Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4706
Mini Review(ISSN: 2637-4706) 
Current Challenges and Obstacles to Drug Development for Chagas Disease Volume 2 - Issue 2
Cauê Benito Scarim* and Chung Man Chin
- Department of Drugs and Medicines, São Paulo State University (UNESP), Brazil
Received: August 29, 2018; Published: September 04, 2018
Corresponding author: Cauê Benito Scarim, Department of Drugs and Medicines, São Paulo State University (UNESP), Brazil
DOI: 10.32474/DDIPIJ.2018.02.000134
Abstract
Chagas disease is a protozoan infection which was first identified more than one hundred years ago. But even now, drugs to treat the latent and chronic phases of the disease are not available. The success of a drug design is highly dependent on activity and toxicity. In the case of Chagas disease, questions remain as to identifying the best treatment (mainly for the chronic phase), and how new drugs and drug combinations compare to current therapy. The principal priority of this mini-review is to report the enormous difficulty that pharmaceutical chemists encounter in the development of new drugs for neglected tropical infectious diseases, in this case Chagas disease.
Keywords: Drug Development; New Drugs; Chagas Disease; T. cruzi
Introduction
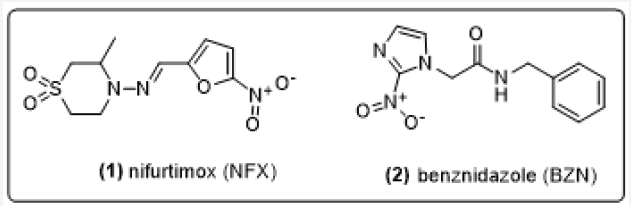
Chagas disease is one of the 17 neglected diseases and was identified 109 years ago by the Brazilian physician and researcher Carlos Justiniano Ribeiro Chagas. Also known as American trypanosomiasis, Chagas disease is endemic in 21 Latin American countries [1-5]. Due to an increasingly globalized world, this parasite has spread to the United States, Japan, Australia, Canada, and even across the European continent [6,7]. It is caused by the hemoflagellate protozoan from the Kinetoplastida order of the Trypanosamatidae family, called Trypanosoma cruzi [2,8,9]. Treatment is restricted to two nitroheterocyclic drugs: nifurtimox (1) and benznidazole (2) (Figure 1). These were discovered during the 1960s and 1970s, respectively [10,11]. Both drugs are more effective in the acute phase of the disease than in the indeterminate and chronic phases [12-21]. Furthermore, there are differences in the efficacies of these drugs in relation to the several strains of T. cruzi [22] (Figure 1).
Figure 1: Structures of available chemotherapy agents for Chagas disease: nifurtimox and benznidazole.
Challenges and Obstacles in Research and Development of New Anti-Chagasic Drugs
Medicinal chemistry is a multidisciplinary area involving knowledge related to biology, chemistry, and the medical and pharmaceutical sciences [23]. Medicinal chemists are constantly searching for bioactive compounds for various diseases, including effective compounds for tropical infectious diseases, such as Chagas disease. However, since tropical diseases do not typically arouse the attention of pharmaceutical companies due to low financial returns (considering the low income of affected people), medicinal chemists are often not motivated to pursue research [24]. The interest of the pharmaceutical industry is extremely important. Characteristics that are indispensable for the search for new lead compounds for Chagas Disease drug development include high activity (< 5μM), low toxicity (ideal selectivity index: >10), and the validation of new molecular targets [25,26]. Overall, neglected tropical diseases (in this case Chagas disease) represent strong obstacles to the development of countries and subsequently maintain the framework of worldwide socioeconomic inequality (Figure 2).
Scientific Expectations
There is an urgent need for new drugs that have potent activity in the chronic phase of Chagas disease. Concerning toxicity, these drugs need to be safe. If the current medicinal chemistry scaffold continues without a solution, there will be a need for combined drug treatments in the future to decrease blood and tissue parasitology, reduce considerably the percentage of inflammatory infiltrates, and restrict fibrin networks. The optimal experimental protocol is the one that best relates to human response. The data reported in the mini-review published by Scarim and co-workers [27] suggest that for experimental protocol in animals chronically infected with Chagas disease, a treatment with lower doses of benznidazole is more advantageous and can be used as a base to compare new drugs with BZN in the chronic phase of Chagas disease.
Conclusion
More than 100 years after the initial identification of Chagas disease, despite all recent advances and new knowledge about this disease, we still lack an effective treatment against all phases of this disease. Thus, the partnership between governmental and non-governmental institutions with the pharmaceutical industry is extremely important to widen scientific knowledge and research for the treatment of this disease.
Acknowledgment
The authors would like to thank the Programa de Apoio ao Desenvolvimento Coordenação de Aperfeiçoamento Pessoal de Nível Superior (CAPES), Científico da Faculdade de Ciências Farmacêuticas da UNESP (PADC/FCF-UNESP), and the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP - 2016/10847- 9) for research fellowships and financial assistance.
References
- World and Health Organization (WHO) (2010) First report on neglected tropical diseases 2010: working to overcome the global impact of neglected tropical diseases. Geneva: WHO.
- World and Health Organization (WHO) (2015) Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec 90(6): 33-43.
- World and Health Organization (WHO) (2017) Neglected tropical diseases.
- W Feasey, N Wansbrough-Jones, DCW Mabey, AW Solomon (2010) Neglected tropical diseases. Br Med Bull 93(1): 179-200.
- JP Cohen, G Sturgeon, A Cohen (2014) Measuring progress in neglected disease drug development. Clin Therapeut 36(7): 1037-1042.
- JR Dias, JCP Coura (1997) Clínica e terapeutica da doença de Chagas, uma abordagem prática para o clínico geral. Cad Saúde Pública Rio de Janeiro, p. 486.
- Z Andrade (2000) Patologia da Doença de Chagas, In: G Koogan (Eds.), Trypanos. cruzi E Doença Chagas 2º. Rio de Janeiro 1(2): 379-388.
- C Chagas (1909) Nova tripanozomiaze humana: estudos sobre a morfolojia e o ciclo evolutivo do Schizotrypanum cruzi n. gen., n. sp., ajente etiolojico de nova entidade morbida do homem. Mem Inst Oswaldo Cruz 1(2): 159-218.
- G Malafaia, ASDL Rodrigues (2010) Centenario do descobrimento da doença de Chagas: desafios e perspectivas, Centen Do Descob Da Doença Chagas Desafios E Perspect 43(5): 483-485.
- JD Maya, M Orellana, J Ferreira, U Kemmerling, R Lopez-Munoz, et al. (2010) Chagas disease: present status of pathogenic mechanisms and chemotherapy. Biol Res 43(3): 323-331.
- World Health Organization (WHO) (2002), Control of Chagas disease: second report of the WHO expert committee World Health Organization (2000: Brasilia, Brazil), Geneva World Heal. Organ 905: 109.
- ALSS de Andrade, F Zicker, RM de Oliveira, SA Silva, A Luquetti, et al. (1996) Randomised trial of efficacy of benznidazole in treatment of early Trypanosoma cruzi infection. The Lancet 348(9039): 1407-1413.
- S Sosa-Estani, E Segura, A Ruiz, E Velazquez, B Porcel, et al. (1998) Efficacy of chemotherapy with benznidazole in children in the indeterminate phase of Chagas’ disease. Am J Trop Med Hyg 59(4): 526-529.
- GMS Da Silva, MFF Mediano, PEAA Do Brasil, MDC Chambela, DJA Silva, et al. (2014) A clinical adverse drug reaction prediction model for patients with chagas disease treated with benznidazole. Antimicrob Agents Chemother 58: 6371-6377.
- CA Morillo, JA Marin-Neto, A Avezum, S Sosa-Estani, A Rassi Jr, et al. (2015) Randomized trial of benznidazole for chronic Chagas’ Cardiomyopathy. N Engl J Med 373: 1295-1306.
- D Soy, E Aldasoro, L Guerrero, E Posada, N Serret, et al. (2015) Population pharmacokinetics of benznidazole in adult patients with Chagas disease. Antimicrob Agents Chemother 59: 3342-3349.
- ML Fernandez, AR Riarte, ME Marson, G Mastrantonio, JC Ramirez, et al. (2016) Pharmacokinetic and pharmacodynamics responses in adult patients with Chagas disease treated with a new formulation of benznidazole. Mem Inst Oswaldo Cruz 111: 218-221.
- AM Hasslocher-Moreno, PEAA do Brasil, AS de Sousa, SS Xavier, C Chambela, et al (2012) Safety of benznidazole use in the treatment of chronic Chagas’ disease. J Antimicrob Chemother 67(5): 1261-1266.
- CD Fernandes, FM Tiecher, MM Balbinot, DB Liarte, D Scholl, et al. (2009) Efficacy of benznidazol treatment for asymptomatic chagasic patients from state of Rio Grande do Sul evaluated during a three years follow-up. Mem Inst Oswaldo Cruz 104: 27-32.
- MC Andrade, MDF Oliveira, AT Nagao-Dias, ICB Coelho, DDS Candido, et al. (2013) Clinical and serological evolution in chronic Chagas disease patients in a 4-year pharmacotherapy follow-up: a preliminary study. Rev Soc Bras Med Trop 46: 776-778.
- JA Castro, MM deMecca, LC Bartel (2006) Toxic side effects of drugs used to treat chagas’ disease (American trypanosomiasis). Hum Exp Toxicol 25(8): 471-479.
- B Zingales, MA Miles, CB Moraes, A Luquetti, F Guhl et al. (2014) Drug discovery for Chagas disease should consider Trypanosoma cruzi strain diversity. Mem Inst Oswaldo Cruz 109(6): 828-833.
- CG Wermuth, CR Ganellin, P Lindberg, LA Mitscher (1998) Glossary of terms used in medicinal chemistry (IUPAC Recommendations 1998) Pure Appl Chem 70(5): 1129-1143.
- R Pink, A Hudson, MA Mouriès, M Bendig (2005) Opportunities and challenges in antiparasitic drug discovery. Nat Rev Drug Discov 4(9): 727-740.
- D Vanderelst, N Speybroeck (2010) Quantifying the lack of scientific interest in neglected tropical diseases. PLoS Negl Trop Dis 4(1): e576.
- E Chatelain, JR Ioset (2018) Phenotypic screening approaches for Chagas disease drug discovery. Expert Opinion on Drug Discovery 13(2): 141-153.
- CB Scarim, AR Ribeiro, JA da Rosa, MC Chung (2018) Response to different benznidazole doses in animal models of chronic phase Chagas disease: a critical review. Rev Soc Bras Med Trop 51(2): 133‐140.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...