Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4722
Research Article(ISSN: 2637-4722) 
Late Intrahospital Bath Is Safety in Newborns Volume 3 - Issue 1
Erika del Carmen Ochoa Correa MD, PhD1, Mónica Janeth Olivares-Ramírez MD1, Isaías Rodríguez Balderrama MD, PhD1, Adriana Nieto Sanjuanero MD, PhD1*, Jorge M. Llaca Diaz MSc2, Christian Alanis Enríquez MD1, Manuel Enrique de la O-Cavazos3
- 1Neonatology Unit, School of Medicine and University Hospital “Dr. José Eleuterio Gonzalez”, Universidad Autónoma de Nuevo León (U.A.N.L.), Monterrey, Nuevo León, México.
- 2Departamento de Patología Clínica, School of Medicine and University Hospital “Dr. José Eleuterio Gonzalez”, Universidad Autónoma de Nuevo León (U.A.N.L.), Monterrey, Nuevo León, México.
- 3Department of Pediatrics, School of Medicine and University Hospital “Dr. José Eleuterio Gonzalez”, Universidad Autónoma de Nuevo León (U.A.N.L.), Monterrey, Nuevo León, México.
Received: December 09, 2020 Published: January 12, 2021
Corresponding author: Adriana Nieto Sanjuanero MD, PhD, Neonatology Unit, School of Medicine and University Hospital “Dr. José Eleuterio Gonzalez”, Universidad Autónoma de Nuevo León (U.A.N.L.), Av. Francisco I. Madero, s/n, Col. Mitras Centro, C.P. 66460, Monterrey, Nuevo Léon, Mexico.
DOI: 10.32474/PAPN.2021.03.000155
Abstract
Introduction: Preterm infants in many neonatal intensive care units (NICU) are routinely bathed despite the lack of evidence to support frequent bathing as a way to reduce the risk of infection. In fact, research has shown that routine bathing causes behavioral and psychological instability in premature infants
Materials and methods: Prospective, randomized clinical trial. Neonates older than 34 weeks of gestation were included, treated in the Neonatal Intermediate Care Unit at the University Hospital in the period from October 1, 2018 to February 28, 2019.
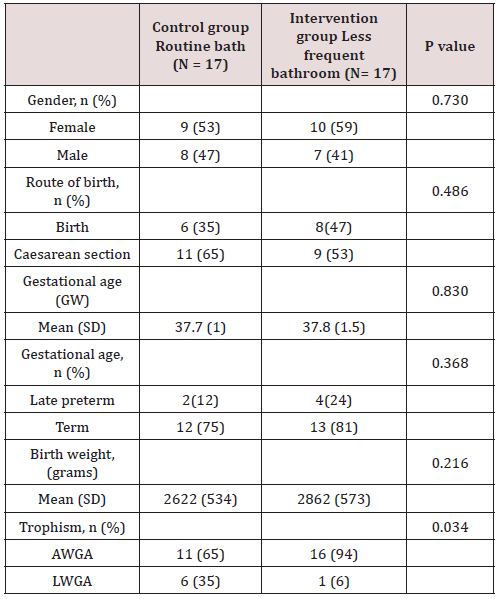
Results: A total of 34 patients were included in the study, which were assigned by lot to two groups, 17 patients corresponded to the control group (daily bath) and 17 patients to the intervention group (bath every 3 days). There was no significant difference between the two groups regarding gestational age, however, a value of p = 0.034 was found regarding trophism.
Conclusion: There were no differences in the presence of saprophytic flora or pathogenic flora that colonize the skin of the neck region between both groups. The main saprophytic microorganism found was coagulase negative Staphylococci; and the main pathogens detected in the cultives of both groups were Staphylococci Aureus and E. Coli.
Keywords: Bath; Colonization; Cultivation; Germ; Newborn; Pathogenic Flora; Saprophytic Flora
Introduction
Globally 3.1 million children die each year in the neonatal period (the first 28 days of life). Half of these deaths occur within the first 24 hours of birth, 75% occur in the early neonatal period (0 to 6 days) due to preterm births, severe infections, and perinatal asphyxia. Essential care can contribute to reducing the incidence of neonatal morbidity and mortality [1]. Recommendations for care in the first week of life include improvement in thermal care practices such as drying, skin-to-skin contact, immediate breastfeeding, and delayed bathing [2]. Although some aspects of care are understood, others have not been adequately investigated or explained, the infant bath is an example. Preterm infants in many neonatal intensive care units (NICU) are routinely bathed despite the lack of evidence to support frequent bathing as a way to reduce the risk of infection. In fact, research has shown that routine bathing causes behavioral and psychological instability in premature infants [3]. The skin functions as a protective barrier against pathogens and toxins, provides thermal regulation, and maintains fluid balance [4]. The skin of the term newborn has a barrier function similar to that of the adult, whereas the skin of the premature newborn provides a poor barrier [3]. Among the clinical implications are greater susceptibility to microbial attack, greater risk of maceration due to moisture retention and greater response to irritants, in addition to the fact that soaps and cleaners can alter the epidermal barrier and the acid mantle causing dry skin and more susceptible to damage [5].
The greater colonization of newborns occurs at birth, the new evidence suggests that factors in labor and the environment at birth such as the route of birth, the frequency of vaginal examination, the use of antibiotics, influence the maternal microbiome and the newborn [6,7]. Newborn skin contains a variety of microorganisms and is determined to some extent by the type of birth, vaginal or cesarean. The skin of newborns obtained vaginally contains bacterial “communities” that resemble the maternal vaginal flora, whereas the skin of newborns by caesarean section reflects the flora of the skin [3]. Vaginal birth remains the most common method of birth and is achieved in 68% of women in the United States. The vaginal microbiome is a mutualistic environment of organisms that depend on environmental conditions and host factors that promote homeostasis in the vagina and inhibit the growth of bacteria that have been associated with preterm labor and chorioamnionitis. When born vaginally, the fetus is exposed to a large number of maternal bacteria from the vagina and the perianal area. Infants delivered via caesarean section, regardless of exposure to prophylactic antibiotics, harbor a less diverse microbiome that resembles the skin, mouth, and bacteria on surrounding maternal surfaces [8]. Infants born via cesarean section tend to harbor a large portion of antibiotic resistant genes compared to those born vaginally [9]. Sponge bathing and bathing in premature newborns can negatively impact some physiological parameters such as heart rate, oxygenation, and present behavioral signals that indicate stress. Due to concerns about stress in premature babies during bathing, studies evaluated the impact of bathing with a mild cleanser every 2 days versus every 4 days on colonization of skin and pathogenic bacteria. Concern for thermoregulation of newborns has traditionally driven bathing practices, as effective self-regulation of body temperature can be a major challenge for newborns [10]. Temperature regulation is of vital importance for nurses caring for newborns. Although there are many benefits of bathing, such as maintaining the integrity of the skin, reducing the risk of infection and elevation of temperature, promoting mobility and maintaining joint function, the act itself has become a ritual and is based on tradition. rather than empirical evidence [11]. The times for bathing are flexible, depending on the characteristics and stability of the newborn and the wishes of your family. However, the best time for the bath to occur is unknown. The newborn’s bath should not last more than 5 minutes [5]. There are 3 types of bath available for infants. Sponge bathing is the act of bathing with a cloth or sponge without being immersed in a bath of water. The bathtub bath is the act of bathing the baby immersed in a bathtub with water. Finally, the wrapped bath is the immersion of the infant, who is first wrapped in a towel, in water, washing 1 limb at a time while maintaining a flexed and contained position throughout the bath. The objective of this study was to correlate the bacterial colonization of the neck region with the presence of sepsis, signs of systemic inflammatory response, or local infections, in neonates with an indication of daily bath versus bath every 3 days. The hypothesis raised was that the second bath of the newborn after 3 days of hospital stay does not increase the incidence of skin or systemic infections.
Materials and Methods
Prospective, randomized clinical trial. Neonates older than 34 weeks of gestation were included, treated in the Neonatal Intermediate Care Unit at the University Hospital in the period from October 1, 2018 to February 28, 2019. Within the selection criteria, the inclusion criteria were: Neonates older than 34 weeks of gestation, admitted to the Neonatal Intermediate Care Unit, euthermic and that the first bath was performed in the first 24 hours of hospital stay. The exclusion criteria were: Newborns admitted to hospital with an infectious history during their pregnancy, patients with the presence of an infectious process at the time of admission and / or administration of antibiotics. The elimination criteria were: Neonates who are discharged within 3 days of hospital stay, who do not perform any of the cultives and / or who present an incomplete clinical record. Informed consent was obtained, which was signed by both parents and two witnesses. The neonates were divided into 2 groups by means of a lottery, in both groups a basal culture was taken in the neck region at the time of admission, then the first bath was performed, and a second culture was taken after 30 minutes. The control group bathed every 24 hours and the intervention group every 3 days, using the sponge bath technique. In all patients, a third culture was collected at 3 days, before bathing and a fourth culture 30 minutes after bathing. The skin cultives were processed in the Microbiology Laboratory of the “Dr. José Eleuterio González”. To obtain the skin cultives, a Stuart culture medium was used, the sample was obtained from the neck region, cleaning 10 times, in 2 directions, 5 horizontal and 5 vertical, in 2cm [2]. The type of skin flora was identified and the number of Colony Forming Unit (CFU) per square centimeter was obtained for each microorganism. It will be determined after 48hr of incubation at 35°C. The sample was processed in the Microbiology Laboratory of the “Dr. José Eleuterio González” within 30 minutes, after obtaining each sample[12].
The normal flora was defined as coagulase-negative staphylococcus and diphtheroids [3], the pathogenic flora as any microorganism other than those mentioned [13]. When a pathogen was identified, an antibiogram was performed, the treating physician was informed, and the result was documented in the clinical record of each patient. The bathing process of the newborns included in the study consists of a sponge bath, a new Grisi® neutral soap was used for each bath and tap water. The bath was carried out in the morning shift, by nursing personnel. After completing the bath, the infants were dressed in diapers and a clean sheet. Descriptive statistics, simple frequencies and percentages were used for the analysis of the studied variables. They were used for data collection in the Microsoft Excel 2013 program and for statistical analysis the SPSS version 21.0 program (IBM Corporation ©). For the comparison between groups, inferential statistics were used by means of the student’s T distribution test for parametric variables and chi square and Pearson’s for non-parametric variables, considering a significant p <0.05. The calculation of the sample size was performed using a formula for equivalence of 2 means with a confidence of 95% and a power of 80% bilateral, with a mean of 18.43 CFU * 105 and standard deviation of 17.25 with an interval to demonstrate the equivalence of +/- 10 required a total of 30 participants per group.
Results
A total of 34 patients were included in the study, which were assigned by lot to two groups, 17 patients corresponded to the control group (daily bath) and 17 patients to the intervention group (bath every 3 days).
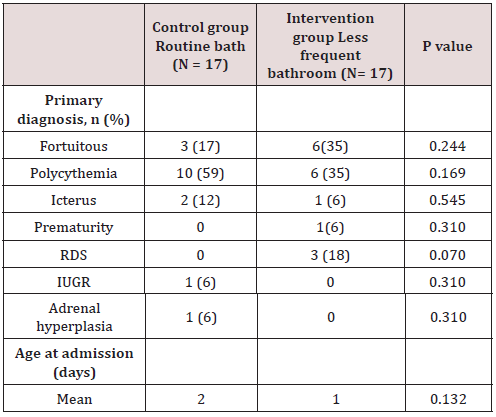
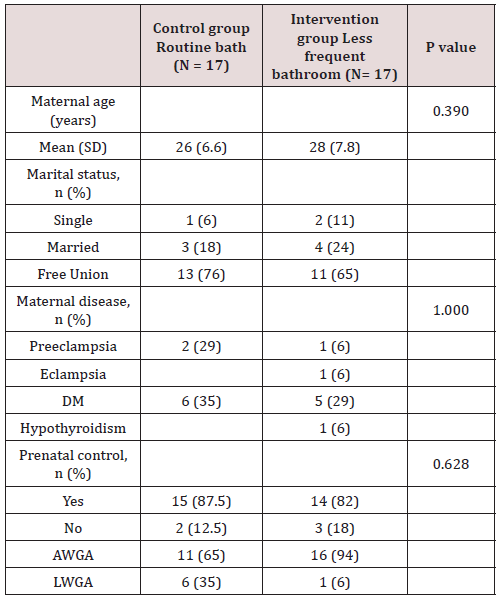
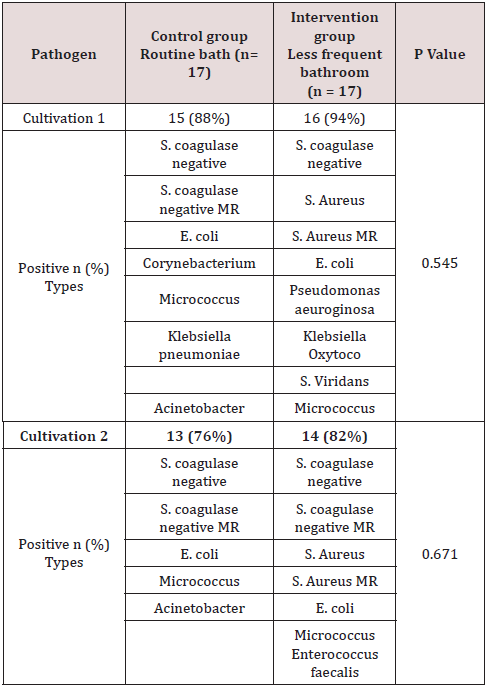
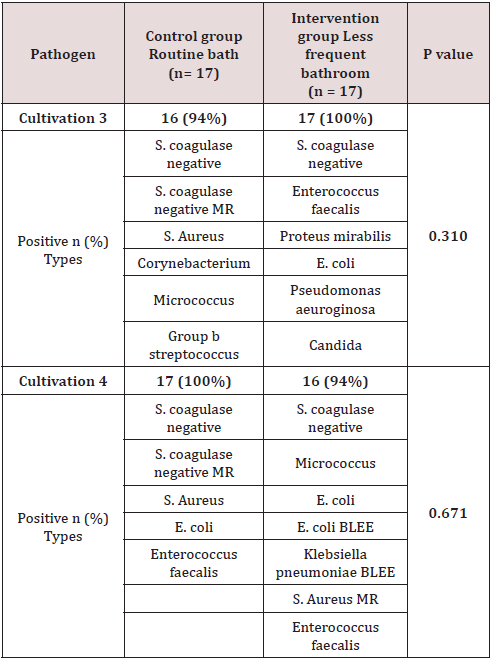
Of the total of 34 patients included, 56% (n=19) corresponded to the female gender and 44% (n=15) to the male gender. Likewise, 41% (n=14) of births in the general population were deliveries and 59% were caesarean sections (n=20). In the control group (group 1) a gestational age was recorded in weeks of 37.7 with a SD ± 1 and, in the intervention group (group 2), a gestational age of 37.8 ± 1.5. 6 late preterm patients were included (between 34-36.6 SDG), 2 corresponding to the first group and 4 to the second, the rest were term patients, no patient was older than 42 weeks of gestation (post-term). Regarding the weight at birth, an average weight of 2622 grams (± 534) was reported in the control group and 2862 grams (± 573) in the intervention group. Of the entire universe of patients, 79% (n=27) had an adequate weight for gestational age (AWGA), 7 patients reported a low weight (LWGA), the majority belonging to the control group. There was no significant difference between the two groups regarding gestational age, however, a value of p = 0.034 was found regarding trophism. Table 1 shows the demographic characteristics of each group. Among the most common admission diagnoses, we find polycythemia 47% of the general population (n=16), accidental delivery 26% (n=9), jaundice 9% (n=3), respiratory distress syndrome 9% (n=3), other diagnoses were prematurity, intrauterine growth retardation, and adrenal hyperplasia. A p = 0.070 was found in the diagnosis of RDS since all patients were from the intervention group (Table 2). Among the maternal characteristics we find an average maternal age of 27 years, 70% (n=24) in a free union marital status. The maternal diseases that occurred during pregnancy were pre-eclampsia and eclampsia. The existing maternal diseases were diabetes mellitus and hypothyroidism. According to the guidelines established by the Official Mexican Standard NOM-007-SSA2-2016, for the care of women during pregnancy, childbirth and the puerperium, and of the newborn, in relation to control, we found that 85% (n=29) had adequate prenatal care in both groups. Baseline cultives were obtained before and after the first bath, and 3 days after admission in each patient. 68 cultives from each group were obtained throughout the study (n=17 for both groups). The results of the cultives of each group are presented in tables 3,4 and 5. Despite the presence of pathogens in the skin cultives, none of the patients developed any signs or symptoms of infection during participation in this study.
Discussion
Controversy exists about when to bathe hospitalized newborns to reduce the risk of colonization of harmful flora and whether this predisposes to local and systemic infections; without affecting physiological and behavioral stress, since an increase in stress has been described, especially in premature babies [12]. In this study, late preterm and term neonates were studied, however, a limitation of this study is that the changes in behavior of the baby were not objectively measured, as to confirm this theory. It is recommended to seek strategies to reduce this stress, one of them is a change in the frequency of bathing in patients hospitalized in the Neonatal Intensive Care Unit. It has been seen that the decrease in the frequency of bathing has shown beneficial effects in the patient admitted to the NICU since it preserves the protective properties of the skin, mainly in premature infants [1]. The newborn’s skin becomes colonized with microorganisms within 24 hours of birth. The first areas to be colonized are armpit, navel, genitals, groin, and abdomen. The prevalent microorganisms are coagulase-negative staphylococcus and diphtheroid [3]. Staphylococcus epidermidis, one of the 13 species of coagulase-negative staphylococci, is the most common vaginal organism and rapidly colonizes the surface of the skin, it is the organism that predominates on the skin of most newborns [10]. In preterm newborns, many invasive infections are caused by this bacteria [13]. Other common microorganisms are Staphylococcus saprophyticus (perineum), Staphylococcus capitis (sebaceous areas), corynebacterium (intertriginous areas), propionibacterium (follicles, sebaceous glands), acinetobacter (moist, intertriginous areas, perineum [14], malas. Lactobacillus spp dominates the vaginal area [6].
Consistent with what has been reported in the literature, the main saprophytic microorganism found in this study was coagulasenegative staphylococcus. The microbiome of neonates requiring admission to the NICU differs from healthy term neonates. The microbiome of the infant in the NICU is less diverse and less in the number of anaerobes, which contribute to an increase in the rates of infection and inflammatory processes in this population [15]. Pammi et al. in 2017 reported that the most abundant phyla in premature skin were Firmicutes and Proteobacteria, and the predominant genus was Staphylococcus. The skin microbiome of preterm infants has increased the abundance of Firmicutes and decreased that of Proteobacteria compared to full-term infants. They found a greater abundance in Staphylococcus, Corynebacterium and Prevotella species and a decrease in the abundance of Brevundimonas, Flavobacterium and Sphingobacterium species at the genus level in premature newborns [16]. In 2018, Young et al. included a total of 61 infants, 41 term newborns and 20 preterm infants, 35 of the term newborns went to joint accommodation with the mother, the rest, including preterm newborns, entered the hospital. NICU area. The dominant bacterial phylum on the skin of neonates was Proteobacteria and Firmicutes. They found that the skin microbiota of preterm infants differs from term newborns, with higher enrichment of Staphylococcus and taxa that are typically associated with the fecal microbiota, such as Escherichi [13]. The results in this study demonstrated no statistical significance in the noxious flora between both assigned groups (daily bath vs bath every third day). According to what is reported in the literature, pathogens such as Staphylococcus Aureus and E. Coli were detected in most of the cultives in both groups, before and after bathing; E. Coli BLEE and MRSA were found in the 4th culture (after bathing) in the intervention group, however, no newborn from any of the assigned groups developed an infection as a result of their participation in this study.
Conclusion
There were no differences in the presence of saprophytic flora or pathogenic flora that colonize the skin of the neck region between both groups. The main saprophytic microorganism found was coagulase negative Staphylococci; and the main pathogens detected in the cultives of both groups were Staphylococci Aureus and E. Coli. There were no manifestations of skin or systemic infectious processes during the study. The findings in this study suggest that late bathing every 3 days in the NICU is safe. This allows the nursing staff to redesign their routines and allocate this time to other priority activities. However, a study with a larger sample size is required to reduce the risk of presenting a type II statistical error.
References
- Akter T, Dawson A, Sibbritt D (2015) What impact do essential newborn care practices have on neonatal mortality in low and lower-middle income countries? Evidence from Bangladesh. J Perinatol 36: 1–6.
- Adejuyigbe EA, Bee MH, Amare Y, Omotara BA, Iganus RB, Manzi F, et al. (2015) “Why not bathe the baby today ?” : A qualitative study of thermal care beliefs and practices in four African sites. BMC Pediatr 15(156):1–7.
- Quinn D, Newton N, Piecuch R (2005) Effect of Less Frequent Bathing on Premature Infant Skin. JOGNN 34(6): 741-746.
- Kuller JM (2014) Newborn & Infant Nursing Reviews Update on Newborn Bathing. NewbornInfant Nurs Rev 14(4):166–170.
- Sarkar R, Basu S, Agrawal RK, Gupta P (2010) Skin care for the newborn. Indian 47: 593–598.
- Dunn AB, Jordan S, Baker BJ, Carlson NS (2017) The Maternal Infant Microbiome: Considerations for Labor and Birth. MCN Am J Matern Child Nurs 42(6): 318–325.
- Rutayisire E, Huang K, Liu Y, Tao F (2016) The mode of delivery affects the diversity and colonization pattern of the gut microbiota during the first year of infants’ life: A systematic review. BMC Gastroenterol. 16(1) :1–12.
- Forslund K, Sunagawa S, Coelho LP, Bork P (2014) Metagenomic insights into the human gut resistome and the forces that shape it. Bioessays 36(3):316–329.
- Behring A, Vezeau TM, Fink R (2003) Timing of the newborn first bath: a replication. NN 22(1): 39–46.
- So HS, You MA, Mun JY, Hwang MJ, Kim HK, Pyeon SJ, et al. (2014) Effect of Trunk-to-Head Bathing on Physiological Responses in Newborns. JOGNN 43(6): 42–751.
- Varda KE (2000) The effect of timing of initial bath on newborn’s temperature. JOGNN 29(1): 27–32.
- Sinha A, Sazawal S, Pradhan A, Ramji S, Opiyo N (2015) Chlorhexidine skin or cord care for prevention of mortality and infections in neonates. Cochrane Database Syst Rev (3): CD007835.
- Younge NE, Araújo pérez F, Brandon D, Seed PC (2018) Early-life skin microbiota in hospitalized preterm and full-term infants. BMC 6(98):1–11.
- Hoath SB, Shah KN (2017) Physiologic Development of the Skin. En: Polin, R. A., Abman, SH Rowitch, DH Benitz, WE Fox, WW (eds). Fetal & Neonatal Physiology. 5a ed. Philadelphia. Elsevier pp.505-506.
- Rodriguez J, Jordan S, Mutic A, Thul T (2017) The Neonatal Microbiome: Implications for Neonatal Intensive Care Unit Nurses. MCN Am J Matern Child Nurs 42(6): 332‐
- Pammi M, Brien JLO, Ajami NJ, Wong MC, Versalovic J, Petrosino JF (2017) Development of the cutaneous microbiome in the preterm infant : A prospective longitudinal study. PLoS One 12(4):1–1.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...