
Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2643-6760
Case Report(ISSN: 2643-6760) 
A Rare Case of Aggravated Hypercalcemia of Malignancy After Surgery Volume 6 - Issue 4
Eun Jang Yoon1, Seungho Jung1, Sungwon Na1, Jiwoo Suh1, Tae-Hwan Yoon2 and Jeongmin Kim1*
- 1Department of Anesthesiology and Pain Medicine, Anesthesia and Pain Research Institute, Yonsei University, South Korea
- 2Department of Orthopaedic Surgery, Arthroscopy and Joint Research Institute, Severance Hospital, Yonsei University, South Korea
Received:July 16, 2021; Published:July 30, 2021
Corresponding author: Jeongmin Kim, Department of Anesthesiology and Pain Medicine, Anesthesia and Pain Research Institute, Yonsei University, South Korea
DOI: 10.32474/SCSOAJ.2021.06.000242
Abstract
Paraneoplastic syndromes are caused by ectopic hormone production in malignant tumors. Hypercalcemia of malignancy is the main manifestation of paraneoplastic syndromes and can lead to acute kidney injury, life-threatening arrhythmia, and cardiac arrest. Here, we present a 70-year-old Asian female with a probable diagnosis of advanced lung cancer with hypercalcemia of malignancy. She underwent surgery to treat a pathologic fracture of the humerus. In this case, hypercalcemia of malignancy was unexpectedly aggravated after surgery, and hypercalcemic complications led to a poor outcome for the patient.
Abbreviations: HCM: Hypercalcemia of Malignancy; BPM: Beats Per Minute; ICU: Intensive Care Unit; AKI: Acute Kidney Injury; POD: Postoperative Day; PVC: Premature Ventricular Complexes; CRRT: A Continuous Renal Replacement Therapy; HHM: Humoral Hypercalcemia of Malignancy; IL: Interleukin; ECG: Electrocardiography; PTH: Parathyroid Hormone; PTHrP: Parathyroid Hormonerelated Peptide
Introduction
Paraneoplastic syndromes refer to a cluster of symptoms that result from malignancy but not attributed to direct tumor invasion or compression [1]. Paraneoplastic syndromes are caused by ectopic hormone production. Hypercalcemia develops due to an increase in parathyroid hormone. However, in cancer patients, it is mostly caused by paraneoplastic syndromes due to malignancy [2]. HCM occurs in more than 30% of patients with cancer, and 8.6% of them have bone metastases [3,4]. In HCM, the mortality rate is 50% within a month and 75% within three months of starting treatment [5]. Lung cancer is the most common cancer associated with HCM, observed in 12.5% of patients with advanced lung cancer [6]. In addition, the survival rate of patients with pathologic metastatic fractures is the lowest at 110 days for patients with lung cancer compared with those with other cancers [7]. The outcomes of HCM can range from mild symptoms to serious complications [2]. Mild symptoms include poor oral intake, general weakness, somnolence, constipation, anorexia, polyuria, and hypertension. Serious complications include psychosis, coma, renal failure, pancreatitis, life-threatening arrhythmia, and cardiac arrest. Here, we present a case of a 70-year-old woman with a probable diagnosis of lung cancer who underwent surgery for a pathologic fracture of the humerus.
Case Report
A 70-year-old woman (height, 160 cm; weight, 65 kg) was hospitalized for surgery at an orthopedic outpatient clinic with a fracture of the right humerus. The patient had a history of breast cancer, stroke, and patent foramen ovale (grade 1). The patient could climb two flights of stairs prior to the injury; however, she had bloody sputum for the past month and experienced dyspnea on exertion. Reduced breathing sound accompanied by crackle was auscultated in the right middle and right lower lobes. On noncontrast chest computed tomography, a huge mass of around 10 cm was observed in the right middle and right lower lobes with associated mucoid impaction and patchy ground-glass opacity, suggesting primary lung cancer.
The patient underwent open reduction and internal fixation for the right humerus fracture. On the day of surgery, electrocardiography, pulse oximetry, continuous arterial blood pressure monitoring in the left radial artery, and tympanic temperature measurement were performed. Initially, her blood pressure was 127/76 mmHg, heart rate was 116 bpm, and SpO2 was 95–97% with 7 L/min oxygen supplied by a nasal cannula. Regional anesthesia was performed with ultrasound-guided supraclavicular brachial plexus block using 0.75% ropivacaine (90 mg) and 2% lidocaine (240 mg). The initial corrected calcium level was 13.4 mg/dL (3.35 mmol/L); thus, we started hydration to treat hypercalcemia (Figure 1). The operation was uneventful. Blood pressure was decreased at 15 min after the start of surgery. Norepinephrine was infused and increased to 0.06 μg/kg/min.
Following norepinephrine infusion, blood pressure was well maintained during the operation, and the lowest blood pressure was 86/52(65) mmHg. After the surgery, she was admitted to the ICU for close monitoring.
Figure 1: Preoperative and postoperative corrected calcium levels: This graph shows changes in the corrected calcium levels over the course of hospital admission of the patient. The serum level of calcium was markedly increased immediately after the operation and was decreased following dialysis at 30 h after the operation. Mild hypercalcemia corresponds to a level of 10.5 to 11.9 mg/dL, moderate hypercalcemia corresponds to a level of 12.0 to 13.9 mg/dL, and severe hypercalcemia corresponds to a level above 14.0 mg/dL [3]. *Ca: calcium, Preop: preoperative period, Postop: postoperative period.
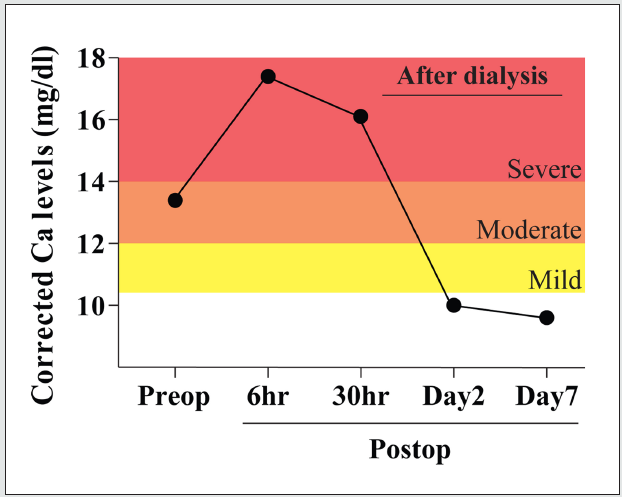
The patient’s initial mental status was alert in the ICU but soon changed to drowsy, confused, and disoriented. The patient also complained of constant thirst at 3 h after admission to the ICU. Notably, the corrected calcium level was rapidly increased to 17.4 mg/dL (4.35 mmol/L), which is indicative of severe hypercalcemia (Figure 1). Other laboratory tests revealed newly onset AKI. Therefore, urine output was monitored, and 4 mL/kg/h plasma solution was administered to treat severe hypercalcemia and AKI. In addition, furosemide and zoledronic acid (4 mg/day) were administered. On POD 1, the corrected calcium level remained high at 16.2 mg/dL (4.0 mmol/L). On POD 2, sinus tachycardia with frequent PVCs and persistent hypercalcemia were noted; thus, CRRT was initiated. Hydration, medication, and CRRT normalized serum calcium levels. After CRRT, the mental status was improved to alert and partially disoriented. Frequent PVCs were no longer observed, and the body temperature was gradually decreased from 39℃ to 37.2℃. However, on POD 5, the general health status rapidly deteriorated. Atrial flutter was observed, and hypoxemia was aggravated; thus, mechanical ventilation had to be applied. The patient developed shock, including a high fever, which increased the demand for continuous norepinephrine administration (0.4 μg/ kg/min). One week after surgery, the patient suffered from cardiac arrest and deceased despite active medical intervention.
Discussion
Although several studies have examined the perioperative consideration of patients with hypercalcemia due to primary hyperparathyroidism, reports on patients with HCM are limited. This report highlighted the exacerbation of HCM after surgery. The mechanism of HCM is well known, which is secondary to the secretion of bioactive substances from neoplastic cells [2]. HHM accounts for 80% of the causes of HCM [3]. The etiological factor for hypercalcemia in HHM is PTHrP, which is structurally similar to PTH. PTHrP causes the renal tubular reabsorption of calcium and, at the same time, increases urinary phosphorus excretion, leading to hypercalcemia and hypophosphatemia. Local osteolytic hypercalcemia accounts for 20% of the cases of cancer-related hypercalcemia and is usually associated with extensive bone metastases and skeletal tumor burden. In metastatic bone cancers, local cytokines such as IL-1, IL-3, IL-6, and tumor necrosis factor α are secreted, leading to excessive osteoclast activation. This increases resorption and promotes the local production of PTHrP, resulting in hypercalcemia. In this patient, the postoperative PTH level was as low as 11.7 pg/mL, and the level of PTHrP was increased to 31.3 pmol/L. The preoperative hypercalcemic state of this patient was characterized by the manifestation of paraneoplastic syndromes, which appear to have been caused by HHM due to suspected primary lung cancer or local osteolytic hypercalcemia resulting from bone metastasis.
Hypercalcemia was rapidly worsened after surgery. In the present case, the corrected serum calcium level before surgery was 13.4 mg/dL (3.35 mmol/L) (moderate hypercalcemia); however, after the surgery, it was significantly increased to 17.4 mg/dL (4.35 mmol/L) (severe hypercalcemia). HCM could be exacerbated after surgery for various reasons. Hypercalcemia may be worsened by immobilization and intraoperative hypotension. In addition, the operation itself may be an aggravating factor of hypercalcemia [8]. Surgery can cause an increase in plasma prostaglandin E2 and interleukin-6, leading to complex systemic reactions. After surgery, the high calcium levels in this patient could be attributed to the activation of osteoclasts due to cytokine secretion. Therefore, the possibility of postoperatively exacerbated hypercalcemia in cancer patients with paraneoplastic syndromes should be considered.
The perioperative management of HCM can be inferred from the management of hypercalcemia caused by hyperparathyroidism; however, there is limited information on the management of HCM. It is important to normalize blood calcium levels before surgery with normal saline, phosphate or furosemide, and bisphosphonates [9]. If dehydration and malnutrition are not corrected, hypotension or circulatory failure may occur with a high probability after anesthetic induction. Neuromuscular monitoring is necessary because it is difficult to predict the effects of neuromuscular blockers. Additionally, continuous ECG monitoring is required for the detection of changes in the cardiac rhythm. Hypotension should be avoided as it can lead to acidosis with an increase in ionized calcium [10]; thus, invasive arterial blood pressure monitoring can be beneficial.
Conclusion
Surgery aggravates HCM and affects patient outcomes: thus, HCM should be corrected with bisphosphonate, furosemide, or dialysis before surgery to prevent complications. Tracking calcium levels, ECG monitoring, and neuromuscular monitoring during surgery are also crucial. As demonstrated in this case, healthcare providers should be aware that hypercalcemia could be aggravated during the postoperative period. Therefore, calcium levels should be closely monitored, and it is necessary to prepare for the management of the hypercalcemic state.
References
- Pelosof LC, Gerber DE (2010) Paraneoplastic syndromes: An approach to diagnosis and treatment. Mayo Clin Proc 85: 838-854.
- Clines GA (2011) Mechanisms and treatment of hypercalcemia of malignancy. Curr Opin Endocrinol Diabetes Obes 18: 339-346.
- Stewart AF (2005) Clinical practice. Hypercalcemia associated with cancer. N Engl J Med 352: 373-379.
- Bahk WJ, Chang JH, Lee SK, Moon MS, Woo YK, et al. (1995) Hypercalcemia associated with bone metastasis. J Korean Orthop Assoc 30: 349-354.
- Mirrakhimov AE (2015) Hypercalcemia of Malignancy: An Update on Pathogenesis and Management. N Am J Med Sci 7: 483-493.
- Li X, Bie Z, Zhang Z, Li Y, Hu X, et al. (2015) Clinical analysis of 64 patients with lung-cancer-associated hypercalcemia. J Cancer Res Ther 11: 275-279.
- Parker MJ, Khan AZ, Rowlands TK (2011) Survival after pathological fractures of the proximal femur. Hip Int 21: 526-530.
- Buvanendran A, Kroin JS, Berger RA, Hallab NJ, Saha C, et al. (2006) Upregulation of prostaglandin E2 and interleukins in the central nervous system and peripheral tissue during and after surgery in humans. Anesthesiology 104: 403-410.
- Papadima A, Lagoudianakis EE, Markogiannakis H, Pappas A, Georgiou L, et al. (2008) Anaesthetic considerations in parathyrotoxic crisis. Eur J Anaesthesiol 25: 772-774.
- Bushinsky DA, Monk RD (1998) Electrolyte quintet: Calcium. Lancet 352: 306-311.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...




