Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4544
Research Article(ISSN: 2637-4544) 
Chlorhexidine Alcohol Versus Povidone Iodine Prior to Elective Cesarean Section: A Randomized Open Label Controlled Trial Volume 3 - Issue 3
Nisreen Aref Albezrah*
- Head of Obstetric and Gynecology Department, Taif University, Saudi Arabia
Received:July 08, 2019; Published: July 12, 2019
Corresponding author: Nisreen Aref Albezrah, Head of Obstetric and Gynecology Department, Taif University, Saudi Arabia
DOI: 10.32474/IGWHC.2019.03.000163
Abstract
Objective: To compare the incidence of SSI(surgicl site infection) betweenanti-sepsis solutions Chlorhexidine-Alcohol (CA) and Povidone-Iodine (PI) before elective CS(cesarean section).
Methods: Aprospective randomised controlled trial was conducted from January 2018 tell February, 2019 at the Obstetrics and Gynecology department of King Abd AL-Aziz Hospital, Saudi Arabia. Pregnant women greater than 18 years andreached 37 week of gestation andwere scheduled for elective cesarean delivery wererecruitmed. These womenwere randomly allocated in the ratio of 1:1 and received skin anti-septic preparation A (Chlorhexidine-Gluconate) or B (Povidone-Iodine). The primary outcome was superficial or deep SSI within 30 days after elective CS while the secondary outcomes assessed were hospital length of stay, readmission, endometritis and adverse skin reactions.
Results: There were 490 pregnant women indicated for elective CS randomized to CA group, while 493 to PI group. Significant difference was observed in SSI betweenCA and PI group (4.3% vs. 7.7%; p-value = 0.014). Moreover, difference was also significant in superficial SSI between the two groups (2.9% vs. 5.9%; p-value = 0.030). However, significant difference was not observed for deep SSI between the two groups (1.4% vs. 2.4%; p-value = 0.362). Among the secondary outcomes, significant difference was observed in the mean hospital stay (4.89 vs. 5.67; p-value = 0.001), while no significant difference was observed in hospital readmission and endometritis between the two groups. In conclusionChlorhexidine-Alcohol for pre-operative skin anti-sepsis in elective CS was associated with significantly lower risk SSI when compared to Povidone-Iodinesolution.
Keywords: Cesarean section; Surgical site infection; Chlorhexidine-Alcohol; Povidone-Iodine
Introduction
It has been estimated that more than 18.5 million Cesarean Delivery (CD) are annually performed globally [1]. In the United States (US) it had been estimated that 1.32 million cesarean were performed in the year 2014, which were 32% of all births [2]. Cesarean Section (CS) is one of most frequent obstetrical procedure being performed globally with recent surveys had demonstrated increased trend of performing CS in developed and also in developing countries [3-4].
As cesarean sections numbers performed are increasing, the post-operative complications incidence is also expected to rise. Surgical site infection (SSI) was reported as one of the commonest complications following CD, with incidence from 3% to 15% globally [5-7]. The SSI following CD had been strongly associated with severe morbidity, prolonged hospitalisation, readmission, re-surgery and eventually increased medical charges [8]. It not only imposed significant financial burden on healthcare system of a country but also imposed psychological burden on mother. Management of SSI following CD can pose challenge for mother and newborns due to bonding interruption and breastfeeding to new born, not many antibiotic options available and logistics related to wound care. Importantly, SSI has been significantly related to maternal mortality rate as high as 3% [9]. Utilization of more efficacious anti-sepsis solution could reduce cesarean-related surgical site infection.
The selection of antiseptic skin reagent before surgery is vital to prevent SSI. Povidone-iodine (PI) being low priced and having broader antimicrobial activity against b bacteria, viruses, protozoa and fungi made the solution as most widely used anti-septic [10]. However, evidence from the literature reported it to be associated with skin irritation and staining and average drying time for optimal function was reported as three minutes [11]. Importantly, the use of PI had been related with a delay in collagen maturation and epithelization, thus impaired wound healing [12].
Chlorhexidine-Alcohol (CA) also had a broad spectrum bactericidal activity and more rapid onset against certain bacteria (i.e. S. aureus and E. coli) with optimal effect within 20 seconds and anti-bacterial effects lasting for greater than 48 hours on the skin [13]. It is high priced compared to PI and having no color thus making hard to identify on the skin of patient once applied are potential disadvantages of CA [14]. However, low rate of skin irritation, residual effect long lasting, not affected by the body fluid and effectiveness against non-sporulating bacteria made the preparation promising to use to reduce SSI [14].
Clinical studies have been conducted comparing incidence of SSI with anti-sepsis use for various surgical procedures. The two meta-analyses that included a total of 7 clinical trials reported that chlorhexidine-based antiseptics were superior to iodine-based products in reducing SSI after numerous surgical procedures but did not include CD [15,16]. The results of the two Randomised Controlled Trial (RCT) that compared the efficacy of Chlorhexidine-Alcohol (CA) compared to Povidone-Iodine (PI) among women undergoing CD reported conflicted findings. Ngai et al. [17] reported no significant difference in the incidence SSI, while Tuuli et al. [18] demonstrated a significant reduction in SSI with CA when compared to PI. The research objective was to compare the incidence of SSI of two antiseptic preparations before elective CS being used at our institution. It was hypothesised that CA would be superior to PI in reducing cesarean related SSI.
Methods
Study design and ethical considerations
A single centre open-label, prospective RCT was conducted from start of January 2018, to February end, 2019 at the Obstetrics and Gynecology department of King Abd AL-Aziz Hospital, Jeddah, Kingdom of Saudi Arabia (KSA). The clinical research was initiated after the ethical approval from the Institutional Review Board (IRB) committee of King Abd AL-Aziz Hospital. All the participants prior enrolling in this research gave written informed consent. The study participants were comprehensively briefed about research objective, interventions, process involved and potential risks and benefits of enrolling in this research. The participation in this research was voluntary with participants had the right to withdraw at any point during the research. Study participant’s data anonymity and confidentiality was ensured during the entire research period and post-research.
Participants
Pregnant women greater than 18 years were eligible for enrollment provided reached 37 week of gestation and were scheduled for elective CD. Subjects were excluded if allergic to iodine or chlorhexidine, skin infection adjacent to operative site, abnormal placentation (previa/ accrete), urogenital tract infection within the time period of 2 weeks of delivery, steroid use for a period of two or more weeks during their pregnancy, diagnosed with clinical chorioamnionitis, auto-immune diseases. Moreover, eligible participants were also excluded if it was perceived that the subject is less willing to come back for post-operative assessment and non-willingness to give written informed consent.
Randomization, allocation and masking
The eligible women recruited in this randomized controlled clinical trial were randomly allocated in the ratio of 1:1 using a computer generated random sequence. Allocation of treatment was written on cards which were kept concealed in identically sealed sequential numbered opaque envelops and were kept in the operating room. Till the consent process was not completed study allocation envelops were kept sealed and subject entered in the operating room picked envelop so that antiseptic solution allocated could be applied as picked by subjects. In this RCT, masking was not possible because antiseptic agents having different colors when applied to skin.
Interventions
The study participants were allocated randomly in the 1:1 ratio to receive skin antiseptic preparation either A or B.
- Skin antiseptic preparation A (CA Group): 2% Chlorhexidine Gluconate in 70% isopropyl alcohol paint of quantity of 26-mL in one step was applied; ChloraPrep, Medtronic.
- Skin antiseptic preparation B (PI Group): Povidone-Iodine aqueous scrub (0.75% available iodine solution) having followed by povidone-iodine aqueous paint (1% available iodine solution) was applied; Covidien, Medtronic.
Following the hospital policy, skin was prepared by the nurse within operating room. Anti-septic preparations were applied as per the manufacturer guidelines. Three minute waiting time was kept between anti-septic agent application and skin incision so that adhesive surgical drapes should stick properly over the dry skin. Cefazolin 1gram IV injection a pre-operative prophylaxis antibiotic was admisntered 1 hour prior operation.
The spinal anesthesia was given to all study participants. The low transverse skin incision was performed. Uterine was closed in two layers through Vicryl 1 and the rectus sheath was also closed continuous suturing of Vicryl 1. The 2/0 monocryl was used for the closure of subcutaneous tissue and finally by subcuticular stiches skin was closed using prolene 2/0. All other procedures related to elective CS and post-operative care was done according to the clinical judgment of surgical team.
Data collection
Data related to demographic (i.e. age) maternal characteristics (i.e. gestational age in weeks, parity, body mass index and previous cesarean section scars), co-morbidities (i.e. hypertension, pre-eclampsia, diabetes mellitus, gestational diabetes mellitus, and renal disease) were noted. Moreover, the operative time (minutes), estimated blood loss (ml), pre-operative hemoglobin (g%), post-operative hemoglobin (g%) and blood transfusionwere recorded.
All eligible participants were followed up for SSI for 30 days pos-operatively. The primary outcome was superficial or deep SSI within 30 days after elective CD as defined by US National Healthcare Safety Network definitions of the Centre of Disease Control and Prevention (CDC) [19,20]. The SSI diagnoses was after ensuring that the CDC criteria were met. Patients having attended the post-operative visit at ≥ 30 days after elective cesarean delivery, with well-healed incision on clinical examination and not mentioned any SSI at this or during post-operative assessment were considered to have no surgical site infection and thus free of primary outcome.
The secondary outcomes assessed were hospital stay, readmission, endometritis and adverse skin reactions (i.e. erythema at operative site, skin irritation and allergic skin reaction). Endometritis was defined as fever post-operatively greater than or equal to 38.4 0C atleast two times within 24 hours after delivery along with tenderness in uterine with persistent offensive lochia [21]. Moreover, additional outcome assessed were other wound complications (i.e. skin separation, seroma, hematoma and cellulitis).
Sample size estimation
We estimated a cesarean related SSI rate as 7.5% with PI and hypothesized. That CA being superior based on existing evidence [19] with assumption of 50% reduction in SSI for CA. Considering a power of 0.80 and alpha of 0.05 for statistically significant difference detection in SSI a minimum of 466 pregnant women indicated for elective CS in each group should be enrolled. To account for the loss of follow-up, the sample size calculated was inflated by 15%. Thus, minimum 536 women were enrolled in each group.
Statistical analysis
The statistical software, SPSS version 22 (SPSS Inc., Chicago, IL) was used for statistical analysis. The data was entered into the software and was checked twice to correct for incorrect entries. Descriptive statistics was performed. Qualitative variables were presented as frequency/ percentage and quantitative variables were presented as Mean ± Standard Deviation (SD). The qualitative variables were compared between the two arms of trial by chi square test. If the assumptions of chi square test were not satisfied Fisher exact test was applied. Test of normality, Kolmogorov–Smirnov test was performed for quantitative variables. Quantitative variables as normally distributed were presented as Mean ± SD. The quantitative variables were compared between the two arms of trial by Independent T Test as the assumptions of normality were satisfied. For inferential statistics purpose, the two tailed p-value < 0.05 was considered significant.
Results
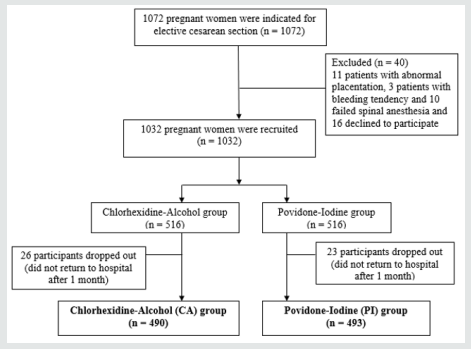
The flow chart of randomization and follow-up of the study participants was shown in Figure 1. After screening there were one thousand and seventy two women indicated for the elective CS. Initially, forty women indicated for elective cesarean delivery were excluded; as eleven presented with abnormal placentation, three had bleeding tendency, ten failed spinal anesthesia and sixteen pregnant women declined to participate. After excluding above mentioned, there were one thousand and thirty two pregnant women were recruited in this open-label, parallel-design prospective randomised controlled trial. As participants were allocated in the ratio of 1:1, thus five hundred and sixteen pregnant women indicated for cesarean delivery were allocated to antisepsis solution CA and similar number for PI. There were twenty six study participants dropped out in Chlorhexidine-Alcohol group, while twenty three participants dropped or did not return for follow-up at one month in PI group. Thus, the CA group constituted the sample of 490, while PI group had the sample of 493. There were few protocol deviations; as 3 patients in PI group received Chlorhexidine-Alcohol as anti-sepsis solution and 9 women in CA group and 11 in PI were recruited and diagnosed late for Chorioamnionitis. However, all randomized patients were analyzed as per protocol with intention to treat analysis (Figure 1).
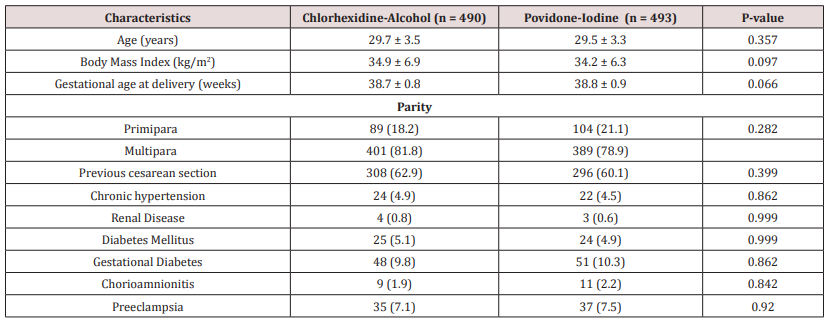
The Table 1 shows the comparison of maternal characteristics between two treatment arms. No significant difference was observed in mean age, BMI, gestational age at delivery between the two groups. Moreover, significant difference was not found when other maternal characteristics i.e. parity, previous cesarean section, co-morbidities, chorioamnionitis and preeclampsia were compared between CA and PI group (Table 1).
Table 1: Comparison of maternal characteristics between Chlorhexidine-Alcohol and Povidone-Iodine group.
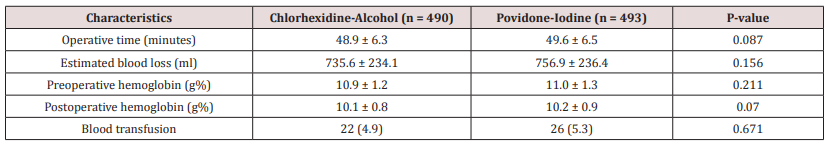
The Table 2 entails details of the comparison of operative characteristics between the two treatment arms. No significant difference was observed in mean operative time, estimated blood loss, pre and post operative hemoglobin and blood transfusion between the two groups. However, as demonstrated in table 2 the mean operative time (48.9 vs. 49.6; p-value = 0.087), estimated blood loss (735.6 vs. 756.9; p-value = 0.156), preoperative hemoglobin (10.9 vs. 11.0; p-value = 0.211), post operative hemoglobin (10.1 vs.10.2; p-value = 0.07) and blood transfusion (4.9% vs. 5.3%; p-value= 0.671) were comparatively lower in CA group in comparison to the PI group (Table 2).
Table 2: Comparison of Operative characteristics between Chlorhexidine-Alcohol and Povidone-Iodine group.
The Table 3 shows details of the comparison of primary and secondary outcomes between the two treatment arms. Significant difference was observed in Surgical Site Infection (SSI) between the CA and PI group (4.3% vs. 7.7%; p-value = 0.014). Moreover, difference was also significant in superficial SSI between the two groups (2.9% vs. 5.9%; p-value = 0.030). However, no difference in significance was observed for deep SSI between the CA and PI group (1.4% vs. 2.4%; p-value = 0.362) (Table 3).
Table 3: Comparison of Primary and Secondary Outcomes between Chlorhexidine-Alcohol and Povidone-Iodine group.
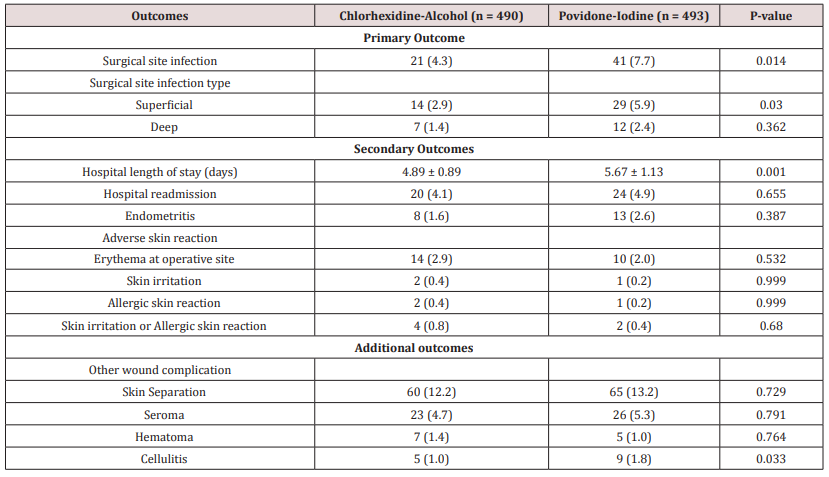
Among the secondary outcomes, significant difference was observed in the mean length of stay (4.89 vs. 5.67; p-value = 0.001) between the CA and PI group. However, no significant difference was observed in hospital readmission and endometritis between the two groups. Though greater proportion of pregnant women indicated for elective CS had readmission and endometritis in PI group. Importantly, though no significant difference was observed in adverse skin reaction but greater proportion of participants allocated to Chlorhexidine-Alcohol group had erythema at operative site (2.9% vs. 2%; p-value = 0.532), skin irritation (0.4% vs. 0.2%; p-value = 0.999), allergic skin reaction (0.4% vs. 0.2%; p-value = 0.999) and skin irritation/ allergic skin reaction (0.8% vs. 0.4%; p-value = 0.680) as compared to Povidone-Iodine group.
Finally, significant difference was not observed in other wound complications (i.e. skin preparation, seroma and hematoma) between the two groups. Though, lesser proportion of participants allocated to CA group had cellulitis (1% vs. 1.8%; p-value = 0.033) as compared to PI group and statistically significant difference was observed.
Discussion
Selection of an efficacious antiseptic skin solution prior surgery is vital to prevent and reduce the incidence of SSI. The present RCT conducted highlighted that Chlorhexidine-Alcohol (CA) was superior to Povidone-Iodine (PI) in decreasing SSI among pregnant women indicated for elective CS. In the two treatment groups the maternal and operative characteristics were comparable, however significant difference SSI, hospital stay and wound complication (cellulitis) was observed between the two groups.
Lower proportion of SSI (4.3%) was observed in the treatment arm of CA as compared to PI group with SSI as 7.7%. Though difference was significant in superficial SSI between the two groups (2.9% vs. 5.9%; p-value = 0.030) but significant difference was not observed for deep SSI between the two groups (1.4% vs. 2.4%; p-value = 0.362). A recent meta-analysis that included six RCTs compared the use of CA with PI as anti-sepsis agents for skin preparation pre-operatively to reduce SSI, reported CA was associated significantly with fewer SSIs as risk ratio was 0.60 with 95% CI in the range of 0.45-0.79 [10]. An RCT reported that that preoperative skin antisepsis CA with Iodine-Alcohol for the prevention of SSI after CD reported that SSI was diagnosed in 23 patients (4.0%) in the CA group and in 42 (7.3%) in the Iodine-Alcohol group with p-value was significant as 0.02 [18]. Moreover, the similar study reported that the rate of superficial SSI was 3.0% in the CA group and 4.9% in the Iodine-Alcohol group (p-value = 0.10) while the rate of deep infection was 1.0% and 2.4%, respectively (p-value = 0.07) [18]. A prospective observational study that compared the incidence of SSI between the CA and PI group among pregnant women indicated for elective CS reported that SSI was 3.7% in the CA group compared with 4.6% in the PI group, with odds ratio as 0.78 and difference was not statistically significant as p-value was 0.35 thus demonstrating that both antiseptic agents were suitable for preparing skin prior elective CS [22]. Moreover, 0.56% patients in the CA group and 0.7% in the PI group developed endometritis (p-value =.74) [22]. Another RCT reported no significant difference in SSI in two treatment arms as incidence was 4.6% vs. 4.5% for PI and CA groups. Moreover, no significant difference was also observed in types of SSI (superficial and deep) between the treatment arms [17]. The RCT comparing the prevalence of bacterial culture positivity at the CD incision site among patients pre-operatively application of Chlorhexidine-Gluconate compared to PI reported that at 18 hour, women in the PI group were seven fold more at risk to women in the Chlorhexidine-Gluconate group for culture positivity (48.5% Vs. 11.1%; with p-value = 0.0023) [23].
However, a recent RCT conducted to compare CA with PI when used as skin antisepsis preparation prior CD reported no difference in significance in SSI both superficial and deep between the two groups [24]. The RCT highlighted that at 30 days follow-up SSI incidence was identified in 6.3% of the CA group and 7.0% in the PI group while incidence of superficial SSI was 4.6% against 5.5%, deep SSI as 0% versus 0.4%; and endometritis as 1.7% versus 1.1% in CA versus PI group respectively [24]. A Cochrane systemic review that included 6 trials and 3607 women reported that Chlorhexidine-gluconate before CS, when compared with PI, made little difference to the incidence of SSI with relative risk as 0.80 and 95% CI as 0.62 to 1.02, while little or no difference to the incidence of endometritis with relative risk as 1.01 was identified [25]. The result of a recent meta-analysis published in 2019 included four randomized controlled trials comparing CA with PI skin preparation solutions for women undergoing CD reported that risk of SSI was significantly reduced around 28% with CA, while superficial or deep SSI alone did not show difference statistically significant [26].
It is difficult to determine the reasons for difference in incidences of SSI following elective CS in the above discussed findings. However, it could be argumented that the difference in results of randomized controlled trial related to cesarean anti-sepsis solutions may be due to institutional perioperative practices, difference in surgical drapes used, adherence level to manufacturer recommendations and extent of anti-sepsis solutions being washed post-surgery may have potentially impacted the efficacy ofthese products. In developing countries and underdeveloped healthcare settings the incidence of SSI following elective CS could be high due to low adherence with the infection control practices. Importantly, most of the researches reported in the literature had been from developed countries i.e. US and others where more efficient infection control measures are adopted. The findings from developing and under developed countries could yield more interesting findings with respect to comparison of SSI incidence with two anti-septic solutions compared in the current RCT.
In the current RCT significant difference in the mean hospital stay (4.89 vs. 5.67; p-value = 0.001) was observed between the CA and PI group. Another RCT reported no significant difference in median length of stay between the two groups as Chlorhexidine-alcohol with Iodine-Alcohol [18]. The Cochrane review reported the mean reduction of 0.10 days with Chlorhexidine gluconate being used pre-operatively to prepare skin for elective CS, however the difference was not significant statistically [25]. Length of stay is of significant importance. The hospital stay could be increased in cases with infections and thereby increasing the cost of care and placing more economic burden on patients and utilization of the healthcare resources which are already scarce. A study had reported that the length of hospitilisation was increased by four days among women diagnosed with SSI with most common organism responsible was Staphylococcus aureus [27]. Moreover, the similar study also reported that among one fourth of these women required readmission [27]. Thus, there has been a very vital linkage between SSI with hospital stay and readmission and thereby the cost of care eventually. This linkage can be broken by using the chlorhexidine-alcohol for pre-operative skin anti-sepsis in elective CS which will reduce the incidence of SSI as highlighted in our study findings.
Significant difference was not observed in other wound complications (i.e. skin preparation, seroma and hematoma) between the two treatment arms but lesser proportion of participants allocated to CA group had cellulitis (1% vs. 1.8%; p-value = 0.033) as compared to PI group and the difference was statistically significant. The findings were similar to a clinical trial that reported no significant difference in other wound complications (i.e. skin preparation, seroma, hematoma and cellulitis) between the two treatment arms as CA with Iodine-Alcohol [18]. In the current study the skin irritation/ allergic skin reaction was 0.8% in CA group and 0.4% in PI group with difference not statistically significant.A study reported no difference in adverse skin reaction between the CA and PI groups with frequency of adverse skin reaction being comparable [18]. A Cochrane systemic review with two trials included reported that Chlorhexidine-gluconate before CS, when compared with PI showed reduction in skin irritation or allergic skin reaction with relative risk as 0.60 [25]. A retrospective analysis had reported that use of CA post CD led to the reduction in cellulitis, seroma as well as hematoma, thereby indicating the use of CA in unscheduled CD where the risk of complications are significantly higher [28].
This RCT concluded thatchlorhexidine-alcohol for pre-operative skin anti-sepsis in elective CS was related with significantly lower risk of SSI when compared to povidone-iodine anti-sepsis solution thus CA is a more suitable anti-septic agent for skin preparation prior elective CS. This study had certain strengths. The large sample recruited along with reduced number of cases being dropped due to loss to follow-up and the randomization process involved for treatment allocation were the main strengths of the study. However, the current study does have certain limitations. Firstly, being a single centre study reduces the generalizability and external validity of the study findings. Secondly, the RCT lack masking of clinical personals to treatment allocation and those involved with the diagnosis of SSI.
References
- Gibbons L, Belizan JM, Lauer JA, Betran AP, Merialdi M, et al. (2012) Inequities in the use of cesarean section deliveries in the world. Am J Obstet Gynecol 206(4): 331.e1-331.e19.
- Curtin SC, Gregory KD, Korst LM, Uddin SF (2015) Maternal morbidity for vaginal and cesarean deliveries, according to previous cesarean history: New data from the birth certificate, 2013. Natl Vital Stat Rep 64(4): 1-12.
- Vogel JP, Betrán AP, Vindevoghel N, Souza JP, Torloni MR, et al. (2015) Use of the robson classification to assess caesarean section trends in 21 countries: A secondary analysis of two WHO multicountry surveys. Lancet Global Health 3(5): e260-e270.
- Li H, Luo S, Trasande L, Hellerstein S, Kang C, et al. (2017) Geographic variations and temporal trends in cesarean delivery rates in China, 2008-2014. JAMA 317(1): 69-76.
- Olsen MA, Butler AM, Willers DM, Devkota P, Gross GA, et al. (2008) Risk factors for surgical site infection after low transverse cesarean section. Infect Control Hosp Epidemiol 29(6): 477-84.
- Schneid Kofman N, Sheiner E, Levy A, Holcberg G (2005) Risk factors for wound infection following cesarean deliveries. Int J Gynecol Obstet 90(1): 10-5.
- Opøien HK, Valbø A, Grinde Andersen A, Walberg M (2007) Post-cesarean surgical site infections according to CDC standards: Rates and risk factors. A prospective cohort study. Acta Obstet Gynecol Scand 86(9): 1097-1102.
- Olsen MA, Butler AM, Willers DM, Gross GA, Fraser VJ (2010) Comparison of costs of surgical site infection and endometritis after cesarean delivery using claims and medical record data. Infect Control Hosp Epidemiol 31(8): 872-75.
- Zuarez Easton S, Zafran N, Garmi G, Salim R (2017) Postcesarean wound infection: Prevalence, impact, prevention, and management challenges. Int J Womens Health 9: 81-88.
- Anggrahita T, Wardhana A, Sudjatmiko G (2017) Chlorhexidine-alcohol versus povidone-Iodine as preoperative skin preparation to prevent surgical site infection: A meta-analysis. Med J Indonesia 26(1):54-61.
- Mangram AJ (1999) Guideline for prevention of surgical site infection, 1999. Infect Control Hosp Epidemiol 20(4): 250-278.
- McKenna PJ, Lehr GS, Leist P, Welling RE (1991) Antiseptic effectiveness with fibroblast preservation. Ann Plast Surg 27(3): 265-268.
- Hibbard JS (2005) Analyses comparing the antimicrobial activity and safety of current antiseptic agents: A review. J Infus Nurs 28(3): 194-207.
- Lim K, Kam PCA (2008) Chlorhexidine - Pharmacology and clinical applications. Anaesth Intensive Care 36(4): 502-12.
- Lee I, Agarwal RK, Lee BY, Fishman NO, Umscheid CA (2010) Systematic review and cost analysis comparing use of chlorhexidine with use of iodine for preoperative skin antisepsis to prevent surgical site infection. Infect Control Hosp Epidemiol 31(12): 1219-1229.
- Noorani A, Rabey N, Walsh SR, Davies RJ (2010) Systematic review and meta-analysis of preoperative antisepsis with chlorhexidine versus povidone-iodine in clean-contaminated surgery. Br J Surg 97(11): 1614-1620.
- Ngai IM, Van Arsdale A, Govindappagari S, Judge NE, Neto NK, et al. (2015) Skin preparation for prevention of surgical site infection after cesarean delivery. Obstet Gynecol 126(6): 1251-57.
- Tuuli MG, Liu J, Stout MJ, Martin S, Cahill AG, et al. (2016) A randomized trial comparing skin antiseptic agents at cesarean delivery. New Engl J Med 374(7): 647-655.
- Menderes G, Athar Ali N, Aagaard K, Sangi-Haghpeykar H (2012) Chlorhexidine-alcohol compared with povidone-iodine for surgical-site antisepsis in cesarean deliveries. Obstet Gynecol 120(5): 1037-1044.
- Centers for Disease Control and Prevention. Surgical site infection. Natl Healthc Saf Netw NHSN Patient Saf Compon Man 9: 1-23.
- Asghania M, Mirblouk F, Shakiba M, Faraji R (2011) Preoperative vaginal preparation with povidone-iodine on post-caesarean infectious morbidity. J Obstet Gynaecol 31(5): 400-403.
- Elshamy E, Ali YZA, Khalafallah M, Soliman A (2018) Chlorhexidine–alcohol versus povidone–iodine for skin preparation before elective cesarean section: a prospective observational study. J Matern Fetal Neonatal Med.
- Kunkle CM, Marchan J, Safadi S, Whitman S, Chmait RH (2015) Chlorhexidine gluconate versus povidone iodine at cesarean delivery: A randomized controlled trial. J Matern Fetal Neonatal Med 28(5): 573-577.
- Springel EH, Wang X, Sarfoh VM, Stetzer BP, Weight SA, et al. (2017) A randomized open-label controlled trial of chlorhexidine-alcohol vs povidone-iodine for cesarean antisepsis: the CAPICA trial. Am J Obstet Gynecol 217(4): 463.e1-463.e8.
- Hadiati DR, Hakimi M, Nurdiati DS, da Silva Lopes K, Ota E (2018) Skin preparation for preventing infection following caesarean section. Cochrane Database Syst Rev.
- Tolcher MC, Whitham MD, El Nashar SA, Clark SL (2019) Chlorhexidine-Alcohol Compared with Povidone-Iodine Preoperative Skin Antisepsis for Cesarean Delivery: A Systematic Review and Meta-Analysis. Am J Perinatol 36(2): 118-123.
- Henman K, Gordon CL, Gardiner T, Thorn J, Spain B, et al. (2012) Surgical site infections following caesarean section at Royal Darwin Hospital, Northern Territory. Healthc Infect 17(2): 47-51.
- Temming LA, Raghuraman N, Carter EB, Stout MJ, Rampersad RM, et al. (2017) Impact of evidence-based interventions on wound complications after cesarean delivery. Am J Obstet Gynecol 217(4): 449.e1-449.e9.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...