Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1709
Research Article(ISSN: 2641-1709) 
A Systematic Literature Review of Music Induced Hearing loss Volume 6 - Issue 4
Supriya Gopal*
- The School of Advanced Education, Research and Accreditation (SAERA), University Isabel I de Castilla, Spain
Received: May 26, 2021 Published: June 08, 2021
Corresponding author: Supriya Gopal, The School of Advanced Education, Research and Accreditation (SAERA), University Isabel I de Castilla, Spain
DOI: 10.32474/SJO.2021.06.000245
Abstract
Music induced hearing loss is caused by listening to loud music on Personal Music Players (PMP) or attending discotheques regularly over a period of time. Exposure to loud music causes physiological changes and damage in the Basilar Membrane especially to the Outer Hair Cells, Inner Hair Cells, and the Auditory Nerve. Damage includes loss of sensitivity to soft sounds, presence of recruitment, tinnitus, reduced frequency selectivity and reduced flow of synapses and information to the auditory cortex. This leads to poor auditory discrimination and reduced temporal fine structure of sounds and poor pitch perception. Hearing loss acquired due to exposure to intense loud music, is typically greatest over the frequency range 3–6 kHz, hence the high frequency hearing loss. Numerous studies done on threshold changes, temporary threshold shift and permanent threshold shift using Pure Tone Audiometry and Oto-Acoustic Emission have indicated high frequency hearing loss in young adults after exposure to loud music at discotheques, overuse of Personal Music Players (PMP) in teenagers and children. Pure Tone Audiometry (PTA) changes weren’t noted, however Oto-Acoustic Emissions (OAE) showed changes and indication of high frequency hearing loss and tinnitus was noted as a symptom after loud music exposure.
Keywords:PTA: Pure Tone Audiometry; OAE: Oto-Acoustic Emissions; PMP: Personal Music Players; MIHL: Music Induced Hearing Loss; TM: Tympanic Membrane
Introduction
Music is a rudimentary part of human life and is important for emotional wellbeing. WHO (2020) estimated 1.1 billion young people (aged between 12–35 years) are at risk of hearing loss due to unsafe listening practices? A disorder in which a person has been exposed to high levels of music over a prolonged period of time is Music Induced Hearing Loss (MIHL). People of all ages, demographics and gender may be affected by MIHL. This disorder is caused due to too much sound intensity going through and into the auditory system with longer period of exposure.
Audition or Hearing
The auditory system is a system for the sense of hearing. The human ear mechanism has a frequency range of 20 Hz- 20 kHz. It is divided into two sub-systems: The Peripheral System consists of outer ear, middle ear (Tympanic membrane, middle ear ossicles, eustachian tube and the middle ear muscles) and inner ear (Vestibular system and Cochlea). The central system is from the cochlear nucleus to the primary auditory cortex. The outer ear is made up of the pinna and the external auditory meatus. The pinna or the ‘auricle’ is commonly known as the ear, is a funnel-like structure, which not only directs sound into the external auditory meatus but also acts as a resonant tube, boosting a peak between 4 and 6 kHz of up to 10 dB HL [1]. The pinna helps in localization of sound in space and collects acoustic energy from the environment and directs it to tympanic membrane via external auditory meatus. Two ears side by side helps in binaural sound localization (Silva et al., 2014). A secondary resonance is provided to the magnitude of 17 to 22 dB HL at roughly 2.7 kHz, as sound waves travel down through the external auditory meatus [2]. At the end of this meatus lies the tympanic membrane. The Tympanic Membrane (TM) marks the boundary between the outer ear and the middle ear and is 55 mm. The eardrum moves synchronously in response to variations in air pressures, which constitute sound waves. The drum’s vibrations are transmitted through the ossicular chain to the cochlea. The diameter of the TM helps in conversion of the acoustic energy to mechanical energy. When sound waves enter the ear, they strike the tympanic membrane and this force of the sound wave strike and transmits the vibrations further in, to the ossicles of the middle ear. The middle ear ossicles are a group of 3 bones, and they are the smallest bones in the human body. The malleus (hammer), incus (anvil) and the stapes (stirrup). The sound waves from the tympanic membrane are amplified by the auditory ossicles. The chain of three tiny bones in the middle ear acts as an impedance transformer, efficiently coupling the relatively large low impendence movement of the ear drum to the smaller, high impedance movement of the fluid in the cochlea of the inner ear.
Inner Ear: It consists of the vestibular system and cochlear system. The vestibular system represents the peripheral balance system. The cochlea system represents the ‘hearing’ part of the inner ear and is situated in the petrous portion of the temporal bone. The cochlea is divided into 3 sections, scale vestibuli, scala media and scala tympani. It interacts with the middle ear via two holes that are closed by membranes: the oval window, which is located at the base of the scala vestibuli and which undergoes pressure from the stapes (‘middle ear’), and the round window, which seals the base of the tympanic membrane and is used to relieve pressure. The scala media houses the organ of Corti which rests on the basilar membrane the surface of the inner hair cells (one row) and the outer hair cells (3 rows) by the pillars or Corti. When sound pressure is transmitted to the fluids of the inner ear by the stapes, the pressure wave deforms the basilar membrane in an area that is specific to the frequency of the vibration. In this way, higher frequencies cause movement in the base of the cochlea, and deeper frequencies work at the apex. This characteristic is known as cochlear tonotopy. The gradual thickening of the basilar membrane from the base (20kHz) to the apex (20Hz). This transmission of sound is known as ‘Travelling wave theory’. This frequency tuning is closely linked to the electro motility of the outer hair cells (OHCs) and is defined by the fibres of the auditory nerve and the inner hair cells (IHCs) that generate the neural signal.
Cochlear fluids: Perilymph: There are two types of perilymph: the perilymph of the scala vestibuli, and that of the scala tympani. Both have a composition similar to cerebro-spinal fluid (CSF): rich in sodium (140mM) and poor in potassium (5mM) and calcium (1.2mM). The perilymph in the scala vestibuli comes from blood plasma across a hemto-perilymphatic barrier, whereas that of the scala tympani originates from CSF.
Endolymph: is created from perilymph. The endocochlear potential is the sum of two potentials: a positive potential caused by active secretion of K+ by the stria vascularis (120mV) and a negative potential created by the passive diffusion of K+ ions from the hair cells (40mV). The spiral ganglion is formed from the primitive otocyst. It differentiates very early, before the organ of Corti. In a human, it is composed of 30 to 35,000 bipolar spiral ganglion neurons (SGNs) of two main types. Large and slightly myelinated type I neurons (accounting for more than 90%) are connected to inner hair cells; small and unmyelinated type II neurons are connected to outer hair cells. Both types have central axons delivering messages to the cochlear nuclei. Central System: auditory messages are conveyed to the brain via two types of pathway: the primary auditory pathway which exclusively carries messages from the cochlea, and the non-primary pathway (also called the reticular sensory pathway) which carries all types of sensory messages.
Primary Auditory Pathway: the first relay of the primary auditory pathway occurs in the cochlear nuclei in the brain stem, which receive Type I spiral ganglion axons (auditory nerve); at this level an important decoding of the basic signal occurs: duration, intensity and frequency. The second major relay in the brain stem is in the superior olivary complex: the majority of the auditory fibres synapse there having already crossed the midline. Leaving this relay, a third neuron carries the message up to the level of the superior colliculus (mesencephalus). These two relays play an essential role in the localisation of sound.
A last relay, before the cortex, occurs in the medial geniculate body (thalamus); it’s here that an important integration occurs preparation of a motor response (e.g., vocal response). The final neuron of the primary auditory pathway links the thalamus to the auditory cortex, where the message, already largely decoded during its passage through the previous neurons in the pathway, is recognized, memorized and perhaps integrated into a voluntary response.
Physiology of Music Induced Hearing loss
Exposure to loud and high levels of music leads to several physiological changes in the cochlea and the auditory pathway leading up to the auditory cortex. Disturbance and damage to the outer hair cells in the cochlea leads to loss of hearing sensitivity to soft sounds, presence of recruitment and decreased frequency selectivity. Disturbance and damage to the inner hair cells or the synapse in the cochlea causes degeneration of the neurons in the auditory nerve and reduced synapses to the auditory cortex causing poor auditory discrimination, reduced pitch perception and poor temporal fine discrimination.
Conceptual Repercussions of Music Induced Damage
Outer Hair Cell (OHC) Damage
Damage to OHC impairs the function and leads to three perceptual reactions. First is the reduction of basilar membrane vibration at the peak. Hearing loss acquired due to exposure to intense loud music, is typically greatest over the frequency range 3–6 kHz, hence the high frequency hearing loss. Second damage is reduced frequency selectivity and this in turn reduces the spectral shape of sounds which is important in distinguishing different musical instruments. The third reaction is the presence of recruitment (abnormal growth of loudness) [3].
Inner Hair Cell (IHC) Damage
Damage to IHC causes disruption in temporal synchrony intervening the basilar membrane wave form and action potentials i.e., spikes, in the auditory nerve. It reduces the spikes along the auditory nerve leading to reduced neural coding and properties of sound. This concealment causes poor discrimination of sound, reduced sensitivity, disorganization of temporal structure of sounds, reduced sound localization and poor pitch perception. When the damage is complete over a certain region of the basilar membrane, then little or no information from that region is conveyed to the auditory nerve, known as dead region. Dead regions are common in rock music listeners or regular discotheques goers [4].
Perceptual Repercussions
Deterioration of OHC’s, IHC’s and neurons lead to reduced synapse to the central auditory cortex, due to this central auditory cortex applies greater gain to the signal coming from the auditory nerve and this leads to Tinnitus and Hyperacusis. Tinnitus occurs due to increased gain amplification applied by the auditory cortex to neuronal noise that would not be audible normally. The presence of hyperacusis could arise from greater gain being applied to high level inputs, resulting in greater than normal loudness.
Cochlear changes due to music exposure
Preventable hearing loss occurs when the cochlea is damaged by the repeated exposure to loud music. The underlying mechanism of music induced hearing loss is outer hair cell dysfunction. This can lead to either cellular trauma in response to transient highintensity sound such as loud rock amplifier exposure or due to metabolic exhaustion of the outer hair cells in response of continuous acoustic input. Prolonged, excessive music exposure can induce metabolic and mechanical changes in the organ of Corti [5]. Excessive noise exposure leads to hearing loss and other symptoms include tinnitus, hyperacusis and dizziness [3].
Tinnitus
The perception of noise in the ears or head when no external sound is present [6,7]. There are a number of types of tinnitus, and not all result from cochlear damage. Often tinnitus is transient, and it is normal to occasionally hear a brief ringing in the ear, which dies away within a few seconds. However, when chronic tinnitus is experienced after exposure to loud sounds, such as rock concerts and discotheques it is not just a warning sign, but a clear manifestation of cochlear injury. Consider the ringing sound to be caused by hair cells and neurons actually in the process of dying. Such cells generate a neural injury discharge because the cell membrane breakdown causes repeated depolarization (excitation) and/or uncontrolled release of neurotransmitters. In the case of severe acoustic trauma, tinnitus can persist and becomes permanent. It has been suggested that the initial neural injury discharge sets up (synaptic) connections in a network of auditory neurons at a more central (cortical) brain level, and that these cells continue to fire spontaneously (perhaps because a local positive feedback circuit is established or because local inhibitory neuron activity is reduced). Chronic tinnitus can be as devastating on quality of life as hearing loss. Clearly, any recreational activity that induces tinnitus should be avoided [8]. Derebery [9] reported that 25% experienced tinnitus post-concert. In a survey of over 9000 young adults, Chung et al. [10] found that 43% had experienced tinnitus after attending a nightclub, and 61 % after a live-music concert.
Hyperacusis
Hyperacusis or sensitive hearing is reduced tolerance to sounds of normal intensity leading to pain and discomfort and this occurs due to the central auditory processing centre perception. It is a psychological, perceptual, and social scope that can affect quality of life living with this condition. People with hyperacusis often perceive every day environmental sounds louder and intolerable that are typically not loud to others. Although all sounds may be perceived as too loud, high frequency sounds may be particularly troublesome. Numerous people experience sensitivity to sounds, but true hyperacusis is rare. It can affect all ages and can be unilateral or bilateral, often associated with tinnitus. Hyperacusis can lead to withdrawal, social isolation, fear of normal sounds (called phonophobia), and depression [11,12]. Symptoms include sensitivity to everyday sounds, often starting in one ear then progressing to both ears, difficulty tolerating ordinary environments and situations, isolation and pain, or physical discomfort with sounds.
Decibel exposure
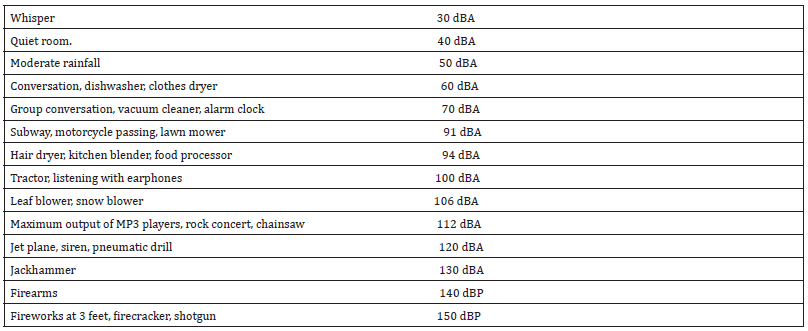
In general, any sound, noise or music exposure of 85 dBA is considered to cause damage to the outer hair cell and listening to music at this level for approximately 8 or more hours is likely to result in permanent damage. Further, as volume increases, the exposure time decreases. A risk to hearing arises from a combination of how loud the sound is and how long a person is exposed to it. It can take many months or even years for the effect on hearing to become apparent but listening to loud music or other media over a long time can gradually cause permanent hearing loss issues. Past literature and studies that indicate the effects of music on hearing is greater than the effects of noise. About one in six Australians has some kind of hearing loss. Experts believe that 37% of the hearing loss experienced by individuals was caused by preventable and repeated exposure to loud music, that is known as Music-induced Hearing Loss [13].
Government Regulations on Acceptable Noise Levels
The prevalence of noise induced hearing loss (NIHL) is a public health concern with government regulations (i.e., POEO EPA Australia) in place to protect general population in occupational settings. Sound pressure levels (SPL) of various recreational sounds including music have been measured and exposure dosages monitored as per the national regulations. Various governmental agencies, such as the Occupational Safety and Health Administration (OSHA) and The National Institute for Occupational Safety and Health (NIOSH) in the United States, the European Agency for Safety and Health at Work (EASHW) in Europe and Work Health and Safety in Australia has regulations and standards for noise exposure. Decibels exceedingly more than 85dB should be limited in a day. According to WHO the exposure standard for noise involves two measures:
a) LAeq, 8h of 85 dB(A) means that a person can’t be exposed to this level of intensity for more than 8 hours a day.
b) LCpeak of 140 dB(C) means that a person can’t be exposed to this level of intensity. Any exposure above this peak can create almost instant damage to hearing.
Pure Tone Audiometry
Clinically, audiometry is used to evaluate hearing and establish thresholds from 250Hz -8000 kHz, and this procedure is used for evaluating MIHL. Audiometrically, MIHL typically presents as a decrease in hearing sensitivity thresholds that generally centers around 4000 Hz, and gradually affects hearing thresholds at surrounding frequencies, corresponding to cochlear damage in adjacent regions [14,15] other symptoms include tinnitus (i.e., the perception of noise in the ears or head when no external sound is present), hyperacusis and dizziness [3].
Otoacoustic Emissions
Clinically, slight changes to the cochlea can go undetected with audiometry, hence objective measures like OAEs are needed for early detection [16]. Otoacoustic emissions (OAE) is non-invasive and low-level sound emitted by the cochlea either spontaneously or evoked by an auditory stimulus. Specifically, OAEs provide information related to the function of the outer hair cells (OHC). The most frequently evoked otoacoustic emissions in the clinical setting are transient stimulus otoacoustic emissions (TOAE), and distortion product otoacoustic emissions (DPOAE). DPOAE in particular is used in detection of Noise Induced Hearing Loss [17] and can be used in differentially diagnosis, separating highfrequency hearing loss from other aetiologies such as Presbycusis and Ototoxicity [18].
Music is noise
Acoustically, music has a similar structure as noise. Both music and noise have significant low frequency fundamental energy which musicians call the tonic (or note name), and higher frequency harmonic or broadband energy. Music, like noise, can have sound levels in the 60-70 dBA region and also sound levels in excess of 110 dBA. One difference is that many sources of industrial noise exposure are steady state, in that the sound levels have minimal variation over time, whereas music is characterised by highly fluctuating levels. This intermittency over time has been studied for decades beginning with the 1966 CHABA report that defined the on/off fraction rule which essentially weighted the exposure by the time varying levels of the noise source – the quieter periods are balanced against the louder periods. This is one reason why music exposure for any given gross measure, such as dBA or Leq, tends to result in less hearing loss for musicians and those who listen to music, than their industrial colleagues. The audiometric configuration of long-term noise exposure is even similar to that of long-term music exposure. It is frequently difficult to differentiate a noise induced hearing loss from a music induced hearing loss purely on audiometric data. A thorough case history is required as the differentiating element. It is therefore not surprising that many of the research results using noise as a stimulus can apply (or have been applied) to music in many national and international regulations and policies.
Temporary Threshold Shifts
Noise can cause reversible hearing loss, and this is called temporary threshold shift (TTS). This occurs on exposure to loud sounds like loud music and this causes fatigue of hair cells and causes tinnitus (ringing). TTS can be caused by listening to ordinary digital music devices at volumes that are not especially high (e.g., 100 dB) [19]. Sound pressure levels can routinely reach levels of 126 dB [21]. Non-occupational noise is also regularly encountered during recreational activities and is a source of premature hearing reduction.
Permanent Threshold Shift
Source: American Speech, Language and Hearing Association, Dangerous and Safe Noise Levels.
Continuous or repeated exposures to noise leads to TTS and exposure over a period of time leads to permanent threshold shift (PTS) if repeated [20]. Therefore, PTS can be defined as noise-induced threshold shift that persists after a period of recovery subsequent to the exposure. In animal studies, recovery has been reported for periods extending up to 3 weeks, therefore it may be premature to define a threshold shift as temporary until at least 3 weeks post-exposure, when a permanent threshold shift arises, as the cochlear hair cells go into permanent fatigue and fail to relax leading to permanent damage. While the smallest level of TTS or PTS that can be reliably measured in an individual has not been well defined given test-to-test variability in individuals, several standards have been set for what is considered a significant hearing loss or “standard threshold shift” (STS). The Occupational Safety and Health Administration (OSHA) states that an STS is a 10 decibel (dB) increase in hearing threshold averaged across 2000, 3000 and 4000 Hz in the same ear from an individual’s baseline or recent annual audiogram (29 CFR 1910.95). An STS is a reportable work-related injury once it has been reconfirmed with a retest within 30 days of the initial test and results in a hearing threshold of at least 25 dB in the affected ear. Therefore, most occupational hearing loss or PTS is under reported since OSHA only requires an STS to be reported. Peak noise levels, in dB, are provided in the following Table 1.
Hearing loss
Hearing loss is not limited to adults. Data from a populationbased study in audiometry conducted during a 1988-94 survey of over 6,100 children and adolescents aged 6 to 19 years indicates that approximately 15% have low-frequency hearing loss of at least 16 dB in one or both ears. Male adolescents were found to have a greater prevalence of high-frequency hearing loss than females. No significant differences were observed by race and ethnic groups; however, children from families with low poverty-to-income ratios have more high-frequency hearing loss than children from the middle and high poverty-to-income ratios, suggesting that class disparities may exist (Niskar, 1998).
Objective
Previous research has demonstrated that occupational exposure to loud noise has led to audiometric evidence of Noise Induce hearing Loss (NIHL). Thus, it raises the question about the impact of occupational exposure to music on hearing in children, teenagers, and young adults. For this purpose, the following research questions will be systematically reviewed on occupational music exposure in children, teenagers, and young adults.
a) What are the factors and associated symptoms of hearing loss due to music in children, teenagers, and young adults?
b) Can occupational music exposure lead to temporary or permanent threshold shifts as evidenced by audiometric testing?
c) Does evidence exist regarding OAEs and early detection of hearing loss in this population?
Method
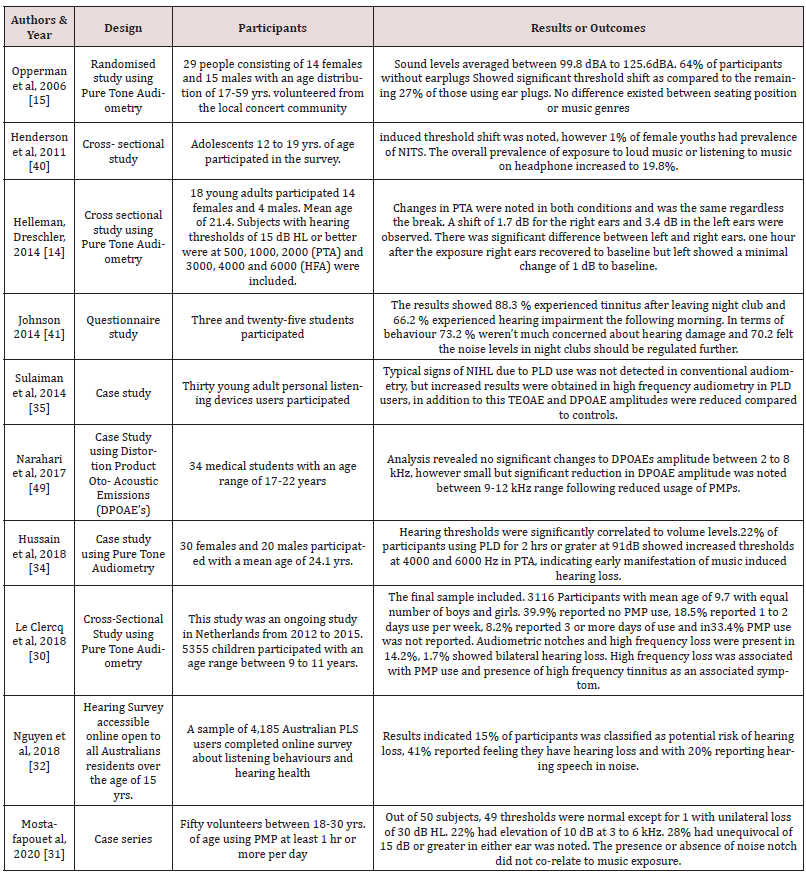
Substantial literature review was conducted on music induced hearing loss from peer reviewed and scholarly journal articles from different database such as PubMed, NCBI (The National Centre for Biotechnology Information advances science and health by providing access to biomedical and genomic information) Google scholar and Cochrane Library. The database was extracted from Google and Safari search engines. Thirty-two journal articles and 2 theses were reviewed and a priori decision was made to use only peer reviewed journals consisting of search strategy phrase ‘children, teenagers, young adults, cause, audiometry testing, Oto Acoustic Emissions, overuse, personal music players, discotheques, music exposure, decibel levels, tinnitus, hyperacusis, cochlear changes and hearing losses. Ten peer reviewed relevant journals relevant to this study are discussed in the following table.
Results
Table 2 summarizes the ten studies chosen for this literature review to answer the objective questions. All studies outline the design, participants, and outcomes. The selected studies measured the outcome of loud music exposure by using personal music players in children and adolescents and attending discotheques with loud music by young adults. The audiologic tests used were Pure Tone Audiometry and Oto-Acoustic Emissions.
Discussion
The review of articles included in this study demonstrates great variability exists in measuring music exposure levels and measuring techniques. The word ‘Music exposure’ compasses a wide range of exposure situations (portable music player use, listening to loud music at discotheque, and parties). This study reported 3 specific reviewed aims related to music induced hearing loss, the factors, and associated symptoms of hearing loss due to music exposure in children, teenagers and young adults, music exposure leading to temporary or permanent threshold shifts as evidenced by audiometric testing and existence of evidence regarding OAEs and early detection of hearing loss in this population. In order to evaluate the results this discussion will be based on the aims along with examining limitations of this study and future directions. Tinnitus presence is reported by children, adolescents, and young adults after extensive exposure to loud music. Tinnitus manifestation is seen in adolescents at primary and high school students, listening to loud music on personal music player, this caused increase in tinnitus frequency [22]. Recent exposure to noise or music was associated with a significant increase in tinnitus including an age-related interaction. An association was observed between recent and lifetime exposure. There are several reports of a high prevalence of temporary tinnitus following acute exposure to sounds, including music [23]. Despite the high prevalence of tinnitus in young population due to loud music exposure, the awareness of hearing protection and the associated risks of loud music exposure are extremely low. Future studies should focus on tinnitus as a warning for music induced hearing loss and emphasis on temporary and permanent music indued damage. Zhao et al. [24] managed a considerable review of research that evaluated the association between music exposure and hearing loss. Evidence and conclusions from that review was the differentiation between studies which found a relationship and those which did not, in the use of sensitive audiologic measuring tools. The use of pure-tone averages (PTA) as a test for hearing loss is not nearly as sensitive to cochlear damage as are OAEs, and high frequency audiometric testing, among other audiological tests.
A large study on recreational music exposure affecting hearing and auditory function was conducted by Carter et al. [25] The scientific weakness in some studies were discussed and concluded that imprecision in pure-tone audiometry existed. When conducting pure tone audiometry in detecting hearing threshold of a subject, many variabilities play a vital role in obtaining the response including subject’s motivation, skill and knowledge of the tester, the equipment used for testing, calibration, and testing environment. Oto-Acoustic Emissions as a measure of cochlear function is utilized in many studies and has signified as a promising and sensitive tool in determining MIHL and preclinical damage, i.e., dysfunction of the outer hair cells that precedes decrease in hearing thresholds. The non-invasive, objective, and rapid nature of OAE testing is considered superior to the subjective nature, time-consuming, labour-intensive method of pure tone audiometry [26]. Previous studies showed that OAEs were a reliable method of measuring MIHL, the results from studies evaluated above [27-29] provided sufficient documentation that supports the use of OAEs in a music-exposed population, revealing measurable decreases in OAE amplitude after music exposure. While OAEs provide direct relationship in hearing loss diagnosis it is difficult to provide accurate evidence to support the claim that noise exposure does leads to dysfunction of the auditory system due to the many extraneous variables that may exist. Even when it is accepted that a population (children, adolescents and young adults), are exposed to hazardous levels of music, it is confounded by the complex nature of hearing loss and dysfunction. For instance, the presence of middle ear pathology and impacted cerumen, may all prove relevant when examining the correlation. Longitudinal studies are necessary to support this further [25].
Most studies reported no significant association between puretone air thresholds and exposure to loud music; however, significant changes in hearing thresholds and otoacoustic emissions and high tinnitus prevalence suggest an association between music exposure and hearing loss in children [30]. No difference in pure‐tone threshold, speech reception threshold, or speech discrimination was found among subjects when segregated by music exposure level [31]. It was also noted that males were at higher risk as they were exposed to increased PMPs as compared to females [32]. Puretone threshold changes were noticed in both conditions i.e., tested immediately after music exposure at discotheques and 2 hrs after exposure and the results were same, inconsiderate of the break. Threshold shifts could be averaged for 1000, 2000, and 4000 Hz [33]. Immediately after the threshold shift and on cessation of the music, a change of 1.7 dB for right ears, and 3.4 dB for left ears was noted. Significant difference between left and right ears was also noted. An hour after the exposure, right ears recovered to baseline conditions significantly whereas left ears showed a minimal but clinically irrelevant remaining shift bordering on 1 dB [33]. Hussain et al. [34] concluded on short term music exposure affects DPOAE’s amplitude and can induce temporary noise induced hearing loss with maximum volume settings on PMPs. TOAE and DPOAE showed reduced amplitudes in teenagers using personal listening devices. The deterioration of extended high-frequency thresholds and the decrease in DPOAE amplitudes were more evident in these users [35].
Prolonged use of PMPs does cause a TTS OR PTS, in the high frequencies and loud music in discotheques do cause TTS and PTS and this reflected in OAE’s but not in hearing thresholds, hence we can say awareness needs to be created among PMP users and regular young adult discotheques attendees. The attitudes toward hearing loss and hearing protection among university students is limited and more awareness on hearing loss and tinnitus is needed in this population on hearing protection [36]. There are a wide range of hearing protection available ranging from simple ear plugs, noise attenuators to earmuffs. Good and ANSI recommended earplugs can provide a sound attenuation of 25 dB to 30 dB. Earmuff (circumaural) hearing protectors, provide up to 30 dB of attenuation [8]. Personal music players are now available to everyone in the form iPod, smart phones with music applications, discman, portable stereo systems. Listening to music through PMPs is extremely popular among all ages. Leisure time activities with high-intensity music exposure are also very popular (music concerts, discotheques, cinema). All these activities may be responsible for an early hearing impairment (temporary or permanent hearing threshold shift, hearing loss) starting at a younger age. Interventions should target students from pre-vocational schools and should focus on increasing adolescents’ knowledge of the risks of loud music and how to protect themselves. Besides, hearing education for adolescents and technical modifications of PMP, volume-level regulations for PMP players may be warranted, even though latest PMP technologies having a decibel warning. There is a need for action by the relevant authorities and institutions to enhance public knowledge on MIHL. Exposure can be controlled and prevented by taking proper actions as suggested by Olusanya et al. [37] The regulation, output limits on PMPs are an incomplete solution. Headphones themselves are not regulated and affect output level, and sound output danger is determined both by the sound level and length of listening time [38-55].
Conclusions
The primary goals of this study were to understand how children and adolescents use PMPs, how their usage pattern contribute to the risk of MIHL in this population, and how attitudes and beliefs about PMP use relate to listening behavior. The study reported on literature review on monitoring listening behavior over time, providing accurate measures of listening habits that were previously unavailable in other literatures [56-60]. For the development of effective interventions, we recommend theorybased longitudinal studies among those frequently exposed to loud music to assess these correlations in greater depth. Interpretation of the significance of these findings in relation to music exposure must be made with caution. MIHL is an additive process and even subtle deficits may contribute to unequivocal hearing loss with continued exposure. Continued education of young people about the risk to hearing from recreational music exposure is warranted.
References
- Kollmeier B (2008) Anatomy, Physiology and Function of the Auditory System. D Havelock, S Kuwano, M Vorländer (Eds.), Handbook of Signal Processingin Acoustics. Springer, New York, USA.
- Silva A, Blasca W, Lauris J, Oliveira J (2014) Correlation between the characteristics of resonance and aging of the external ear. CoDAS 26(2): 112-116.
- Moore BCJ (2016) Effects of Sound-Induced Hearing Loss and Hearing Aids on the Perception of Music. Journal of the Audio Engineering Society 64(3): 112-113.
- Kähäri K, Zachau G, Eklöf M, Sandsjö L, Möller C (2003) Assessment of hearing and hearing disorders in rock/jazz musicians: International Journal of Audiology 42(5): 279-288.
- Keppler H, Dhooge I, Vinck B (2015) Hearing in young adults. Part II: The effects of recreational noise exposure. Noise & health 17(78): 245–252.
- Møller AR (2010) Epidemiology of tinnitus in adults. Moller AR, Langguth B, DeRidder D, Kleinjung T (Eds.,), Textbook of tinnitus: Diagnosis and treatment. Springer: New York, USA pp. 29–38.
- Roberts LE, Eggermont JJ, Caspary DM, Shore SE, Melcher JR, et al. (2010) Ringing Ears: The Neuroscience of Tinnitus. Journal of Neuroscience 30(45): 14972–14979.
- Harrison RV (2008) Noise-induced hearing loss in children: A 'less than silent' environmental danger. Paediatrics & Child Health 13(5): 377–382.
- Derebery MJ, Vermiglio A, Berliner KI, Potthoff M, Holguin K (2012) Facing the music: pre- and post-concert assessment of hearing in teenagers. Otology & Neurotology 33(7): 1136–1141.
- Chung JH, Des Roches CM, Meunier J, Eavey RD (2005) Evaluation of noise-induced hearing loss in young people using a web-based survey technique. Pediatrics 115(4): 861-867.
- Baguley DM (2003) Hyperacusis. Journal of the Royal Society of Medicine 96(12): 582–585.
- Halevi-Katz DN, Yaakobi E, Putter-Katz H (2015) Exposure to music and noise-induced hearing loss (NIHL) among professional pop/rock/jazz musicians. Noise & Health 17(76): 158–164.
- Safe Work Australia (2021) Occupational Noise Induced Hearing loss in Australia pp. 1-4.
- Helleman HW, Dreschler WA (2012) Overall versus individual changes for oto-acoustic emissions and audiometry in a noise-exposed cohort. International Journal of Audiology 51(5): 362- 372.
- Opperman DA, Reifman W, Schlauch R, Levine S (2006) Incidence of spontaneous hearing threshold shifts during modern concert performances. Otolaryngology. Head and Neck Surgery 134(4): 667–673.
- Kemp TD (2002) Otoacoustic emissions, their origin in cochlear function, and use, British Medical Bulletin 63(1): 223–241.
- Jansen EJM, Helleman HW, Dreschler WA (2009) Noise induced hearing loss and other hearing complaints among musicians of symphony orchestras. International Archives Occupational Environmental Health 82: 153–164.
- Meyer-Bisch C (1996) Epidemiological evaluation of hearing dam- age related to strongly amplified music (personal cassette players, discotheques, rock concerts)-high-definition audiometric survey on 1364 subjects. Audiology 35: 121-142.
- Le Prell C, Dell S, Hensley B, Hall J, Campbell K, et al. (2012) Digital Music Exposure Reliably Induces Temporary Threshold Shift in Normal-Hearing Human Subjects. Ear & Hearing 33(6): e44-e58.
- Lonsbury-Martin BL, Martin GK, Bohne BA (1987) Repeated Temporary Threshold Shift exposures in monkeys: alterations in hearing, cochlear structure, and single-unit thresholds. Journal of Acoustical Society of America 81(5): 1507-1518.
- Breinbauer HA, Anabalón JL, Gutiérrez D, Cárcamo R, Olivares C, Caro J (2012) Output capabilities of personal music players and assessment of preferred listening levels of test subjects: outlining recommendations for preventing music-induced hearing loss. Laryngoscope 122(11): 2549-2556.
- Bulbul S, Bayar Muluk N, Çakir E, Tufan E (2009) Subjective tinnitus and hearing problems in adolescents. International Journal of Pediatric Otorhinolaryngology 73(8): 1124-1131.
- Gilles A, Van Hal G, De Ridder D, Wouters K, Van de Heyning P (2013) Epidemiology of Noise-Induced Tinnitus and the Attitudes and Beliefs towardsNoise and Hearing Protection in Adolescents. Plos ONE 8(7): 70297.
- Zhao F, Manchaiah VK, French D, Price SM (2010) Music exposure and hearing. disorders: An overview. International Journal of Audiology 49(1): 54-64.
- Carter L, Williams W, Black D, Bundy A (2014) The leisure-noise dilemma: hearing loss or hearsay? What does the literature tell us? 35(5): 491-505.
- Mehrparvar A, Mollasadeghi A, Karimi G, Vahidy M, Mostaghaci M, et al. (2012) Amplitude changes in otoacoustic emissions after exposure to industrial noise. Noise & Health 14(56): 28.
- Bhagat SP, Davis AM (2008) Modification of otoacoustic emissions following ear-level exposure to MP3 player music. International Journal of Audiology 47(12): 751-760.
- Gopal K, Chesky K, Beschoner E, Nelson P, Stewart B (2013) Auditory risk assessment of college music students in jazz band-based instructional activity. Noise & Health 15(65): 246.
- Santos L, Morata T, Jacob L, Albizu E, Marques J, et al. (2007) Music exposure and audiological findings in Brazilian disc jockeys (DJs). International Journal of Audiology 46(5): 223-231.
- Le Clercq C, Goedegebure A, Jaddoe V, Raat H, Baatenburg de Jong R, et al. (2018) Association Between Portable Music Player Use and Hearing Loss Among Children of School Age in the Netherlands. JAMA Otolaryngology–Head & Neck Surgery 144(8): 668.
- Mostafapour, Sam P, Lahargoue, Kelli, and Gates, George A. (1998) Noise‐induced Hearing Loss in Young Adults: The Role of Personal Listening Devices and Other Sources of Leisure Noise. The Laryngoscope 108(12): 1832-1839.
- Nguyen J, Beach E, Barr C, Gilliver M (2017) Personal Listening Devices in Australia: Patterns of Use and Levels of Risk. Seminars In Hearing 38(4): 282-297.
- Helleman HW, Dreschler WA (2015) Short-term music-induced hearing loss after sound exposure to discotheque music: The effectiveness of a break in reducing temporary threshold shift. International Journal of Audiology 54(Sup 1): S46-S52.
- Hussain T, Chou C, Zettner E, Torre P, Hans S, et al. (2018) Early Indication of Noise-Induced Hearing Loss in Young Adult Users of Personal Listening Devices. Annals of Otology, Rhinology & Laryngology 127(10): 703-709.
- Sulaiman AH, Husain R, Seluakumaran K (2014) Evaluation of early hearing damage in personal listening device users using extended high-frequency audiometry and otoacoustic emissions. Europe Archives of Otorhinolaryngology 271: 1463–1470.
- Alnuman N, Ghnimat T (2019) Awareness of Noise-Induced Hearing Loss and Use of Hearing Protection among Young Adults in Jordan. International Journal of Environmental Research and Public Health 16(16): 2961.
- Olusanya BO, Neumann KJ, Saunders JE (2014) The Global Burden of Disabling Hearing Impairment: A Call to action. Bulletin of World Health Organisation 92: 367–373.
- Chasin M (2018) Music is noise. ENT & Audiology News 27(3).
- Goedert C (2021) Hearing Conservation. OSHA Instruction. Washington, USA.
- Henderson E, Testa MA, Hartnick C (2011) Prevalence of noise-induced hearing-threshold shifts and hearing loss among US youths. Pediatrics 127(1): e39-46.
- Johnson O, Andrew B, Walker D, Morgan S, Aldren A (2014) British university students' attitudes towards noise-induced hearing loss caused by nightclub attendance. Journal of Laryngology and Otolology 128(1): 29-34.
- Knobel KAB, Lima MCMP (2012) Knowledge, habits, preferences, and protective behavior in relation to loud sound exposures among Brazilian children. International Journal of Audiology 51(sup1): S12-S19.
- Lean M (2020) Decibel Level Chart.
- Levey S, Fligor B, Ginocchi C, Kagimbi L (2012) The Effects of Noise-Induced Hearing Loss on Children and Young Adults. Contemporary Issues in Communication Science and Disorders 39(3): 76-83.
- Martin WH, Sobel J, Griest SE, Howarth L, Yongbing SHI (2006) Noise induced hearing loss in children: Preventing the silent epidemic. Journal of Otology 1(1): 11-21.
- McGinnity S, Beach EF, Mulder J, Cowan R (2018) Caring for musicians’ ears: insights from audiologists and manufacturers reveal need for evidence-based guidelines. International Journal of Audiology 57(sup1): 12-19.
- Moore DR, Zobay O, Mackinnon RC, Whitmer WM, Akeroyd MA (2017) Lifetime leisure music exposure associated with increased frequency of tinnitus. Hearing research 347: 18–27.
- Morata TC (2007) Young people: Their noise and music exposures and the risk of hearing loss, International Journal of Audiology 46(3): 111-112.
- Narahari P, Bhat J, Nambi A, Arora A (2017) Impact of usage of personal music systems on Oto-acoustic emissions among medical students. Noise and Health 19(90): 222.
- Niskar AS, Kieszak SM, Holmes A, Esteban E, Rubin C, et al. (1998) Prevalence of Hearing Loss Among Children 6 to 19 Years of Age: The Third National Health and Nutrition Examination Survey. JAMA 279(14): 1071–1075.
- Noise (2021) Rockville: American Speech and Hearing Association p. 2.
- Noreña AJ (2011) An integrative model of tinnitus based on a central gain controlling neural sensitivity. Neuroscience and biobehavioral reviews 35(5): 1089–1109.
- Portnuff CD (2016) Reducing the risk of music-induced hearing loss from overuse of portable listening devices: understanding the problems and establishing strategies for improving awareness in adolescents. Adolescent Health, Medicine and Therapeutics 7: 27–35.
- Robles L, Ruggero M (2001) Mechanics of the Mammalian Cochlea. Physiological Reviews 81(3): 1305-1352.
- Safe Work Australia (2020) Managing noise and preventing hearing loss at work. Safe Work Australia pp. 5-13.
- Sha S, Schacht J (2017) Emerging therapeutic interventions against noise-induced hearing loss. Expert Opinion on Investigational Drugs 26(1): 85-96.
- Sliwinska-Kowalska M, Jedlinska U (1998) Prolonged exposure to industrial noise: Cochlear pathology does not correlate with the degree of permanent threshold shift but is related to duration of exposure. Journal of Occupational Health 40: 123-131.
- Sliwinska-Kowalska M, Kotylo P (2001) Otoacoustic emissions in industrial hearing loss assessment. Noise & Health 3(12): 75–84.
- Vogel I, Brug J, Van der Ploeg CPB, Raat H (2009) Prevention of adolescents’ music-induced hearing loss due to discotheque attendance: A Delphi study, Health Education Research 24(6): 1043-1050.
- Zeng FG (2013) An active loudness model suggesting tinnitus as increased central noise and hyperacusis as increased nonlinear gain. Hearing Research 295: 172–179.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...