Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1644
Research articleOpen Access
The Study of Magnesium sulphate vs Diazepam in Eclampsia Volume 2 - Issue 1
Tariq Masood Khan* and Aisha Malik
- Department of Obstetrics and Gynecology, Pakistan
Received: November 29, 2018; Published: December 10, 2018
Corresponding author: Tariq Masood Khan, Family-Emergency Medicine Specialist, Department of Obstetrics and Gynecology, Pakistan
DOI: 10.32474/OAJRSD.2018.02.000129
Abstract
Eclampsia is a life-threatening condition with highest mortality and morbidity characterized by uncontrolled hypertension unconsciousness fits albumin urea in the setting the of Pre-Eclampsia.
Study: An extensive elaborative study carried out from 2005 to 2009, to study the therapeutic and prophylactic role of Magnesium Sulphate and Diazepam in Eclampsia.
Study Period: Four years 2005 to 2009
Objective: To study MgSo4 Vs Diazepam reducing MMR, NMR in Eclampsia
Study Design: This was a comparative study. Total 500 patients of fulminent pre-Eclampsia/Eclampsia were studied included booked/un-booked patients attending Lady Willingdon Hospital were admitted evaluated assessed and put on MgSo4 Vs Diazepam divided in two groups. The results were compared which showed of MgSo4 as superior in efficacy of compared to Diazepam in improving overall mortality morbidity in Eclampsia.
Material and Methods: Total 500 patients of Eclampsia admitted and studied in Unit-2 Lady Willingdon Teaching Hospital of King Edward Medical University Lahore managed in ICU according to a special design proforma, protocol and were given MgSo4 and Diazepam for control of fits, Hydralazine, Labetalol and Isoket infusions for lowering-blood pressure (MgSO4) A+B. (Diazepam) with multi-disciplinary involvement divided in two groups compared and followed up.
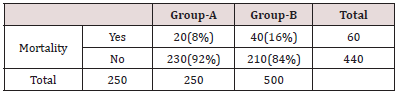
Results: In Group-A there were 20 mothers and in Group-B 40 mothers died. It was observed that maternal mortality was significantly higher in Group-B women as compared to that of Group-A. i.e. (p-value=0.005) In Group-A there were 20 mothers and in Group-B 40 mothers died. It was observed that maternal mortality was significantly higher in Group-B women as compared to that of Group-A. i.e. (p-value=0.005).
Conclusion: The comparative study and of role of MgSO4 Vs Diazepam in reducing Mortality, morbidity in maternities, neonates, efficacy showed the superiorness of Magnesium Sulphate as compared to Diazepam without any doubt.
Keywords: Therapeutic; Prophylactic role; Magnesium Sulphate; Diazepam; Eclampsia
Abrevations: MgSO4: Magnesium Sulphate; MMR: Maternal Mortality Rate; NMR: Neonatal Mortality Rate
Introduction
Eclampsia is defined as the onset of convulsions/ seizures activity during pregnancy or post-partum in a patient who have signs and symptoms of Fulminent Pre-eclampsia eclampsia i.e. hypertension & albuminuria mostly after the 20th week of Pregnancy. Eclampsia is a life-threatening emergency that continues to be a major cause of serious maternal neonatal morbidity and mortality worldwide. In developing countries eclampsia accounts for about one third of maternal deaths. Complicated cases and mismanaged cases are responsible for most maternal deaths which are usually due to cerebrovascular accidents, pulmonary edema, renal hepatic, or respiratory failure, or multi system failure, DIC, Hypertensive encephalopathy/Aspiration, Mendleson Aspiration Syndrome. Pulmonary Embolism etc.
In addition, it is associated with high perinatal mortality and neonatal morbidity. The patho physiology of eclamptic seizures is not understood. The main causes of perinatal mortality and neonatal morbidity from Eclampsia are pre-term delivery, fetal growth retardation and abruptio placentae. Fetal distress, prematurity, Asphyxia, Neonatorum. Our objective of study was to study Magnesium Sulphate Vs Diazepam in reducing mother and child mortality in Pre-Eclampsia and Eclampsia. Urgent interventions are required to manage patients/and increase public awareness, the need for case referral and to raise the level of understanding, improving attitudes and practice in relation to Eclampsia. Preeclampsia/ Eclampsia has been recognized as a clinical entity since the time of Hippocrates. In 1916 zweifel first termed “toxemia the disease of theories”.
Many of the theories attributed to pre-eclampsia/ eclampsia describe pathological features of clinical presentation which are the result rather than the cause of the disease process. It has been thought to be a neurological, renal, hepatic, hypertensive and more recently a placental disorder. It is the result of both pathological change and maternal response. In different women, the rate of progression and the organ systems affected can be different. There needs to be an initial placental trigger, but it is the maternal response that probably modifies the disease presentation and progression. By trying to understand the variations of disease presentation and progression, management of the affected women can become clearer and outcomes are predictable. One thing, that is agreed by all, is that placenta is required for the development and maintenance of pre-eclampsia/eclampsia and delivery, with removal of placenta-remains the ultimate cure and is evidenced based that the role of Magnesium Sulphate Vs Diazepam remains superior to Diazepam in both reducing maternal/neonatal morbidity/mortality. Evidence in support of the multiple modular approach to pre-eclampsia includes.
a) Poor Placentation
i. Deficient trophoblast invasion.
ii. Failure of adaptation of maternal vessels.
iii. Increased incidence of placental insufficiency.
iv. Hyperplacentosis.
v. Increased incidence in twin pregnancy.
vi. Increased incidence in diabetics.
vii. Increased incidence in rhesus incompatibility.
viii. Association with molar pregnancy.
b) Fetal/Placental Response
i. Need for placenta to be present to develop the disease.
ii. Need for placenta to be present for continuation of disease.
iii. Abnormalities of villous formation.
c) Systemic Reaction
Activation of circulating neutrophils
i. Vcam-1 is elevated in the seizures.
ii. Abnormalities of lymphocyte function.
iii. Increased cytokine activity.
iv. Increased lipid peroxide production.
d) Maternal Response
i. Decreased cellular protection for free radical activity.
ii. Generalized membrane instability.
iii. Diminished vascular endothelial function.
e) Increased vascular resistance/vasoconstriction
i. Hypertension.
ii. Renal impairment.
iii. Convulsions.
iv. Liver function.
v. Platelets consumption.
vi. Hemolysis.
Diagram of components of the pathophysiology of preeclampsia/ eclampsia that make up the multi system disorder. Preeclampsia / eclampsia only occurs in the presence of placenta. It usually but not always associated with placental insufficiency and intra uterine growth restriction IUGR. Hypertension in Pregnancy does not cause growth retardation but co-events with placental functions resulting in the restriction of growth. Both normal and abnormal implantation is likely to be influenced by maternal/ fetal immunological interactions. Large granular Macrophages and lymphocytes, as they are important to mother’s ability to recognize the invading trophoblast and respond appropriately to it. An abnormal or excessive maternal immunological response may lead to deficient implantation and poor placentation.
Maternal Immunology
Evidence for and against an immunological basis to preeclampsia.
For
a. More common in primigravida
b. More common in twin pregnancies
c. Incidence increased by change of partner.
d. A higher incidence of HLA homozygosity.
Against
a. The increase in ABO, HLA, linked compatibility
b. The incidence in monozygotic/dizygotic reproductions.
c. Similar placental findings are found in IUGR.
The “utero-placental ischemia is responsible for pre-eclampsia.” It is believed that placental ischemia results in the release of a substance “Factor X”. It is the maternal reaction to any placental factor that produces the signs & symptoms of pre-eclampsia. Immunocyte-clinical studies have localized neutrophil diastase in term placenta, decidua and myometrium in women with PET. The cell adhesion molecule V-cam is elevated in peripheral circulation. TNFX has been shown to be increased in PET.
a) Abnormalities in various cell types could explain the immunological activity red cell fragility, platelet activation, hepatocyte dysfunctions, glomerular endotheliosis and neuronal sensitivity. Therefore, all the clinical manifestations of pre-eclampsia can be explained by cellular dysfunction secondary to response to circulating factors in maternal blood.
b) Alterations in the membrane of the red cell can alter blood physiology and tendency to lyse and can explain many of the clinical finding in this condition.
c) The red cell in women with pre-eclampsia is deficient in intracellular free radical scavengers, opening them to increased damage and membrane stability. Levels of antioxidant activity correlate with plasma levels of prostacyclin and thromboxane is common with pregnancy induced hypertension. These changes particularly those found with superoxide dismutase (SOD) have been found in neutrophils. This decreased SOD activity would appear to be a secondary phenomenon.
d) The most common physical presentation of the eclampsia is hypertension. A vascular Endothelial cell dysfunction is certainly part of the disease process, though not permanent endothelial damage.
The Maternal Debus Theory
The substance responsible for the systematic reaction may be pregnancy placental villi.
Hereditary Factors
Multiple studies have suggested that pre-eclampsia is a familial disease. It seems likely that there may be genetic factors that modulate the process, the so-called MODULATOR GENES. Recent studies confirmed this family linkage and results are consistent with a single gene dominance with 48% inheritance.
a. Gene linkage with in Family groups is being investigated
b. ANGIO TENSINOGEN: a candidate gene involved in preeclampsia.
Arngrusson et al 27 in a study of several generations of women found evidence for role of AGT on neighboring gene in predisposition to pre-eclampsia. This was true for women with both Protein uric and non-pre-Protein uric and especially with Protein uric hypertension having increased significant level.
c. This suggests that genetic factor may be related to hypertension of PET and risk of convulsions is related to another, may be genetic cause.
TNF & Gene in Pre-Eclampsia
TNF can affect cells in many ways including (1) altering the balance between oxidant and antioxidant.
MGSO4 Vs Diazepam
Though MgSO4 is the drug of choice for fulminent pre- eclampsia and eclampsia, with a very narrow range of safety margins, monitoring of drug levels is mandatory, with the 3R Parameters, Renal output more than 30ml/hr. diminished/respiratory rate less than 12/min, then reduce or stop altogether Reduce or absent reflex activity 0.5gm/hr, 1-2gm/hr. is the maintenance dose MgSO4 is given as Prophylactic and therapeutic dose, Diazepam the drug, is given very slowly, as it can depress respiratory rate, MgSO4 52% better than Diazepam and 67% than phenytoin. The level of MgSO4 is in maintenance 4-7 in Eq/lit, as there is narrow therapeutic range, so MgSO4 level be monitored critically.
Review of Literature
Historical Background
Despite lack of complete understanding, preeclampsia and eclampsia have been recognised and described many centuries ago. Hippocrates observed that headaches, convulsions, fits and drowsiness are ominous signs in association with pregnancy. In his treatise on gynecology Varandeous coined the term eclampsia in 1619. Evidence exist that ancient civilizations of China, Egypt and India have all recognized and narrated this disease as well as the bleak maternal and fetal prognosis it has Association with hypertension and proteinuria, the two cardinal features were noted in mid to late 19th century (hypertension) and the early 20th century proteinuria. Delivery was further identified as the key and salient feature of management in the 20th century. Notwithstanding the extensive literature and progress in our understanding, these facts remains the basis of our management of preeclampsia and eclampsia [1,2].
Epidemiology
Eclampsia is defined as the onset of seizures/fits in the setting of preeclampsia and without another neurologic or medical causes or condition. The spectrum of hypertensive disease in pregnancy, which includes preeclampsia-eclampsia, is one of the leading causes of maternal morbidity and mortality both in the United States and worldwide, resulting in 10-15% of maternal deaths [3-5]. Eclampsia is further associated with increased morbidity due to risks of hypoxic-ischemic brain damage and intracranial hemorrhage from seizure activity [6]. Of eclampsia cases, 2-20% are complicated by perinatal loss, while 1-20% are complicated by maternal fatality, with the highest rates of morbidity and mortality in developing countries [5,7,8]. The risk of eclamptic seizures is approximately 2% in patients with untreated severe preeclampsia, while it is roughly 0.5% in patients with mild preeclampsia [9].
Eclampsia may occur antenatally, intrapartum, or postnatally. The incidence of eclampsia is cited to be on the order of 2-8 cases per 10,000 deliveries in developed countries and up to 16-69 cases per 10,000 in developing countries. Several recent studies from Canada and Ireland have demonstrated a decline in eclampsia incidence over time [7,10]. Liu et al found a decline from 12.4 per 10,000 to 5.9 from 2003 through 2009 in Canada, while O’Connor et al 4 found a decline from 5.4 per 10,000 to 3.5 over a span of 30 years in Ireland. It is believed that the use of magnesium sulfate prophylactically has been associated with a decrease in incidence of eclampsia [5,7,10,11].
Etiologic and Risk Factors for Eclampsia
Genetic predisposition, immunology, endocrinology, nutrition, abnormal trophoblastic invasion, coagulation abnormalities, vascular endothelial damage, cardiovascular maladaptation, dietary deficiencies or excess, and infection have been proposed as etiologic factors for preeclampsia/eclampsia. Imbalanced prostanoid production and increased plasma antiphospholipid have also been implicated in eclampsia [1,12,13].
Risk Factors for Eclampsia
The following are considered risk factors for eclampsia:
a) Nulliparity
b) Family history of preeclampsia, previous preeclampsia and eclampsia [13].
c) Poor outcome of previous pregnancy, including intrauterine growth retardation, abruptio placentae, or fetal death.
d) Multifetal gestations, hydatid mole, fetal hydrops, primigravida.
e) Teen pregnancy.
f) Primigravida.
g) Patient older than 35 years.
h) Lower socioeconomic status.
The following preexisting medical conditions are also considered risk factors: [1,14]
i) Obesity.
j) Chronic hypertension.
k) Renal disease.
l) Thrombophilia’s-antiphospholipid antibody syndrome.
m) Protein C deficiency and protein S deficiency.
n) Antithrombin deficiency.
o) Vascular and connective tissue disorders.
p) Gestational diabetes.
q) Systemic lupus erythematosus [14].
Pathophysiology of Eclampsia
Inhibition of Uterovascular Development
Many uterovascular changes occur when a woman is pregnant. It is believed that these changes are due to the interaction between fetal and maternal allografts and result in systemic and local vascular changes. It has been shown that in patients with eclampsia, the development of uteroplacental arteries is hindered [1].
Hindrance of Cerebral Blood Flow Regulation
It is believed that in eclampsia there is abnormal cerebral blood flow in the setting of extreme hypertension. The regulation of cerebral perfusion is inhibited, vessels become dilated with increased permeability, and cerebral edema occurs, resulting in ischemia and encephalopathy. In extreme hypertension, normal compensatory vasoconstriction may become defective. Several autopsy findings support this model and consistently reveal swelling and fibrinoid necrosis of vessel walls [1,13]
Endothelial Dysfunction
Factors associated with endothelial dysfunction have been shown to be increased in the systemic circulation of women suffering from eclampsia. These include the following [1]
i. Cellular fibronectin.
ii. Von Willebrand factor.
iii. Cell adhesion molecules (i.e., P-selectin, vascular endothelial adhesion molecule-1 [VCAM-1].
iv. Intercellular adhesion molecule-1 [ICAM-1]).
v. Cytokines (i.e., interleukin-6 [IL-6]).
vi. Tumor necrosis factor-α [TNF-α].
In addition, it is believed that antiangiogenic factors, such as placental protein fms-like tyrosine kinase 1 (sFlt-1) and activin A, antagonize vascular endothelial growth factor (VEGF). Elevated levels of these proteins cause a reduction of VEGF and induce systemic and local endothelial cell dysfunction. Leakage of proteins from the circulation and generalized edema are sequelae of the endothelial dysfunction and thus a defining factor associated with preeclampsia and eclampsia [1].
Oxidative Stress
Evidence indicates that leptin molecules increase in the circulation of women with eclampsia, inducing oxidative stress, another factor in eclampsia, on cells. (The leptin increase also results in platelet aggregation, most likely contributing to the coagulopathy associated with eclampsia.) [13,15] Oxidative stress has been found to stimulate the production and secretion of the antiangiogenic factor activin A from placental and endothelial cells [16]. Studies in pregnant mouse models have proposed that there is a dysregulation in the reactive oxygen species (ROS) signaling pathway [15-17]. Studies also suggest that increased systemic leukocyte activity plays a role in the mediation of oxidative stress, inflammation, and endothelial cell dysfunction. Histochemistry studies indicate that there is predominantly an increase in neutrophil infiltration of vasculature in patients with eclampsia [1,17].
Time of Onset of Eclampsia
The onset of eclamptic convulsions can be antepartum, intrapartum, or postpartum. The reported frequency of antepartum convulsions among recent series has ranged from 38%to53%. The frequency of postpartum eclampsia has ranged from 11% to 44%. Although most cases of postpartum eclampsia occur within the first 48h, some cases can develop beyond 48h postpartum and have been reported as late as 23 days postpartum [8,14,18- 21]. The cases of eclampsia that occur after 48h postpartum, but less than 4 weeks after delivery, are defined as late postpartum eclampsia. These women will have signs and symptoms consistent with preeclampsia in association with convulsions. Some of these women will demonstrate a clinical picture of preeclampsia during labor or immediately postpartum (56%), whereas others will demonstrate these clinical findings for the first time more than 48 h after delivery (44%) [19,21,22]. Of interest is the fact that late postpartum eclampsia developed despite the use of prophylactic magnesium during labor and for at least 24h postpartum in previously diagnosed preeclamptic women [19,21]. Therefore, women in whom convulsions develop in association with hypertension and/or proteinuria or with headaches or blurred vision after 48h of delivery should be considered to have eclampsia and initially treated as such [22].
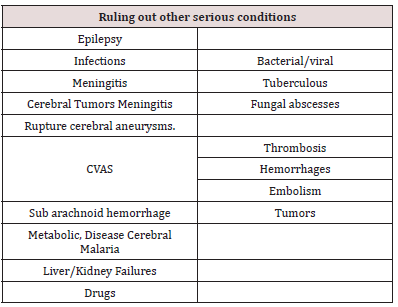
Differential Diagnosis
The presenting symptoms, clinical findings, and many of the laboratory findings overlap with several medical and surgical conditions [21,23-25]. The most common cause of convulsions developing in association with hypertension and/or proteinuria during pregnancy or immediately postpartum is eclampsia. Rarely, other etiologies producing convulsions in pregnancy or postpartum may mimic eclampsia. These diagnoses are particularly important in the presence of focal neurologic deficits, prolonged coma, or atypical eclampsia. In addition, in some patient’s gestational hypertension or preeclampsia may develop in association with these disorders (connective tissue disease, thrombophilia’s, seizure disorder, hypertensive encephalopathy), further contributing to the diagnostic difficulty [23]. Therefore, an effort should be made to identify an accurate diagnosis, given that management strategies may differ among these conditions. The diagnosis and management of the conditions that mimic eclampsia are beyond the scope of this report [26].
Differential Diagnosis of Eclampsia
Cerebrovascular Accidents
a) Hemorrhage.
b) Ruptured aneurysm or malformation.
c) Arterial embolism or thrombosis.
d) Cerebral venous thrombosis.
e) Hypoxic ischemic encephalopathy.
f) Angiomas.
I. Hypertensive encephalopathy.
II. Seizure disorder.
III. Previously undiagnosed brain tumors.
IV. Metastatic gestational trophoblastic disease.
V. Metabolic diseases.
i. Hypoglycemia, hyponatremia.
ii. Reversible posterior leukoencephalopathy syndrome.
iii. Thrombophilia.
iv. Thrombotic thrombocytopenic purpura.
v. Postdural puncture syndrome.
vi. Cerebral vasculitis [26].
Maternal–Perinatal Complications
Although eclampsia is associated with an increased risk of maternal death in developed countries (0-1.8%), the mortality rate is as high as 14% in developing countries. In developed countries, hemorrhagic stroke is the most common cause of death in patients with eclampsia and resulted in as many as 60% of all eclampsia-related deaths [8,18,19,27-30]. The high maternal mortality reported from developing countries is noted primarily among patients who had multiple seizures outside the hospital and those without prenatal care [18]. In addition, this high mortality rate could be attributed to the lack of resources and intensive care facilities needed to manage maternal complications from eclampsia [31]. Pregnancies complicated by eclampsia are also associated with increased rates of maternal morbidities, such as abruptio placentae (7-10%), disseminated intravascular coagulopathy (7-11%), pulmonary edema (3-5%), acute renal failure (5-9%), aspiration pneumonia (2-3%), and cardiopulmonary arrest (2-5%) [8,14,28].
The risks of diffuse intravascular coagulation (8%); hemolysis, elevated liver enzymes, low platelets (HELLP) syndrome (10-15%); and liver hematoma (1%) are similar in eclamptic and severely preeclamptic patients. It is important to note that maternal complications are significantly higher among women who develop antepartum eclampsia, particularly among those who develop eclampsia remote from term [8,14,28]. Perinatal mortality and morbidities remain high in eclamptic pregnancies. The reported perinatal death rate in recent series ranged from 5.6% to 11.8%.77 This high perinatal death rate is related to prematurity, abruptio placentae, and severe fetal growth restriction. 65,66 The rate of preterm delivery is approximately 50%, with approximately 25% of these occurring before 32 weeks of gestation [8,19,27,31].
Evaluation
Eclampsia always should be considered in a pregnant patient with a seizure episode. A pregnant patient who has been involved in an unexplained trauma (such as a single-vehicle auto accident) and has exhibited seizure activity should be evaluated for eclampsia. Eclampsia can occur during the antepartum, intrapartum, and postpartum periods. Ninety percent of eclampsia cases occur after 28 weeks’ gestation. Preeclampsia can quickly develop into eclampsia. The natural progression of the disease is from symptomatic severe preeclampsia (differentiated from preeclampsia by specific vital signs, symptoms, and laboratory abnormalities) to seizures [1,13,32].
a) Features of eclampsia include the following:
i. Seizure or postictal state (100%).
ii. Headache (80%), usually frontal.
iii. Generalized edema (50%).
iv. Vision disturbance (40%), such as blurred vision and photophobia.
v. Right upper quadrant abdominal pain with nausea (20%).
vi. Amnesia and other mental status changes.
b) The incidences of signs or symptoms before seizure include the following: [1,13,32]
i. Headache (83%).
ii. Hyperactive reflexes (80%).
iii. Marked proteinuria (52%).
iv. Generalized edema (49%).
v. Visual disturbances (44%).
vi. Right upper quadrant pain or epigastric pain (19%).
c) The absence of signs or symptoms before seizure include the following: [1,13,32]
i. Lack of edema (39%).
ii. Absence of proteinuria (21%).
iii. Normal reflexes (20%).
d) The relation of seizure to delivery is as follows:
i. Before delivery (>70%).
ii. Before labor (antepartum) (25%).
iii. During labor (intrapartum) (50%).
iv. After delivery (postpartum) (25%).
Although patients with severe preeclampsia are at greater risk for seizures, 25% of patients have symptoms consistent with mild preeclampsia before the seizures. A study by Cooray et al found that the most common symptoms that immediately precede eclamptic seizures are neurologic symptoms (i.e., headache, with or without visual disturbance), regardless of degree of hypertension. This suggests that closely monitoring patients with these symptoms may provide an early warning for eclampsia [33].
Physical Findings
Most patients with eclampsia present with hypertension and seizures, along with some combination of proteinuria and edema. Findings at physical examination may include the following: [1,13,32]
i. Sustained systolic BP greater than 160 mm Hg or diastolic BP greater than 110 mm Hg.
ii. Tachycardia.
iii. Tachypnea.
iv. Rales.
v. Mental status changes.
vi. Hyperreflexia.
vii. Clonus.
viii. Papilledema.
ix. Oliguria or anuria.
x. Localizing neurologic deficits.
xi. Right upper quadrant or epigastric abdominal tenderness.
xii. Generalized edema.
xiii. Small fundal height for the estimated gestational age.
xiv. Apprehension.Cervical examination of the patient with eclampsia should not be overlooked, because the delivery mode may largely depend upon the patient’s cervical status [1,13,32].
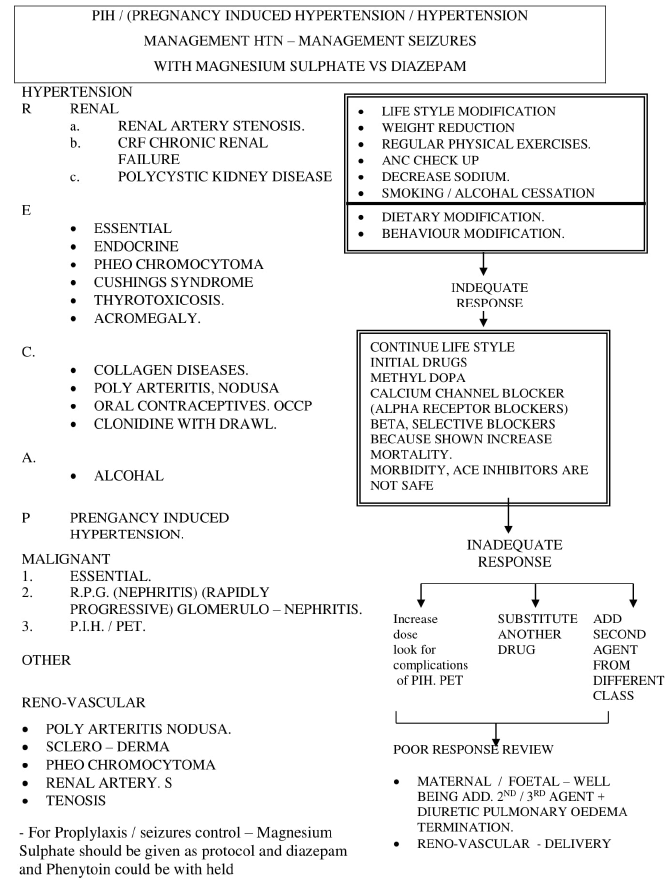
Management
The first priority in the management of eclampsia is to prevent maternal injury and to support respiratory and cardiovascular functions. During or immediately after the acute convulsive episode, supportive care should be given to prevent serious maternal injury and aspiration, assess and establish airway potency, and ensure maternal oxygenation. During this time, the bed’s side rails should be elevated and padded, a padded tongue blade should be inserted between the teeth (avoid inducing gag reflex), and physical restraints may be needed. To minimize the risk of aspiration, the patient should lie in lateral decubitus position, and vomitus and oral secretion are suctioned as needed. During the convulsive episode, hypoventilation and respiratory acidosis often occur. Although the initial seizure lasts only a few minutes, it is important to maintain oxygenation by supplemental oxygen administration via a face mask with or without oxygen reservoir at 8-10L/min [22]. After the convulsion has ceased, the patient begins to breathe again and oxygenation is rarely a problem. However, maternal hypoxemia and acidosis may develop in women who have had repetitive convulsions and in those with aspiration pneumonia, pulmonary edema, or a combination of these factors. It is recommended to use transcutaneous pulse oximetry to monitor oxygenation in all eclamptic patients [22].
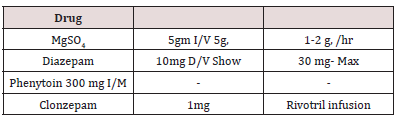
Arterial blood gas analysis is required if the pulse oximetry results are abnormal (oxygen saturation at or below 92%). The next step in the management of eclampsia is to prevent recurrent convulsions. Magnesium sulfate is the drug of choice to treat and prevent subsequent convulsions in women with eclampsia [11,34,35]. It is recommend giving a loading dose of 6 g over 15- 20min, followed by a maintenance dose of 2g/h as a continuous intravenous infusion. Approximately Seminars in perinatology 37 (2013) 207-224 214. 10% of eclamptic women will have a second convulsion after receiving magnesium sulfate [36]. In these women, another bolus of 2g magnesium sulfate can be given intravenously over 3-5min. An occasional patient will have recurrent convulsions while receiving adequate doses of magnesium sulfate. In this patient, recurrent seizures can be treated with sodium amobarbital, given 250mg intravenously over 3-5min. Serum magnesium levels are not monitored during the infusion because there is no established serum magnesium level that is considered “therapeutic.” Patients should be monitored for signs and symptoms of magnesium toxicity by serial evaluations of reflexes, respiratory rate, and urinary output. The signs and symptoms of magnesium toxicity in relationship to the magnesium level [37,38].
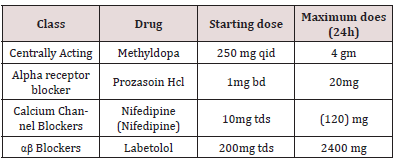
In case of magnesium toxicity, management is described. The next step in the management of eclampsia is to reduce the blood pressure, if hypertensive emergency is present. A hypertensive emergency is defined as sustained (430min), severe hypertension (≥160 mmHg systolic or ≥110 mmHg diastolic) [37-39]. The objective of treating severe hypertension is to avoid the loss of cerebral autoregulation and to prevent congestive heart failure without compromising cerebral perfusion or jeopardizing uteroplacental blood flow that is already reduced in many women with eclampsia. If not treated expeditiously, it can result in complications such as cerebral hemorrhage and maternal death. Thus, morbidity and mortality risk reduction for women with eclampsia requires management and avoidance of severe systolic and diastolic hypertension. The aim of blood pressure management is to keep the BP in a safe range but at the same time to avoid significant hypotension. We recommend a target systolic blood pressure between 140 and 160mmHg and diastolic blood pressure between 90 and 105mmHg. The rationale for keeping maternal blood pressures at these levels is to avoid potential reduction in either uteroplacental blood flow or cerebral perfusion pressure. Acute first-line therapy includes both IV labetalol and hydralazine as well as oral nifedipine [39].
Guideline recommendations for the dosing of IV labetalol are to begin with 20mg intravenously followed at 10-min intervals by doses of 40mg, then 80mg, and then 80mg for a total of 220-300mg. A fall in blood pressure within 5-10min is expected. If desired blood pressure levels are not achieved after 220mg, switch to another drug. Intravenous hydralazine is also recommended, although a meta-analysis has demonstrated a slight increase in adverse events compared with labetalol, but without enough data to recommend one drug over the other [39-41]. Hydralazine dosing begins with 5mg intravenously followed at 20min intervals by a 5-10mg bolus depending upon the initial response. A drop-in blood pressure within 10-30min is expected. If there is no success with a total of 25mg intravenously, consider using another antihypertensive medication. A recent, double-blind, randomized controlled trial demonstrated similar effectiveness with oral nifedipine and intravenous labetalol regimens in the acute control of severe hypertension [42].
Oral rapid acting nifedipine dosing begins with 10-20mg orally and continued every 30min for a maximum dose of 50mg in 1h. Other potent antihypertensive medications such as sodium nitroprusside or nitroglycerine are rarely needed in eclampsia. Diuretics are not used except in the presence of pulmonary edema. The introduction of guidelines in both the United States and the United Kingdom for the management of patients with eclampsia and increased awareness of the importance of blood pressure reduction has been associated with a decrease in the incidence of adverse maternal outcomes [43-46]. The next step in the management of a patient with eclampsia is to manage associated complications, such as disseminated intravascular coagulopathy (DIC) and pulmonary edema. If DIC is suspected, it is important to maintain blood volume and blood pressure with aggressive replacement with crystalloids and/or blood products as indicated. In general, the treatment of DIC is mainly supportive in nature. Specific details regarding the management of DIC are beyond the scope of this discussion. Initial management of pulmonary edema includes administration of oxygen and stimulating greater urine output by giving 20-40mg of IV furosemide over 1-2min. Further details regarding management of pulmonary edema is beyond the scope of this discussion. The next step in the management of an eclamptic patient is to begin induction/delivery within 24h of the onset of eclampsia. Maternal hypoxemia and hypercarbia cause fetal heart rate and uterine activity changes during and immediately following a convulsion. Fetal heart rate changes can include bradycardia, transient late decelerations, decreased beatto- beat variability, and compensatory tachycardia. Changes in uterine activity can include increased frequency and tone [47]. These changes usually resolve spontaneously within 3-10min after the termination of convulsions and the correction of maternal hypoxemia. The patient should not be rushed for an emergency cesarean delivery based on these findings, especially if the maternal condition is not stable. It is considered to be advantageous to the fetus to allow in utero recovery from hypoxia and hypercarbia due to maternal convulsions. However, if the bradycardia and/ or recurrent late decelerations persist beyond 10-15min despite all resuscitive efforts, then a diagnosis of abruptio placentae or non-reassuring fetal status should be considered. The presence of eclampsia is not an indication for cesarean delivery. The decision to perform cesarean delivery should be based on fetal gestational age, fetal condition, presence of labor, and cervical Bishop score. We recommend cesarean delivery for those with eclampsia before 30 weeks of gestation who are not in labor and whose Bishop score is below 5 [22].
Patients having labor or rupture of membranes are allowed to deliver vaginally in the absence of obstetric complications. When labor is indicated, it is initiated with either oxytocin infusions or prostaglandins in all patients with a gestational age of 30 weeks or more, irrespective of the Bishop score. A similar approach is used for those before 30 weeks of gestation if the cervical Bishop score is at least 5. Maternal pain relief during labor and delivery can be provided by either systemic opioids or epidural anesthesia as recommended for women with severe preeclampsia. Either epidural, spinal, or combined techniques of regional anesthesia can be used for cesarean delivery. Regional anesthesia is contraindicated in the presence of coagulopathy or severe thrombocytopenia (platelet count less than 50,000/mm3) [22]. In women with eclampsia, general anesthesia increases the risk of aspiration and failed intubation due to airway edema and is associated with marked increases in systemic and cerebral pressures during intubation and extubation. Women with airway or laryngeal edema may require awake intubation under fiber optic observation with the availability of immediate tracheostomy. Changes in systemic or cerebral pressures may be attenuated by pretreatment with labetalol or nitroglycerine injections [22].
The Use of Magnesium Sulphate for the Treatment of Eclampsia
The Magpie Trial
Magnesium sulphate (MgSO4) was first introduced to control convulsions in 1925, but it was the Collaborative Eclampsia Trial in 1995 that confirmed the efficacy of MgSO4 in the treatment of severe preeclampsia and eclampsia. The trial (also called Magpie trial) was a randomized, placebo-controlled study that enrolled over 10,000 women in 33 countries and across a wide variety of clinical settings. Four centers in Nigeria Ibadan, Sagamu, Port Harcourt and Sokoto participated in the study [48]. Women treated with MgSO4 had a 52% and 67% lower recurrence of convulsions than those treated with diazepam and phenytoin, respectively. Use of MgSO4 in patients with severe pre-eclampsia reduced the risk of progression to eclampsia by more than half and reduced maternal mortality. The effect of MgSO4 on perinatal outcomes was also studied, demonstrating significantly improved outcomes for newborns compared to phenytoin. Recently, the 2-year outcome following the use of MgSO4 in the Magpie trial was published. The reduction in the risk of eclampsia following prophylaxis with MgSO4 was not associated with an excess of death or disability for the women after 2 years in the group that had MgSO4 compared to placebo. The children whose mothers were treated with MgSO4 were also studied at the age of 18 months. The use of the MgSO4 was not associated with a difference in the risk of death or disability for the children at 18 months of age compared to those whose mothers were treated with placebo [29,34,49].
Mechanism of Action
The mechanism of action of MgSO4 is not completely understood. It is thought to cause dilatation of cerebral blood vessels thus reducing cerebral ischemia. It is also thought that the magnesium blocks calcium receptors by inhibiting N-methyl- Daspartate receptors in the brain [50]. Magnesium also produces a peripheral (predominantly arteriolar) vasodilatation [51] thus reducing the blood pressure. It also acts competitively in blocking the entry of calcium into synaptic endings thus altering neuromuscular transmission. This transmission is affected by a preponderant presynaptic as well as a post-synaptic effect. The presynaptic release of acetylcholine is also reduced thus altering neuromuscular transmission [52]. The precise mechanism of action for the tocolytic effects of MgSO4 is not clearly defined but may be related to the action of magnesium as a calcium blocker thus inhibiting muscle contractions [53].
Availability OF MgSO4
On the basis of the available evidence, The World Health Organization (WHO) has recommended MgSO4 as the most effective, safe, and low-cost drug for the treatment of severe preeclampsia and eclampsia. There are indeed several reports of its successful introduction in several countries including Nigeria and its effectiveness and safety for mother and baby [54-57]. However, the drug has remained largely unavailable in several developing countries where it is incidentally needed the most. Leading advocates, researchers, non-governmental organizations, representatives of the WHO and national health ministries from all over the world recently met and identified the main barriers to the use and availability of MgSO4. These included the lack of guidelines on its use, non-inclusion in many national essential drug lists, the wrong perception that the drug is meant for use only at the highest level of facilities (such as those with intensive care facilities), lack of training of health workers on its use, little incentive for pharmaceutical companies to commercialize the drug, and ready availability of prepackaged forms of less effective drugs [58].
MgSO4 Regimens
There are principally two main regimens available for the administration of MgSO4. In the Pritchard Regimen, the loading bolus dose of 4g of MgSO4 is given slowly intravenously over 5-10min and this is followed by 10g given intramuscularly (5g in each buttock). Subsequently, 5g is given intramuscularly into alternate buttocks every 4h. In the Zuspan regimen, the loading dose consists of an initial intravenous dose of 4g slowly over 5-10min followed by a maintenance dose of 1-2g every hour given by an infusion pump [59]. A gravity fed infusion set can be used in the absence of the pump especially in the developing countries. It should be noted that for the 50% MgSO4, 1 ml of the solution contains 0.5 g of MgSO4, while for the 20% solution, 1ml contains 0.2 g of MgSO4. Monitoring is important to ensure that the right doses are administered, and this is not an easy task. Whatever regimen chosen, the drug should be administered till 24h after delivery or after the last fit (whichever comes last). The choice of which regimen to use depends on a number of factors such as availability of staff to monitor the drug as well as the expertise of the staff. In resource-constrained settings, the Pritchard regimen may be easier to administer since it is given intramuscularly (could thus be administered by lower cadre of health workers).
It, however, has the disadvantage of being very painful, a situation which is not desired for a patient on whom efforts are been made to lower the blood pressure. To counteract this, the intramuscular dose could be administered with about 2ml of 1% xylocaine in the same syringe. Some workers have reported modifications in the above-mentioned regimens. MgSO4 has been used with the dose reduced to a loading dose of 4.5g intravenously and maintained on intramuscular 1.5g every 4h until 12h after delivery or the last fit [60]. In another study, the loading dose was 10g intramuscularly followed by a maintenance dose of 2.5g intramuscularly every 4h for 24h. The drug has been used as in Pritchard regime, but the duration of its administration reduced to 12h after the initial loading dose. The feto maternal outcome was similar to the two more famous regimens (Pritchard and Zuspan) [61,62].
Clinical Detection of Toxicity
The main fear of toxicity was also laid to rest with the Magpie trial. Toxicity of the drug was monitored using clinical parameters. The parameters that need to be monitored are the knee jerk (should be present), respiratory rate (should be more than 16/minute), and urine output (should be more than 25ml/min). These clinical parameters have been compared with serum levels of MgSO4. The first warning sign of toxicity is loss of the knee jerk which occurs at serum magnesium level of 3.5-5mmol/l. Respiratory paralysis occurs at 5-6.5 mmol/, cardiac conduction is altered at more than 7.5mmol/ while cardiac arrest occurs when serum magnesium exceeds 12.5mmol/l However, with the above-mentioned protocols, the expected serum range of magnesium is 2-3.5 mmol/ l10. Using the Pritchard regimen, a mean serum magnesium level of 2.1mmol/l was found. Should toxicity be detected, however, the antidote is 1g of 10% calcium gluconate given intravenously slowly over 10minutes [48,63,64].
Training on MgSO4
The need has now emerged for refresher trainings for health workers in the use of MgSO4. Clinical protocols are particularly useful in guiding such workers. The Federal Ministry of Health has developed a national clinical service protocol for obstetric care. The protocol outlines the management of eclampsia and how MgSO4 can be used and monitored. There is need to distribute this protocol and train health workers all over the country on its use. It is also recommended that the protocol should be utilized nationally as a guideline thus ensuring universal dosage regimen that will also help in uniform studies and research. Some workers have also reported the utilization of the protocol to suite the working environment in respect of the available facilities, staff, investigations and even the regimen of MgSO4 used. In Kano state, for example, the protocol was institutionalized under the guidance of the state safe motherhood committee to incorporate the role played by nonphysicians in the care of patients with eclampsia including referral where necessary [65].
Objective
I. To study MgSO4 versus Diazepam/in reducing maternal neonatal mortality in Eclampsia.
II. To screen, evaluate/manage high risk cases of eclampsia according to a set proforma/Protocol by using MgSO4 Vs Diazepam.
III. To study maternofetal outcome by using MgSO4 Vs Diazepam.
Material & Methods
Study Period
Three years from January 2005 to December 2009 in Unit -II 2 LWH, KEMU.
Study Design
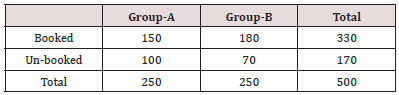
500 patients of Eclampsia were studied included booked/unbooked patients attending Lady Willingdon Hospital were admitted evaluated and put on MgSo4 Vs Diazepam and divided in two groups. The results were compared which showed a MgSo4 superior as compared to Diazepam in improving overall mortality morbidity in Eclampsia. Our study included 500 cases eclampsia with of convulsive disorders with diagnosis of pregnancy or at any time during first 6 weeks post-Partum admitted during year Jan 2005 to Dec 2009 at Lady Willingdon Hospital only, those patients were excluded which turned out to be cerebral thrombosis “Epileptic on medical record. Lady Willingdon Hospital is a tertiary care referral hospital affiliated with KEMU. Journal of obstetrics & Gynecology Deployment of MRI & TCD for Eclampsia Pt should enable a precise diagnosis and exclude many other sinister conditions and allow more precise monitoring. Maternal mortality rate MMR <1% to 20% 130 to 300/1000. Other Diagnostic Technique is improving rapidly clinical skills to interpret results, Doctor from different specialties working together (Table 1).
Procedure of Data Collection
All the patients of pregnancy between 28 weeks to 39 weeks with Eclampsia were admitted in ICU and managed according to set protocol and were given MgSO4 and Diazepam divided in two groups and their role compared. Blood Pressure was controlled with Hydralazine, Labetalol, Isoket infusion. Results were given in table form.
Of the total patients 50 were excluded from the study, 10 of them could not be traced in the follow up and their outcome are not known. 20 had been induced, pre-term delivery due to medical conditions i.e. uncontrolled hypertension & albuminuria due to eclampsia.
All patents were examined and investigated thoroughly, managed and regular follow up ensured while comparison is made between the role of MgSO4 & Diazepam in patients. The study conducted in accordance with standard criteria / protocols set up and Data analyzed by recording on an especially designed proforma for evaluation and later analysis and comparison made between when the acute emergency is over, the patients are stabilized managed accordingly till convalescence and followed regularly.
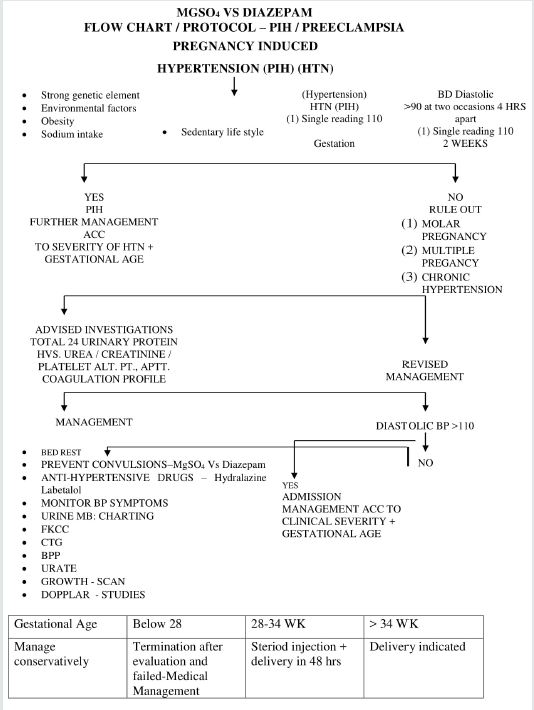
The maternal fetal outcome/prognosis depends upon the severity of signs and Gestational age. Mostly the uncomplicated pregnancy was carried to the terms also if maternal and fetal condition allows i.e. 37 completed weeks and the 34 completed weeks was taken a cutoff point to prematurity and role & comparison of use of MgSO4 Vs Diazepam in eclampsia, analyzed.
Regular antenatal care in high risk pregnancy admission of Eclampsia use of MgSO4 Vs Diazepam control of blood pressure by Hydralazine and Labetalol and urgent delivery markedly reduces MMR, NMR. All women fulfilling the study criteria were counselled especially the OPD patients and meet the average ages between the age (28-39) of gestation. Complete set of history as shown in the proforma, including precious, obstetric, Gynecological/ medical/especially previous history of hypertension/PIH, Fits unconsciousness. PET/ Eclampsia/surgical/drug and psychological history was obtained at first antenatal or first emergency admission (in case of coma from the relatives available) and entered on to a proforma. Gestational age was determined from menstrual history and confirmed by:
a) Dating / Growth, follow up scan
b) BPD (Biparietal) (FL Femur length) in the last Trimester Scan.
As part of routine / emergency care, full thorough and detailed General physical examination was conducted followed by systemic examinations of CVS, GIT, respiratory and special emphasis was laid on CNS examination and Fundo-Scopic points to be particularly important were following points.
i. Neck rigidity.
ii. Level of consciousness.
iii. Reflexes superficial, Deep tendon reflexes/Ankle Clonus Plantares Reflex.
iv. Input/Output.
v. Respiratory rate.
vi. Obstetrical complications like bleeding P/V. Pains/ Leaking.
These symptoms and signs are not only prognostic markers but also helps in monitoring of Management especially Mag sulphate and Diazepam which is the drug of choice for convulsions. The patient being managed according to a management protocol which includes [19].
I. Expectant (After ruling out fulminent Preeclampsia) Management
II. General Management Fulminent (Eclampsia management & role and usage of Magnesium Sulphate Vs Diazepam ICU.
III. Medical/ Management IV. Obstetrical
V. Follow up according to management in high dependency unit with multi-specialty involvement according to severity of symptoms and gestational age.
a. Expectant Management is continuous till maternal/fetal conditions allows and expected maternity achieved.
b. Medical includes also of Management of Fulminent cases of eclampsia.
i. HELLPS Syndrome, DIC,
ii. and Failures, cardiac/respiratory renal/hepatic, acute pulmonary edema. Thrombo-embolic-phenomenas.
Preferably in ICU HDU and equipped with Resuscitation/ monitoring equipment e.g. monitors, Resuscitators/ventilators, defibrillators/suckers etc. Strict monitoring of fluid balance is essential, as fluid over load leads to Pulmonary edema, careful Monitoring is preferred, CVP as Cardio-Vascular system is very labile gives way in seconds and can be prevented.
Data was collected from 500 consecutive eclamptic patients admitted to Lady Willingdon Hospital during the year Jan 2005 to a 2009. L.W.H. is a 235 bedded tertian care hospital attached to KEMU Lahore. It receives complicated and many serious cases coming from distant places, on admission a detailed history was taken from patient or medics attendants / relatives accompanying with any available record, then a thorough clinical and detailed general and systemic examinations and a bed side test of Proteinuria (urine R/E) dipstick method while measuring BP, until Karatokoff “V” sound heard, Neurologist/Physicians were involved in patients care where deemed necessary. During the period of study MgSO4 or/Diazepam was used as anticonvulsant and Hydralazine infusion, Nefidipine was used as antihypertensive, Data was collected on a specifically designed self-administered Performa. The cause of death was determined by a clinical history/exanimations/ investigations and previous management if any etc. Post mortem examination was not under taken in any case, statistical analysis was carried out using chi-squared test of significance.
Data Analysis
Identifying the risk factors during meticulous antenatal screening and looking for increase in hypertension, proteinuria and evaluating the warning signs of pre-eclampsia and eclampsia. The drugs are the MgSO4 and Diazepam for control of fits, control of blood pressure Hydralazine, Labetalol, Isoket infusion and study of MgSO4 and Diazepam already discussed in detail.
Results
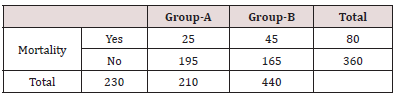
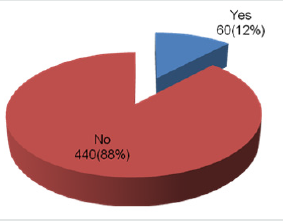
Table 2 in Group-A there were 150 women who had booked for their delivery while the remaining 100 women were un-booked for their delivery. In Group-B there were 180 women were who had booked for their delivery while 70 women were not booked for their delivery (Figure 1). Table 3 in Group-A there were 20 mothers and in Group-B 40 mothers died. It was observed that maternal mortality was significantly higher in Group-B women as compared to that of Group-A. i.e. (p-value=0.005) (Table 4). In Group-A perinatal mortality was seen in 25 neonates while in Group-B perinatal mortality was seen in 45 neonates. According to p-value it was observed that perinatal mortality was significantly lower in Group-A as compared to Group-B. i.e. (p-value=0.002).
Group-A= MgSO4
Group-B= Diazepam
Group-A= MgSO4
Group-B= Diazepam
Chi-Square Test=7.576
p-value= 0.005
Group-A= MgSO4
Group-B= Diazepam
Chi-Square Test=9.148
p-value= 0.002
Discussion
Pre-eclampsia is a condition unique to pregnancy that is characterized by new onset of hypertension and proteinuria [66]. Pre-eclampsia is a relatively common complication of pregnancy and can occur at any time during the second half of pregnancy or in the first few weeks after delivery. For many women who have pre-eclampsia the maternal outcome is good, but severe disease can lead to death or serious problems for the woman. Severe preeclampsia is associated with multiple organ system involvement including renal failure, cerebral hemorrhage and edema, hepatic failure and rupture and thrombocytopenia in addition to preeclampsia [67]. Pre-eclampsia and eclampsia are not distinct disorders but the manifestations of the same condition. In preeclampsia, hypertension and proteinuria are present, and when convulsions occur after other causes of convulsion are excluded in addition to these signs, the condition is referred to as eclampsia [66]. Eclampsia is a serious complication increasing the risk of maternal morbidity and mortality [68].
Current strategies for prevention of pre-eclampsia can be broadly classified as antenatal surveillance, modification of lifestyle, nutritional supplementation and pharmacological therapy. The only definitive treatment for pre-eclampsia or eclampsia is to end the pregnancy. The aim of interventions for women with eclampsia is to prevent further seizures, to minimize and treat any complications and, if not delivered, to optimize the timing of birth for the baby. Currently, standard care for women with severe preeclampsia/ eclampsia is to use an anticonvulsant drug to control the immediate fit, and to continue maintenance treatment to prevent further seizures [68]. The principal goals of treatment in eclampsia are stopping the convulsions and preventing further fits which are associated with reduction in adverse outcome. This has been reported to depend on the type of anticonvulsant used. It has also been revealed, in a typical systematic quantitative review and analysis, that magnesium sulphate was more effective than other interventions in preventing recurrent seizures in eclampsia and in preventing the first seizure in pre-eclampsia. Another metaanalysis by Duley et al. has reported a substantial reduction in the risk recurrence of further fits (RR=0.44, 95% CI 0.34 to 0.57) in eclamptic pregnant women [34,69].
In this study it was observed that maternal and perinatal mortality was high in Group-B (diazepam) however In Group-A (MgSO4) maternal and perinatal mortality was low. This difference in mortality in both treatment groups was statistically significant. According to Khan, magnesium sulphate was the better anticonvulsant than diazepam infusion in terms of total morbidity and maternal deaths (Null versus 5%) [70]. A Randomized Double- Blind Trial of Magnesium Sulphate and Diazepam in Lagos, Nigeria also showed that the use of magnesium sulphate was found to be significantly associated with less serious morbidity in comparison to diazepam use [71]. A similar significant reduction in maternal deaths was reported in Tanzania [72]. Gizat M. Kassie in his study evaluated maternal outcomes of magnesium sulphate and diazepam use in the management of severe pre-eclampsia and eclampsia in Jimma University Specialized Hospital. In his results he reported that Three pregnant women from the magnesium sulphate treated group and eleven pregnant women from diazepam treated group had at least one convulsion after taking the drug. Greater proportion of patients in the magnesium sulphate treated group had less than four days postpartum stay as compared to the diazepam treated patients (82.3% versus 66.2%). Seizure occurrence, duration of postpartum hospital stays and birth outcome had a statistically significant association with the type of anticonvulsant used [73].
Chaudhary P in his study determined the incidence and impact of changes in the intervention strategy for the management of eclampsia in a maternity hospital on maternal and perinatal outcome. In his results he reported that marked improvement was noticed in terms of recurrence of fit (19.13% vs 73.91%) with change in the intervention strategy. Perinatal deaths were fewer in study period B (20% vs 33%). Overall, it seems that care of eclamptic patients and use of magnesium sulphate as anticonvulsant has resulted in positive impact on maternal outcome [57]. Eclampsia is still commonly perceived as the end of a linear spectrum that stretches from normal pregnancy, through mild hypertension, preeclampsia and finally eclampsia. However, eclampsia may precede pre-eclampsia and an alternative view is that seizures are one of the range of signs and symptoms caused by the widespread endothelial cell damage secondary to an ischemic placenta. Pre-eclampsia is a multisystem disease of poorly understood etiology and the extent of involvement of various organs are unpredictable. Another important fact is that treatment of this disease is entirely empirical and symptomatic, and controversy still exists in the choice of the most appropriate treatment especially for prevention and control of fits.
It is understood that treatment of eclampsia is symptomatic as underlying cause is unknown. Most clinicians agree that diazepam should be used to abort initial fit as it is effective, inexpensive, easily available and can be used by nursing staff. However, the risks include respiratory depression, respiratory arrest and aspiration pneumonia. The general aim of treatment in eclampsia is prevention of further fits as it is the recurrent fits that leads to significant cerebral anoxia and its associated adverse outcome. The greater efficacy of magnesium sulphate compared to diazepam or phenytoin for prevention of recurrence of fits is now accepted worldwide in many studies (Figures 2-4) and (Flowcharts 1 & 2).
Conclusion
The comparative study and of role of MgSO4 Vs Diazepam in reducing Mortality, morbidity in maternities, neonates showed the efficacy and superiorness of Magnesium Sulphate as compared to Diazepam without any doubt.
Guide Lines / Key Points Symptomatology / Management Protocol Pre-Eclampsia, Eclampsia
Review MgSO4 Vs Diazepam Key Points
Eclampsia is defined as generalized seizures accompanied with fulminent Pre-eclampsia. Pre-eclampsia eclampsia developing after 20 weeks and at the end of 1st week of Postpartum treatment is I/V Mg Sulphate I/V Hydralazime / Labetalol and rapid delivery.
Key Points Score: Criteria to score Preeclampsia
I. New onset protinuric hypertension and at least one of the following:
a) Symptoms of central nervous system dysfunction:
Blurred vision, scotomata, altered mental status, severe headache.
b) Symptoms of liver capsule distention:
Right upper quadrant or epigastric pain.
c) Hepatocellular injury:
Serum transaminase concentration at least twice normal.
d) Severe blood pressure elevation:
Systolic blood pressure 160 mm Hg or diastolic 110 mg Hg. On two occasions at least six hours apart.
e) Thrombocytopenia:
Less than 100,000 platelets per cubic millimeter
f) Proteinuria:
Over 5 grams in 24 hours or 3+ or more on two random samples four hours apart.
Oliguria <500 mL in 24 hours.
Intrauterine fetal growth restriction.
Pulmonary edema or cyanosis.
Cerebrovascular accidents (CVAS).
Coagulopathy / DIC (Disseminated Intravascular Coagulation).
Review Mgso4 Vs Diazepam
Eclampsia / Pre-Eclampsia
I. Key Points Management Protocol Eclampsia
a) Immediate Care:
Maintain airway, left lateral position, oxygen administration.
b) Abort convulsions:
diazepam 10 mg. 1.v. or clonazepan 1 mg i.v.
c) Seizure prophylaxis / therapeutics:
Magnesium sulphate.
d) Maintain diastolic blood pressure of 95-105 mmHg
e) Coagulation screen/renal function / platelet count.
f) Haemodynamic stabilization followed by delivery within 6-8 h.
g) Postpartum:
24-48 h of intensive care
h) N.B. Ventilatory support for at. Least 24 hours if:
poor arterial blood gases.
Unconsciousness/Glasgow Come scale <8
Extreme restlessness
Laryngeal edema (Tables 5 & 6).
II. Key Points for Clinical Practice:
i. There may be a strong family history.
ii. Hypertension may not be the primary presenting symptom.
iii. The placenta is the initial trigger to disease development.
iv. The presentation of pre-eclampsia is very varied, any body system can be affected.
v. Maternal response is the main controller of disease severity.
vi. Upper abdominal pain is a concerning feature suggestive of HELLP syndrome.
vii. Eclampsia is not an inevitable progression from preeclampsia.
viii. Pulmonary edema can occur with a normal CVP due to capillary leak.
III. Recommendations for Management- key point for clinical practice
We make the following recommendations for the management of patients with eclampsia and severe pre-eclampsia:
a. All severe Fulminent pre-eclamptics and eclamptics should be managed in special regional centers with the appropriate expertise.
b. Continuous monitoring of blood pressure pulse rate, ECG and central venous pressure is required as the cardio-vascular system is extremely labile in this condition and can deteriorate in seconds.
c. Magnesium Sulphate is the drug of choice, in fulminent pre eclampsia / eclampsia.
d. Magnesium Sulphate both proplylactically therapeutically, evidenced based, proved beyond any doubt.
e. The airway should be maintained and protected. Any patient with a Glasgow Coma Scale of less than 9 should be intubated. Nursing staff at district hospitals and community clinics should be taught how to position an unconscious patient, insert oral airway and administer oxygen.
f. An arterial partial pressure of oxygen of at least 100 mmHg should be maintained. Mechanical ventilation may be necessary.
g. Blood pressure should be carefully and slowly lowered the diastolic pressure should be lowered by not more than 30 mmHg in order to maintain cerebral perfusion.
h. Seziures should be prevented or terminated as soon as possible MgSO4 is the anticonvulsant of choice for this purpose. Efficient transport facilities must be available and personnel at district hospitals and community clinics should be capable of administering anticonvulsants.
i. The fetus should be delivered within 6-12 hrs of admission: Caesarean section is often indicated. General anesthesia, administered by skilled anesthetist, is recommended. Where these facilities are not available, epidural anesthesia would be adequate, provided a hypotensive episode is prevented with sufficient intravenous pre-loading and coagulopathy is excluded by estimation of crude clotting time, fibrinogen levels and platelet counts.
j. An eclampsia team should be organized, since the problems developed by these patients are multifactorial. Personnel (an obstetician, obstetric anesthetist and critical care nurse) experienced in the specific obstetric management of these patients need to work together, in order to improve patient outcome.
While prevention of pre-eclampsia/eclampsia must await an understanding of its etiology, improvement in antenatal care, together with active management of the disease when it develops, will improve both fetal and maternal prognosis.
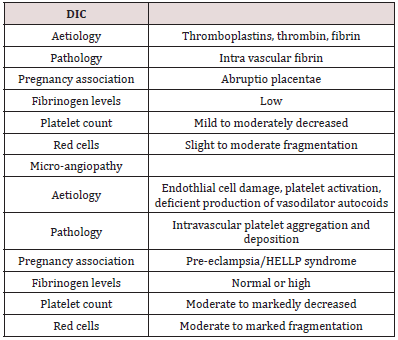
Pre-Eclampsia/Eclampsia – DIC (Table 7)
For:
a) More common in primigravida.
b) More common in twin pregnancies.
c) Incidence increased by change of partner.
d) A higher incidence of HLA homozygosity.
Against
a) No increase in ABO. HLA or Y-linked compatibility.
b) The incidence is similar in monozygotic and dizygotic twin pregnancies.
c) Similar placental findings are found in intra-uterine growth retardation.
Key Points - MgSO4 Vs Diazepam
a) Eclampsia is an obstetrical emergency that occurs in 4 to 5 per 10,000 live births in developed countries, with higher rates in under developing countries.
b) Approximately one-third of cases are not preventable.
c) Both the fetus and the mother are at immediate risk of death or life-long neurologic disability.
d) The goals of management are to stabilize the mother, prevent recurrent convulsions, treat severe hypertension, and initiate delivery of the fetus.
e) Delivery is the only curative treatment, but immediate cesarean birth is not usually necessary.
f) Maternal prognosis is good with prompt treatment and in the absence of cerebrovascular hemorrhage. The fetal prognosis is primarily dependent upon the gestational age at delivery.
g) Two percent of women will experience an eclamptic seizure in a future pregnancy, 9 to 25 percent will develop severe preeclampsia, and up to 65 percent will develop preeclampsia.
h) Magnesium sulfate is the drug of choice for prevention of both primary and recurrent eclamptic seizures as compared to diazepam Phenytoin.
References
- Ross MG (2014) Eclampsia.
- Jido T, Yakasai I (2013) Preeclampsia: a review of the evidence. Annals of African medicine 12(2): 75-85.
- Practice ACoG. (2002) Practice bulletin# 33: diagnosis and management of preeclampsia and eclampsia. Obstetrics & Gynecology 99(1): 159- 167.
- Main EK (2010) Maternal mortality: new strategies for measurement and prevention. Current opinion in Obstetrics and Gynecology 22(6): 511-516.
- O Connor HD, Hehir MP, Kent EM, Foley ME, Fitzpatrick C, et al. (2013) Eclampsia: trends in incidence and outcomes over 30 years. American journal of perinatology 30(8): 661-664.
- Richards A, Graham D, Bullock R (1988) Clinicopathological study of neurological complications due to hypertensive disorders of pregnancy. Journal of Neurology, Neurosurgery & Psychiatry 51(3): 416-421.
- Liu S, Joseph K, Liston RM, Bartholomew S, Walker M, et al. (2011) Incidence, risk factors, and associated complications of eclampsia. Obstetrics & Gynecology 118(5): 987-994.
- Douglas KA, Redman C (1994) Eclampsia in the United Kingdom. Bmj 309(6966): 1395-1400
- Sibai BM (2004) Magnesium sulfate prophylaxis in preeclampsia: lessons learned from recent trials. American journal of obstetrics and gynecology 190(6): 1520-1526.
- Tan K, Kwek K, Yeo G (2006) Epidemiology of pre-eclampsia and eclampsia at the KK Women’s and Children’s Hospital, Singapore. Singapore medical journal 47(1): 48-53.
- Altman D, Carroli G, Duley L, Farrell B, Moodley J, et al. (2002) Do women with pre-eclampsia, and their babies, benefit from magnesium sulphate? The Magpie Trial: a randomised placebo-controlled trial. The Lancet 359(9321): 1877-1890.
- Nodler J, Moolamalla SR, Ledger EM, Nuwayhid BS, Mulla ZD (2009) Elevated antiphospholipid antibody titers and adverse pregnancy outcomes: analysis of a population-based hospital dataset. BMC pregnancy and childbirth 9(1):11.
- Gabbe SG, Niebyl JR, Galan HL, Jauniaux ER, Landon MB, et al. (2012) Obstetrics: normal and problem pregnancies: Elsevier Health Sciences.
- Mattar F, Sibai BM (2000) Eclampsia VIII. Risk factors for maternal morbidity. American journal of obstetrics and gynecology 182(2): 307- 312.
- Banerjee S, Randeva H, Chambers AE (2009) Mouse models for preeclampsia: disruption of redox-regulated signaling. Reprod Biol Endocrinol 7(4).
- Reddy A, Suri S, Sargent IL, Redman CW, Muttukrishna S (2009) Maternal circulating levels of activin A, inhibin A, sFlt-1 and endoglin at parturition in normal pregnancy and pre-eclampsia. PloS one 4(2): 4453.
- Cadden KA, Walsh SW (2008) Neutrophils, but not lymphocytes or monocytes, infiltrate maternal systemic vasculature in women with preeclampsia. Hypertension in Pregnancy 27(4): 396-405.
- Katz VL, Farmer R, Kuller JA (2000) Preeclampsia into eclampsia: toward a new paradigm. American journal of obstetrics and gynecology 182(6): 1389-1396.
- Chames MC, Livingston JC, Ivester TS, Barton JR, Sibai BM (2002) Late postpartum eclampsia: a preventable disease? American journal of obstetrics and gynecology 186(6): 1174-1177.
- Thornton C, Dahlen H, Korda A, Hennessy A (2013) The incidence of preeclampsia and eclampsia and associated maternal mortality in Australia from population-linked datasets: 2000-2008. American journal of obstetrics and gynecology 208(6): 476.
- Lubarsky SL, Barton JR, Friedman SA, Nasreddine S, Ramadan MK, et al. (1994) Late postpartum eclampsia revisited. Obstetrics & Gynecology 83(4): 502-505.
- Hart LA, Sibai BM, editors. (2013) Seizures in pregnancy: epilepsy, eclampsia, and stroke. Seminars in perinatology: Elsevier 37(4).
- Witlin AG, Friedman SA, Egerman RS, Frangieh AY, Sibai BM (1997) Cerebrovascular disorders complicating pregnancy-beyond eclampsia. American journal of obstetrics and gynecology 176(6): 1139-1148.
- Hinchey J, Chaves C, Appignani B, Breen J, Pao L, et al. (1996) A reversible posterior leukoencephalopathy syndrome. New England Journal of Medicine 334(8): 494-500.
- Stott V, Hurrell M, Anderson T (2005) Reversible posterior leukoencephalopathy syndrome: a misnomer reviewed. Internal medicine journal 35(2): 83-90.
- Sibai BM (2005) Diagnosis, prevention, and management of eclampsia. Obstetrics & Gynecology 105(2): 402-410.
- Leitch C, Cameron A, Walker J (1997) The changing pattern of eclampsia over a 60‐year period. BJOG: An International Journal of Obstetrics & Gynaecology 104(8): 917-922.
- López-Llera M (1992) Main clinical types and subtypes of eclampsia. American journal of obstetrics and gynecology 166(1): 4-9.
- Group TETC (1995) Which anticonvulsant for women with eclampsia? Evidence from the Collaborative Eclampsia Trial. The Lancet 345(8963): 1455-1463.
- Martin Jr JN, Thigpen BD, Moore RC, Rose CH, Cushman J, et al. (2005) Stroke and severe preeclampsia and eclampsia: a paradigm shift focusing on systolic blood pressure. Obstetrics & Gynecology 105(2): 246-254.
- Sibai BM (1990) Eclampsia: VI. Maternal-perinatal outcome in 254 consecutive cases. Am J Obstet Gynecol 163(3): 1049-1054.
- Stead LG (2011) Seizures in pregnancy/eclampsia. Emerg Med Clin North Am 29(1): 109-116.
- Cooray SD, Edmonds SM, Tong S, Samarasekera SP, Whitehead CL (2011) Characterization of symptoms immediately preceding eclampsia. Obstet Gynecol 118(5): 995-999.
- Duley L, Gülmezoglu A, Henderson-Smart D (2007) Magnesium sulphate and other anticonvulsants for women with pre-eclampsia. Cochrane Database Syst Rev 10(11).
- Lucas MJ, Leveno KJ, Cunningham FG (1995) A comparison of magnesium sulfate with phenytoin for the prevention of eclampsia. N Engl J Med 333(4): 201-205.
- Witlin A, Sibai B (1998) Magnesium sulfate therapy in preeclampsia and eclampsia. Obstet Gynecol 92(5): 883-889.
- Ono Y (2008) Management of eclampsia.
- Yaqub S, Zafar B, Mahmood KT, Amin F (2011) Management of Eclampsia. Journal of Pharmaceuticals Science and Technology 3: 528-535.
- American College of Obstetricians and Gynecologists (2011) ACOG Practice Bulletin No. 125: Chronic hypertension in pregnancy. Obstet Gynecol 119(2 Pt 1): 396-407.
- Committee on Obstetric Practice (2011) Committee Opinion no. 514: emergent therapy for acute-onset, severe hypertension with preeclampsia or eclampsia. Obstet Gynecol 118(6): 1465-1468.
- Magee LA, Cham C, Waterman EJ, Ohlsson A, Von Dadelszen P (2003) Hydralazine for treatment of severe hypertension in pregnancy: Metaanalysis. BMJ 327(7421): 955-960.
- Shekhar S1, Sharma C, Thakur S, Verma S (2013) Oral nifedipine versus intravenous labetalol for acute blood pressure control in hypertensive emergencies of pregnancy: a randomised trial. Obstet Gynecol 122(5): 1057-1063.
- Menzies J, Magee LA, Li J, MacNab YC, Yin R, et al. (2007) Instituting surveillance guidelines and adverse outcomes in preeclampsia. Obstet Gynecol 110(1): 121-127.
- Von Dadelszen P, Sawchuck D, McMaster R, Douglas MJ, Lee SK, et al. (2010) The active implementation of pregnancy hypertension guidelines in British Columbia. Obstet Gynecol 116(3): 659-666.
- Cantwell R, Clutton-Brock T, Cooper G, Dawson A, Drife J, et al. (2011) Saving mothers’ lives. Reviewing maternal deaths to make motherhood safer: 2006-2008. BJOG 118(1): 1-203.
- McClure J, Cooper G, Clutton-Brock T (2011) Saving Mothers’ Lives: reviewing maternal deaths to make motherhood safer: 2006-2008: a review. British journal of anaesthesia 107(2): 127-32.
- Paul RH, Koh KS, Bernstein SG (1978) Changes in fetal heart rate-uterine contraction patterns associated with eclampsia. Am J Obstet Gynecol 130(2): 165-169.
- Ekele B (2006) Magnesium sulphate: The gold standard for the treatment of eclampsia and severe pre-eclampsia. Trop J Obstet Gynaecol 23(1): 1-2.
- Ekele B (2006) Magnesium sulphate: The gold standard for the treatment of eclampsia and severe pre-eclampsia. Trop J Obstet Gynaecol 23(1): 1-2.
- Sadeh M (1989) Action of magnesium sulfate in the treatment of preeclampsia-eclampsia. Stroke 20(9): 1273-1275.
- Vigorito C, Giordano A, Ferraro P, Acanfora D, De Caprio L, et al. (1991) Hemodynamic effects of magnesium sulfate on the normal human heart. Am J cardiol 67(16): 1435-1437.
- Krendel DA (1990) Hypermagnesemia and neuromuscular transmission. Semin Neurol 10(1): 42-45.
- Dube L, Granry JC (2003) The therapeutic use of magnesium in anesthesiology, intensive care and emergency medicine: a review. Can J Anaesth 50(7): 732-746.
- Omu A, Al-Harmi J, Vedi H, Mlechkova L, Sayed A, et al. (2008) Magnesium sulphate therapy in women with pre-eclampsia and eclampsia in Kuwait. Med Princ Pract 17(3): 227-232.
- Adewole I, Oladokun A, Okewole A, Omigbodun A, Afolabi A, et al. (1999) Magnesium sulphate for treatment of eclampsia: the Nigerian experience. Afr J Med Sci 29(3-4): 239-241.
- Shamsuddin L, Nahar K, Nasrin B, Nahar S, Tamanna S (2005) Use of Parenteral Magnesium Sulphate in Eclampsia and Severe Pre-eclampsia Cases in a Rural set up of Bangladesh. Bangladesh Med Res Counc Bull 31(2): 75-82.
- Chaudhary P (2005) Eclampsia: before and after magnesium sulphate. Journal of Nepal Medical Association. 44(160).
- Langer A, Villar J, Tell K, Kim T, Kennedy S (2008) Reducing eclampsiarelated deaths-a call to action. Lancet 371(9614): 705-706.
- Zuspan FP (1978) Problems encountered in the treatment of pregnancyinduced hypertension. A point of view. Am J Obstet Gynecol 131(6): 591- 597.
- Abdul MA IU, Tukur J, Yusuf MD (2013) Low dose Magnesium sulphate in the management of eclamptic fits: A Randomised controlled trial. Arch Gynecol Obstet 287(1): 43-46.
- Begum R, Begum A, Johanson R, Ali MN, Akhter S (2001) A low dose (‘Dhaka’) magnesium sulphate regime for eclampsia. Acta Obstet Gynecol Scand 80(11): 998-1002.
- Ekele B, Ahmed Y (2004) Magnesium sulfate regimens for eclampsia. International Journal of Gynecology & Obstetrics 87(2): 149-150.
- Lu JF, Nightingale CH (2000) Magnesium sulfate in eclampsia and preeclampsia. Clin pharmacokinet 38(4): 305-314.
- Ekele B, Badung S (2005) Is serum magnesium estimate necessary in patients with eclampsia on magnesium sulphate? Afr J Reprod Health 9(1): 128-132.
- Tukur J (2009) The use of magnesium sulphate for the treatment of severe pre-eclampsia and eclampsia. Ann Afr Med 8(2): 76-80.
- Mizutani S, Wright J, Kobayashi H (2011) A new approach regarding the treatment of preeclampsia and preterm labor. Life sci 88(1): 17-23.
- Mundle S, Regi A, Easterling T, Biswas B, Bracken H, et al. (2012) Treatment approaches for preeclampsia in low-resource settings: A randomized trial of the Springfusor pump for delivery of magnesium sulfate. Pregnancy Hypertens 2(1): 32-38.
- Vigil-De Gracia P (2009) Maternal deaths due to eclampsia and HELLP syndrome. Int J Gynaecol Obstet 104(2): 90-94.
- Tannirandorn Y (2005) Is magnesium sulfate for prevention or only therapeutic in preeclampsia? J Med Assoc Thai 88(7): 1003-1010.
- Khan I (2010) Magnesium Sulfate Versus Diazepam Infusion in Eclampsia. Annals of King Edward Medical University 15(3).
- Ola RE, Odeneye O, Abudu OO (2004) Eclampsia: A Randomized Double- Blind Trial of Magnesium Sulphate and Diazepam in Lagos, Nigeria. Trop J Obstet Gynaecol 21(2): 143-147.
- Muganyizi PS, Shagdara MS (2011) Predictors of extra care among magnesium sulphate treated eclamptic patients at Muhimbili National Hospital, Tanzania. BMC Pregnancy Childbirth 11(1): 41.
- Kassie GM, Negussie D, Ahmed JH (2014) Maternal outcomes of magnesium sulphate and diazepam use in women with severe preeclampsia and eclampsia in Ethiopia. Pharma pract 12(2): 400.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...