Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4706
Research Article(ISSN: 2637-4706) 
Cytotoxicity and Antioxidant Potentials of Barringtonia Asiatica Essential Oil Volume 2 - Issue 2
Isaac John Umaru1,2*, Chinedu Imo2, Kayode Arowora2, Silas Verwiyah Tatah2 and Hauwa A Umaru3
- 1Faculty of Resource Science and Technology, University of Malaysia Sarawak, Malaysia
- 2Department of Biochemistry, Federal University, Nigeria
- 3Department of Biochemistry, Modibo Adama University of Technology, Nigeria
Received: August 11, 2018; Published: August 21, 2018
Corresponding author: Isaac John Umaru, Faculty of Resource Science and Technology, University of Malaysia Sarawak, Malaysia
DOI: 10.32474/DDIPIJ.2018.02.000131
Abstract
Essential oils are liquid mixture of volatile compound obtained from aromatic plants. It was observed that many essential oils have antioxidant properties and can present antimicrobial activities. The Aim of the present study were:
a) to determine the cytotoxicity lethality of the leafs and stem-bark of Barringtonia asiatica using brine shrimp (Artimia salina).
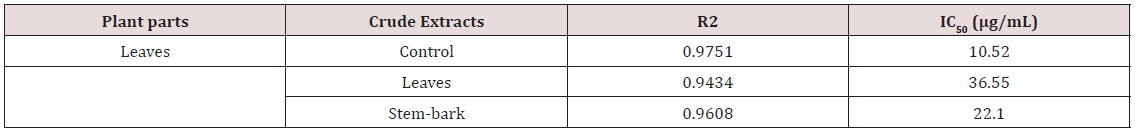
b) to evaluate the antioxidant potential of composition of the leaf and stem-bark essential oil using the DPPH (2,2-diphenyl-1- picrylhydrazyl) free radical scavenging assay. The essential oil exhibited a mild toxicity and significant IC50 of 36.55μg/mL and 22.10μg/mL value of antioxidant potential leaf and stem-bark of Barringtonia asiatica respectively.
Keywords: Barringtonia asiatica; essential oil; cytotoxicity; antioxidant
Introduction
Right from ancient time drugs of plants have been used as herbal formulations in crude forms, like tinctures, teas, powders and poultices, for their growing interest as alternative therapies for the prevention or treatment of various diseases [1]. Essential oils from medicinal and aromatic plants are still considered as rich sources of a huge number of antimicrobial and antifungal components [2]. Many of them show a great potential as anticancer therapeutic agents [3]. However, essential oils are chemically characterized as complex mixtures of low molecular weight compounds and, some of them, are highly volatile and capable of generating flavors and aromas [4]. Scientific studies have shown the role of essential oils in biological interactions among plants and their potential therapeutic including anti-inflammatory, analgesic, anti-tumor, antifungal, and antibacterial activities [5- 9]. In addition to the above-mentioned properties, many essential oils have been confirmed to possess antioxidant activity. They are extremely important for disease prevention, since they inhibit and delay the oxidation of biomolecules by preventing the initiation or propagation of chain oxidation reactions [10-12]. Thus, because of the harmful effects that synthetic antioxidants may cause, such as toxicity and carcinogenicity, the interest in the discovery of natural antioxidants has increased considerably [13]. The growing interest in natural bioactive compounds has led to conduct further studies addressing the replacement of synthetic chemical agents in the industrial sector, since natural products are less harmful to health [14], in addition to being biodegradable and usually exhibiting low toxicity in mammals [15]. This plant Barringtonia asiatica was considered to extract and test for its cytotoxicity and antioxidant potentials. However, Barringtonia asiatica is one of the species belonging to the family Barringtonia, native to mangrove habitats on the tropical, it is a common plant in the Malaysian Mangroves and wetlands such as the Kuching wetlands Sarawak and Bako National Park, it is also found in tropical Africa, Nigeria and Madagascar. Other species belonging to this genus are Barringtonia racemosa, Barrington acutangula, Barringtonia edilus, Barringtonia lanceolate, Barringtonia macrostachya, and Barringtonia spicata [16]. The literature survey revealed that there are no scientific Citation: Isaac J U, Chinedu I, Kayode A, Silas V T, Hauwa A U. Cytotoxicity and Antioxidant Potentials of Barringtonia Asiatica Essential Oil. Drug Des Int Prop Int J 2(2). DDIPIJ.MS.ID.000131. DOI: 10.32474.DDIPIJ.2018.02.000131. 173 studies carried out regarding antioxidant and cytotoxicity of the essential oil of the leaves, stem- barks of Barringtonia asiatica. Hence, the present study is focused to evaluate the antioxidant potentials including determining cytotoxicity (leafs and stembarks) by using Artemia salina and 2,2-diphenyl-1-picrylhydrazyl.
Materials and Methods
Extraction of Essential Oil
The plant samples were subjected to water distillation for 8 hours using Clevenger apparatus to extract the oils quantitatively, according to method described by Samsiah et al. [17] and Fasihuddin & Ibrahim [18]. Approximately 100 g of fresh cut and ground samples of Barringtonia asiatica leaf and stem bark were weighted, transferred into 2 L round flask and mixed with 1.35 L of distilled water. The flask was assembled to the Clevenger trap, connected to the condenser and heated. The sample was heated for 8 hours for hydro distillation process. After 8 hours, the oil trapped in the Clevenger was left to cool at room temperature. The water layer at the bottom of the oil was first drained out to separate it with the oil. The collected oily layer was treated with anhydrous sodium sulphate to remove any trace of water. The experiment was performed triplicates for each sample. The essential oil obtained was kept in a vial and stored in refrigerator at 4°C prior to analysis.
Brine Shrimp (Artemia salina) Lethality Test
Toxicity test against brine shrimp (Artemia salina) developed by McLaughlin et al. [19] was used in this study. Leached brine shrimp eggs were hatched in seawater and incubated for 48 hours at 25ºC. Exactly 3 mg of sample was dissolved in 3mL methanol, and the mixture was sonicated to ensure homogeneity of the extract. Four different volumes of 500, 250, 50 and 5μL each from the stock solution were transferred into NUNC multidish in triplicate. Solvent was allowed to evaporate under a running fume hood for overnight and followed by the addition of 0.2mL DMSO and 4.8mL seawater to give final concentration of 100, 50, 10 and 1μg/mL, respectively. Ten brine shrimp nauplii were transferred into each concentration in NUNC multidish, and was observed every 6 hours for 24 hours. The amount of dead nauplii were calculated. Thymol was used as positive control, whereas 0.2mL DMSO and 4.8mL seawater was used as negative control. The data was analysed to determine the concentration of the samples that kill 50% of brine shrimp at 24 hours or known as LC50. LC50 was calculated and determined by performing Probit analysis in IBM SPSS Statistic software of version 21.
DPPH (2,2-diphenyl-1-pycryl-hydrazyl) Free Radical Scavenging Assay
The free radical scavenging assay of compound 2,2-diphenyl- 1-pycryl-hydrazyl (DPPH) was used to evaluate the antioxidant properties of the crude extract and the essential oil. The measurement was based on the method described by Wang et al. [20]. The sample was prepared by diluting 6 mg of crude extract into 6 mL of methanol, producing a concentration of 1000μg/mL. The stock solution was sonicated to ensure the homogeneity of the sample. Three other concentrations were prepared at 10, 50 and 100μg/mL, diluted from the 1000μg/mL stock solution. Sample of 5000μg/mL was prepared separately by diluting 25 mg of crude extract into 5 mL of methanol. Approximately 3 mL of 0.1mM solution of 2,2-diphenyl-1-pycrylhydrazyl (DPPH) in methanol was each added into five series of prepared concentrations (10, 50, 100, 1000 and 5000μg/mL) of sample solutions (1mL). Analysis was done in triplicate. The solution was mixed vigorously and left to stand at room temperature for 30 minutes in the dark after which its absorbance was measured spectrophotometrically at 517 nm using Jasco ultra violet spectrophotometer model V-630. Methanol was used as blank (only methanol) and negative control (1 mL methanol mixed with 3 mL DPPH), while ascorbic acid (vitamin C) as the standard. The concentration of the sample required to inhibit 50% of the DPPH free radical was calculated as IC50 and the value was determined using Log dose inhibition curve which performed by using PRISM version 3.02 software, based on the calculated values of the DPPH scavenging activity (%) of the sample [21]. DPPH scavenging activity (%) was calculated with formula = A0 - A1/A0 x 100, where A0 was the absorbance of the control, while A1 was the absorbance in presence of the sample. making a concentration of 1000μg/mL. The procedure used was similar to the procedure used to evaluate the antioxidant properties of the crude extract, as mentioned earlier.
Statistical Analysis
Statistical analysis for biological activities was performed using SPSS programme. The LC50 values for toxicity assay was calculated using the Probit Analysis option in the SPSS. The IC50 for DPPH free radical scavenging assay was statistically determined using Log dose inhibition curve in PRISM programme.
Results
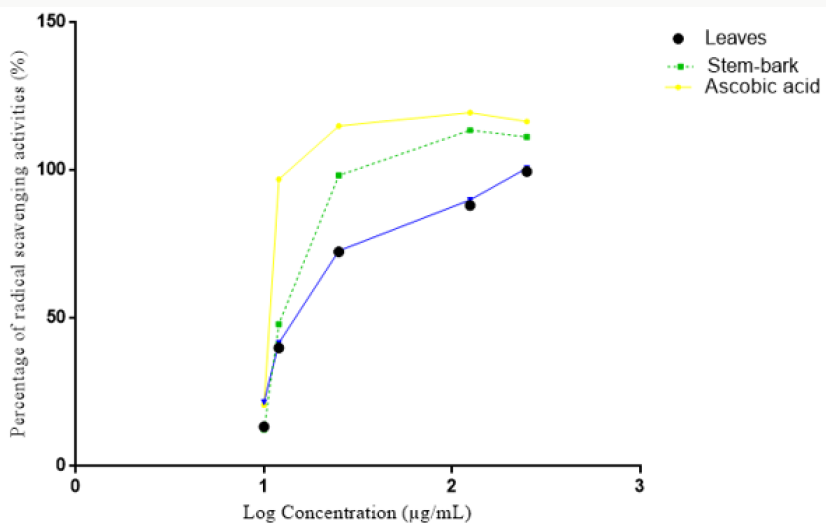
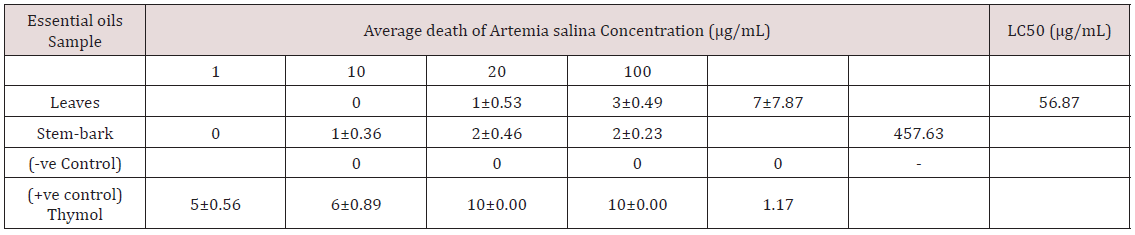
The plant samples were subjected to water distillation for 8 hours using Clevenger apparatus to extract the oils quantitatively. The antioxidant studies of the essential oils of Barringtonia asiatica leaves and stem bark extract indicated higher scavenging activity against DPPH with the IC50 values of 36.55 and 22.10μg/mL respectively. The result also suggested that, the Leaves and stembark cytotoxicity LC50 are 56.87 and 457.63μg/mL for the essential oil. This show the cytotoxicity of the oil. The samples of Barringtonia asiatica leaves and stem bark extract might capture less free radicals formed by DPPH resulting an increase of absorbance and decrease in IC50 value (Tables 1 & 2) and (Figure 1).
Table 1: Average death of Artemia salina at different concentrations of essential oils of Leafs and Stem-bark of Barringtonia asiatica.
Discussion
The essential oil of the plant parts of Barringtonia asiatica tested showed a significant lervical activity when compared with the control and the report of [22]. The lethality concentration of the leaves and the stem-bark essential oil were observed at 1μg/mL -100 μg/mL as presented on the table above. The cytotoxicity for the leaves and the stem-bark essential oil LC50 to be (56.87μg/mL and 457.63μg/mL) respectively. The leaves were indicated to be higher when compared to the is positive control (Thymol) LC50 (1.17) and very toxic when compared to the fact that less than 1000μg/mL toxic as reported by Meyer et al. [22]. Antioxidants are tremendously important substances which possess the ability to protect the body from damage caused by free radical induced oxidative stress. The antioxidant potential of the essential oil of Barringtonia asiatica leaves and stem-bark extracts was investigated in the search for new bioactive compounds from natural resources. It became clear that the Barringtonia asiatica leaves and stem-bark oil present the highest antioxidant activity compared with reference Antioxidant Vitamin C for DPPH scavenging activity. However, differences of essential oil’s antioxidant activity might be observed in some species which may likely be different in different location due to the variation in their chemical composition [23]. Thus, the leaves and stem-bark extract showed a good antioxidant activity with IC50 value of (10.52 and 36.55μg/mL) respectively. This indicated that the plant parts of Barringtonia asiatica shown a promising candidate for use as natural products based-antioxidant for the health of human being.
Conclusion
This study showed that Barringtonia asiatica (L.) essential oil from leaves and Stem-bark have high antioxidant activity. Thus, further studies should be carried out to establish the bioactive compound, its application and practicability of incorporating Barringtonia asiatica (L.) essential oil in any healthy processed products such as capsulated daily supplements.
Acknowledgement
The authors acknowledge to Federal University Wukari for study fellow and Universiti Malaysia Sarawak for Laboratory facility and the support of ZAMALA UNIMAS.
References
- Chhetri BK, Ali NAA, Setzer WN (2015) A survey of chemical compositions and biological activities of Yemeni aromatic medicinal plants. Medicines 2(2): 67-92.
- Said-Al Ahl HA, Sarhan AM, Dahab ADMA, Abou-Zeid ESN, Ali MS, et al. (2015) Essential Oils of Anethum graveolens L.: Chemical Composition and Their Antimicrobial Activities at Vegetative, Flowering and Fruiting Stages of Development. International Journal of Plant Science and Ecology 1(3): 98-102.
- Russo R, Corasaniti MT, Bagetta G, Morrone LA (2015) Exploitation of cytotoxicity of some essential oils for translation in cancer therapy. Evidence-Based Complementary and Alternative Medicine.
- Trombetta D, Castelli F, Sarpietro MG, Venuti V, Cristani M, et al. (2005) Mechanisms of antibacterial action of three monoterpenes. Antimicrobial agents and chemotherapy 49(6): 2474-2478.
- SIANI AC, Siani AC, SIANI AC, SIANI AC, Siani AC, et al. (2000) Óleos essenciais: potencial antiinflamatório. 16: 38-43.
- Silva J, Abebe W, Sousa SM, Duarte VG, Machado MIL, et al. (2003) Analgesic and anti-inflammatory effects of essential oils of Eucalyptus. Journal of ethnopharmacology 89(2-3): 277-283.
- De Sousa AC, Gattass CR, Alviano DS, Alviano CS, Blank AF, et al. (2004) Melissa officinalis L. essential oil: antitumoral and antioxidant activities. Journal of pharmacy and pharmacology 56(5): 677-681.
- Osei-Safo D, Addae-Mensah I, Garneau FX, Koumaglo HK (2010) A comparative study of the antimicrobial activity of the leaf essential oils of chemo-varieties of Clausena anisata (Willd.) Hook. f. ex Benth. Industrial Crops and Products 32(3): 634-638.
- Kaileh M, Berghe WV, Boone E, Essawi T, Haegeman G (2007) Screening of indigenous Palestinian medicinal plants for potential anti-inflammatory and cytotoxic activity. Journal of ethnopharmacology 113(3): 510-516.
- Kaur C, Kapoor HC (2001) Antioxidants in fruits and vegetables–the millennium’s health. International journal of food science & technology 36(7): 703-725.
- Bamoniri A, Ebrahimabadi AH, Mazoochi A, Behpour M, Kashi FJ, et al. (2010) Antioxidant and antimicrobial activity evaluation and essential oil analysis of Semenovia tragioides Boiss. from Iran. Food chemistry 122(3): 553-558.
- Pandini JA, Pinto FGS, Scur MC, Santana CB, Costa WF, et al. (2017) Chemical composition, antimicrobial and antioxidant potential of the essential oil of Guarea kunthiana A. Juss. Brazilian Journal of Biology (ahead) 78(1): 53-60.
- Losso JN, Shahidi F, Bagchi D (Eds.) (2007) Anti-angiogenic functional and medical foods. CRC Press.
- Gao CY, Lu YH, Tian CR, Xu JG, Guo XP, et al. (2011) Main nutrients, phenolics, antioxidant activity, DNA damage protective effect and microstructure of Sphallerocarpus gracilis root at different harvest time. Food chemistry 127(2): 615-622.
- Figueiredo AC, Barroso JG, Pedro LG, Scheffer JJ (2008) Factors affecting secondary metabolite production in plants: volatile components and essential oils. Flavour and Fragrance journal 23(4): 213-226.
- Isaac John Umaru, Fasihuddin A, Badruddin, Zaini B Assim, Hauwa A Umaru (2018) Cytotoxicity (Brine shrimp Lethality Bioassay) of Barringtonia racemosa Leaves, Stem-Bark and Root Extract. Journal of Biotechnology and Bioengineering 2(2): 45-50.
- Samsiah J, Fasihuddin BA, Laily D, Zuriati Z (2015) Essential oils from different parts of Goniothalamus ridleyi plant. Sains Malaysiana 44(11): 1579-1585.
- Fasihuddin BA, Ibrahim J (2003) Chemical constituents of the essential oils of Goniothalamus uvariodes King. Flavour and Fragrance Journal 18(2): 128-130.
- McLaughlin JL (1991) Crown gall tumours on potato disc and brine shrimp lethality: two simple bioassays for higher plants screening and fractionation. Assay for Bioactivity. San Diego: Academic Press. pp. 2-32.
- Wang H, Zhao M, Yang B, Jiang Y, Rao G (2008) Identification of polyphenols in tobacco leaf and their antioxidant and antimicrobial activities. Food Chemistry 107(4): 1399-1406.
- Tailor CS, Goyal A (2014) Antioxidant activity by DPPH radical scavenging method of Ageratum conyzoides Linn. leaves. American Journal of Ethnomedicine 1(4): 244-249.
- Meyer BN, Ferrigni NR, Putnam JE, Jacobsen LB, Nichols DE, et al. (1982) Brine shrimp: a convenient general bioassay for active plants constituents. Plant Med 45(5): 31-34.
- Pattamapan L, Kittisak S, Phanida P, Worawan K, Krit T, et al. (2015) In vitro biological activities of black pepper essential oil and its major components relevant to the prevention of Alzheimer’s disease. The Thai Journal of Pharmaceutical Sciences 39(3): 94-101.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...