deficiency. J Clin Immunol 21(5): 303-309.
- Kanoh T, Mizumoto T, Yasuda N (1986) Selective IgA deficiency in Japanese blood donors: frequency and statistical analysis. Vox Sang 50: 81-86.
- Watanabe S, Naito Y, Yamamoto T (2020) Host Factors That Aggravate COVID-19 Pneumonia. Int J Fam Med Prim Care 1(3): 1011.
- IBM (2020) SPSS Ver24. Tokyo, Japan.
- 2020 Ito S, Per capita consumption of rice, wheat and corn in the world and their changes. Kyushu University. Faculty of Agriculture. World Food Statistics and Graphics.
- Masashi Idogawa, Shoichiro Tange, Hiroshi Nakase, Takashi Tokino (2020) Interactive web-based graphs of novel coronavirus COVID-19 cases and deaths per population by country. Clinical Infectious Diseases 71(15): 902-903.
- USDA.
- https://vdata.nikkei.com/newsgraphics/coronavirus-japan-chart/#d7
- Wahlqvist ML (2016) Food structure is critical for optimal health. Food Funct 7(3): 1245-1250.
- Wahlqvist ML, Specht RL (1998) Food variety and biodiversity: Econutrition. Asia Pac J Clin Nutr 7(3/4): 314-9.
- Knoops KT, de Groot LC, Kromhout D, Perrin AE, Moreiras-Varela O, et al. (2004) Mediterranean diet, lifestyle factors, and 10-year mortality in elderly European men and women: the HALE project. JAMA 292(12): 1433-9.
- Henderson LA, Canna SW, Schulert GS, Volpi S, Lee PY, et al. (2020) On the Alert for cytokine storm: Immunopathology in COVID-19. Arthritis Rheumatol 72(7): 1059-1063.
- Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, et al (2020) COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 395(10229): 1033-1034.
- Tay MZ, Poh CM, Rénia L, MacAry PA, Ng LFP (2020) The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol 20(6): 363-374.
- Watanabe S, Hirakawa A, Mizuno S, Wakino S, Adachi K (2020) What should we eat for healthy longevity? Necessity of tailor-made nutrition. Acta Scientific Nutritional Health 4(3): 01-05.
- Wahlqvist ML, Keatinge JD, Butler CD, Friel S, McKay J, et al. (2009) A Food in Health Security (FIHS) platform in the Asia-Pacific Region: the way forward. Asia Pac J Clin Nutr 18(4): 688-702.
- Hamer HM, Jonkers D, Venema K, Vanhoutvin S, Troost FJ, et al. (2008) Review article:the role of butyrate on colonic function. Aliment Pharmacol Ther 27: 104-19.
- Bachem A, Makhlouf C, Binger KJ, de Souza DP, Tull D, et al. (2019) Microbiota-derived short-chain fatty acids promote the memory potential of antigen-activated CD8(+) T cells. Immunity 51(2): 285-97.
- Watanabe S, Hirakawa A, Nishijima C (2016) Food as medicine: The new concept of “medical rice”. Adv Food Technol Nutr Sci Open J 2(2): 38-50.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...