Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2690-5760
Research Article(ISSN: 2690-5760) 
Combat Medic efast with Novel and Conventional Portable Ultrasound Devices: A Prospective, Randomized, Crossover Trial Volume 3 - Issue 3
CPT Roland F Salazar1*, Jonathan D Monti1, Aaron J Cronin1, Brian J Ahern1, Brett C Gendron2, Michael D Perreault1 and Jason F Naylor1
- 1Madigan Army Medical Center, Joint Base Lewis McChord, Washington, USA
- 2Brooke Army Medical Center, JBSA Fort Sam Houston, Texas, USA
Received:August 05, 2021 Published: August 13, 2021
Corresponding author:Roland F Salazar, 9040 Jackson Avenue, Joint Base Lewis McChord, Washington, USA
DOI: 10.32474/JCCM.2021.03.000166
Abstract
Background: Extended Focused Assessment with Ultrasonography in Trauma (eFAST) reliably identifies non-compressible torso hemorrhage (NCTH), a major cause of battlefield death. Increased portability of ultrasound (US) enables eFAST far forward on the battlefield, and published data demonstrate combat medics can learn and reliably perform US exams. Sonivate has developed an US device with an intuitive graphical user interface (GUI) and novel, finger-worn transducer with built-in linear and phased arrays, the SonicEye®. We evaluated combat medic eFAST performance between the novel SonicEye® and a conventional device made by General Electric, the Vscan Extend™.
Methods: This was a prospective, randomized, crossover trial completed at a single U.S. military installation. Subjects were U.S. Army combat medics with no previous US experience. Subjects performed an eFAST on a live human and a simulation model with both devices after a brief training intervention. Our primary outcome was time in seconds for eFAST completion, limited to 600 seconds. Secondary outcomes included: diagnostic accuracy, technical adequacy using a validated task-specific checklist, and enduser appraisal of device ease-of-use with 5-point Likert items. This study was approved by the local institutional review board.
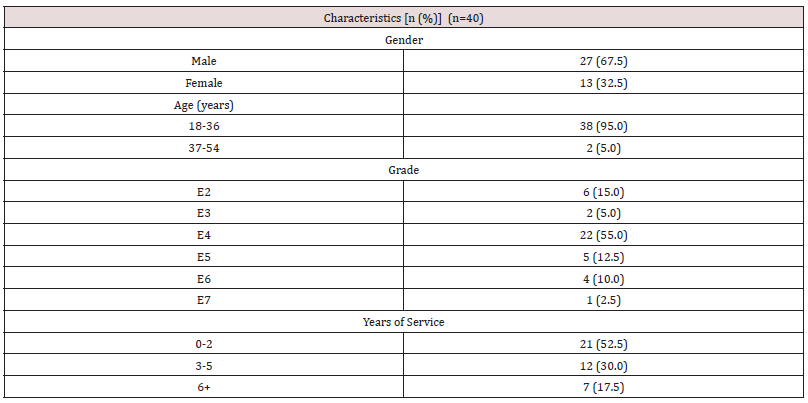
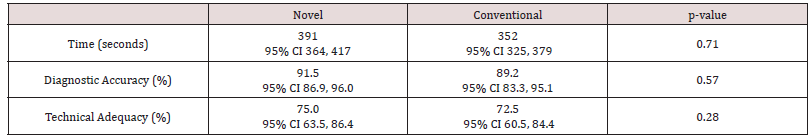
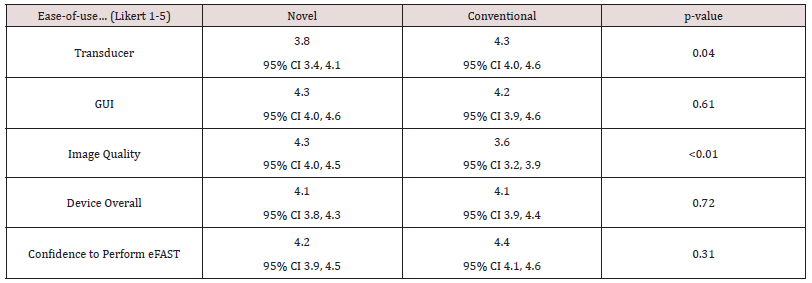
Results: Forty subjects volunteered, most were male (67.5%), less than 36 years old (95.0%), and in the grade E-4 or below (75.0%). Subjects performed a total of 160 eFAST (80 novel, 80 conventional). We found no significant difference in time for eFAST completion between the novel and conventional devices (391 seconds [95% CI 364, 417] versus 352 seconds [95% CI 325, 379]; p = 0.71). We also found there no significant differences between the novel and conventional devices with respect to diagnostic accuracy (91.5% versus 89.2%; p = 0.28) and technical adequacy (75.0% versus 72.5%; p = 0.28). However, we did find that subjects preferred the image quality of the novel device (4.3 versus 3.6; p< 0.01), while favoring the conventional transducer (3.8 versus 4.3; p = 0.04).
Conclusion: Combat medic eFAST performance across devices did not differ with respect to time to completion, diagnostic accuracy, and technical adequacy. Medics with limited US experience performed diagnostically accurate eFAST after a brief training intervention. Future research should assess learning gaps and skill retention in order to guide development of U.S. military US training programs for combat medics..
Keywords: Postoperative Fever; Gynecological Surgery; Surgical Site Infection
Introduction
Background
During the recent conflicts in Afghanistan and Iraq, noncompressible torso hemorrhage (NCTH) was the most common cause of preventable death on the battlefield and required surgical intervention [1-4]. Extended focused assessment with sonography in trauma (eFAST) reliably diagnoses non-retroperitoneal NCTH, and if performed far forward on the battlefield may enable rapid diagnosis and evacuation of casualties with NCTH to surgical facilities [5-9]. Multiple, previous studies demonstrate combat medics can learn and reliably perform diagnostically accurate ultrasound (US) examinations for pulmonary, soft tissue, and musculoskeletal structures [10-13]. A recent study reported combat medics completed timely and diagnostically accurate eFAST after a short training intervention [14]. With recent advances in technology, US devices may now be portable and rugged enough for battlefield utilization [15-17]. Sonivate Medical developed a handheld US device specifically for battlefield medic use. This device couples an intuitive graphical user interface (GUI) and a finger-worn US transducer with built-in linear and high-frequency arrays. Although US device and transducer miniaturization are often considered advantageous, their impact on US exam performance remains unclear, particularly amongst novice sonographers in the austere combat setting. The purpose of this study was to evaluate combat medic eFAST performance with the novel device (Sonivate’s SonicEye®) in comparison to a widely available portable US device (General Electric’s Vscan Extend™).
Goals of this Study
We compare combat medic eFAST completion times between novel and conventional US devices. Secondarily, we evaluate diagnostic accuracy, technical adequacy, and end-user impressions of device ease-of-use, between devices.
Methods
Study oversight and design
The U.S. Army Regional Health Command-Pacific Institutional Review Board approved this prospective, randomized, crossover trial (protocol #218078). All subjects were consented.
Subjects and Materials
We conducted all study activities within the Medical Simulation Training Center at Joint Base Lewis-McChord, Washington. We utilized a classroom for all instruction and a simulated aid station for practical training and testing. The simulated aid station was indoors, temperature-controlled, and with optimal lighting for interpretation of eFAST images displayed on device GUI. We enrolled subjects from locally assigned military units. Our inclusion criteria included medics (military occupational specialty 68W or 18D) on active-duty status 18-54 years old. We excluded subjects who were pregnant or reported previous formal US trainingdefined as a 1-month US training program, an US fellowship, or diagnostic medical sonographer training. We sought medics with minimal US training to avoid potential confounding from device or exam experience. All investigators have emergency medicine residency training. Three investigators (JM, AC, MP) have completed emergency ultrasound fellowship training. All investigators dressed in civilian attire to preclude undue influence from military rank and status.
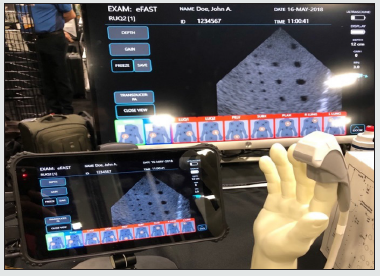
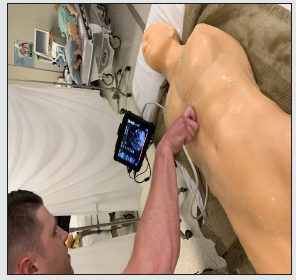
We utilized two portable US devices. The first was a prototype manufactured by Sonivate called the SonicEye® (SonicEye®, Sonivate Medical, Inc., Portland, Oregon, U.S.). The SonicEye® comprises of a finger-worn transducer with built-in linear and highfrequency arrays, thereby eliminating the need to connect multiple, separate transducers during the performance of an eFAST (Figure 1). The SonicEye® is connected by cable to a GUI that employs an intuitive menu system with prompts specifically designed to guide the user through eFAST execution. The second device used was widely available during the time of this study, General Electric’s Vscan Extend™ (Vscan Extend™, General Electric Company, Boston, Massachusetts, U.S.). The Vscan Extend™ also possesses a single transducer with built-in linear and high-frequency arrays; however, these two arrays are situated on opposite ends of the transducer as opposed to one end and it is not designed to be worn on the finger (Figure 2). The Vscan Extend™ transducer is also connected by cable to its GUI that also utilizes a menu-driven system, which includes eFAST but without prompts that guide the sonographer through eFAST execution. We used two models for eFAST training and testing. One was the Blue PhantomTM “FAST Exam Real Time Ultrasound Training Model” (Item Number BP-FAST1800; CAE Blue Phantom, CAE Health Care, Sarasota, Florida, U.S.). Blue PhantomTM models produce realistic sonographic images (Figure 3). We utilized them to generate abnormal US findings; however, Blue PhantomTM models do not replicate lung physiology and cannot produce sonographic lung sliding. After we manipulated the Blue PhantomTM models to create the desired abnormalities, two different investigators performed eFAST on them to validate expected exam findings before subjects tested. The other model was living humans without medical or surgical histories that would produce abnormal eFAST findings. Since these models could not produce abnormal eFAST findings, we did not use them to measure diagnostic accuracy. We incorporated living human models into our study to provide normal eFAST pulmonary findings and evaluate for differences in eFAST performance between living and simulated tissue.
Study protocol
After consenting and enrolling subjects, a single investigator (JM) provided all medics with a standardized, 60-minute lecture on eFAST in a classroom setting. Afterwards, we utilized a random number sequence generator (Random Sequence Generator, Randomness and Integrity Services Ltd., Dublin, Leinster, Ireland) to randomize subjects into one of two groups. Group 1 trained and tested on the novel device first and then repeated the same on the conventional device. Group 2 trained tested on the conventional device before the novel device. By groups, subjects moved to the simulated aid station for device orientation, eFAST exhibition, and eFAST practice exams on a living human model and a Blue PhantomTM model. Investigators demonstrated GUI operation and transducer handling before performing an eFAST on both models for training benefit. Then each subject performed a practice eFAST on both models while being observed by an investigator who provided real-time feedback. After all subjects completed both practice eFAST, subjects returned to the classroom before testing so that investigators could manipulate the Blue PhantomTM models. Then, investigators tested each subject individually on the device they just trained, one eFAST per model. After all subjects tested, we provided an hour-long break for lunch before repeating the same sequence of events, except this time with the other US device. After the second iteration of training and testing, all subjects completed a survey in the classroom before being released from the study.
Outcomes
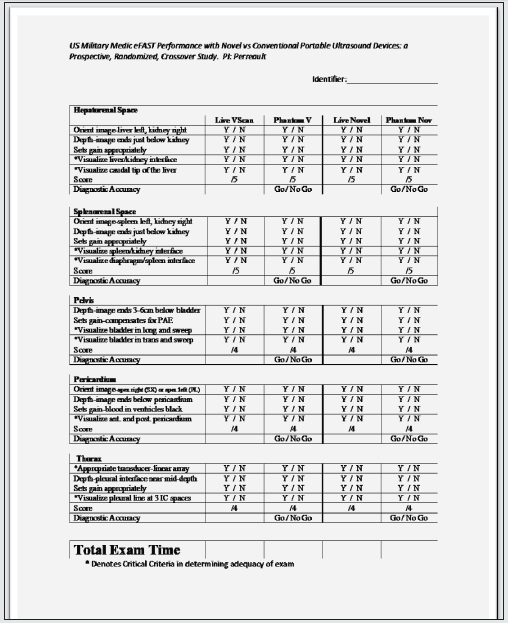
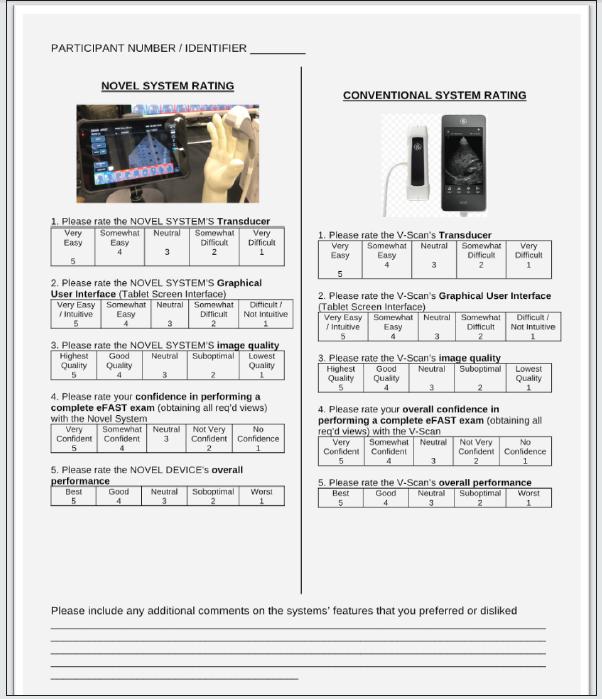
The primary outcome for our study was time to complete an eFAST in seconds. Time started when the subject touched the US transducer and ended when the medic stated the exam was complete or when the maximum allotted time of 600 seconds elapsed. We recorded incomplete exams (i.e., any of the five views were omitted) as the maximum time. We did not include incomplete exams or exams reaching the time limit in our time analysis. We selected 600 seconds as the time limit based on the results of previous research on combat medics performing eFAST [14,18]. Our secondary outcomes included diagnostic accuracy, technical adequacy, and device easeof- use appraisals. For diagnostic accuracy, we only used the Blue PhantomTM models. We required participants to vocalize “normal” or “abnormal” in each of the five eFAST views. Study investigators assessed the participants’ responses as diagnostically correct or incorrect by comparing them to the preset Blue PhantomTM model conditions. We assessed technical adequacy by utilizing a modified version of an image quality checklist validated for FAST (Appendix 1) [14, 19]. An investigator watched a single participant conduct an eFAST and recorded performance of 22 total items. We assessed technical adequacy by total scores; however, in order for an eFAST to be considered technically adequate 9 of the 22 items (indicated by an asterisk; Appendix 1) had to be performed since they are considered critical to maximize the sensitivity of detecting abnormalities [14,19]. We used 5-point Likert items (1=most difficult, 5=easiest) to evaluate medic ease-of-use impressions between devices (Appendix 2). We utilized survey questions validated in previous research on medical eFAST performance [14].
Statistical Analysis
We utilized JMP Pro®, Version 14. (SAS Institute Inc., Cary, NC) to analyze study data. We used a t-test to analyze study data and for crossover effects. We report continuous variables as means with standard deviations and ordinal data as proportions with 95% confidence intervals. We defined statistical significance as p <0.05. We performed pre-study power analysis with a beta of 0.80 and alpha of 0.05 to detect a clinically meaningful difference of 30 seconds between devices, utilizing a time for conventional eFAST completion derived from the results of previous research [14]. Our power analysis determined a sample size of 146 eFAST (73 per group) was required.
Results
From April to May of 2019, 40 combat medics volunteered. All 40 were enrolled, none were excluded, and none withdrew early from the study. Most subjects were male (67.5%), less than 36 years old (95.0%), in the grade E-4 or below (75.0%), and with less than 6 years of military service (82.5%) (Table 1). Subjects performed a total of 160 eFAST exams (80 novel, 80 conventional). Six of 160 (3.8%; # novel3 novel, #3 conventional) eFAST exceeded the time limit and were excluded from comparative time analysis. A total of 794 of 800 (99.25%) possible views were available for secondary outcome analysis. We found eFAST times between the novel and conventional devices were not statistically significant (391 seconds [95% CI 364, 417] versus 352 seconds [95% CI 325, 379]; p = 0.71) (Table 2). Diagnostic accuracy between devices did not differ significantly (91.5% [95% CI 86.9%, 96.0%] versus 89.2% [95% CI 83.3%, 95.1%; p=0.57). And technical adequacy did not differ significantly between devices (75.0% [95% CI 63.5%, 86.4%] versus 72.5 [95% CI 60.5%, 84.4%]; p=0.28). We did, however, find that subjects favored the image quality of the novel device (4.3 [95% CI 4.0, 4.5] versus 3.6 [95% CI 3.2, 3.9]; p<0.01), while preferring the conventional transducer (3.8 [95% CI 3.4, 4.1] versus 4.3 [95% CI 4.0, 4.6]; p=0.04) (Table 3). Analysis of crossover effects demonstrated the treatment effects observed were valid.
Discussion
In this study we evaluated combat medic performance and appraisal of device ease-of-use for eFAST. Subjects favored the novel device’s image quality and the conventional device’s transducer, while endorsing similar assessments for the GUI, device as a whole, and confidence to perform the eFAST. Time for eFAST completion, diagnostic accuracy, and technical adequacy did not differ between the novel and conventional devices. However, we did find that the majority of subjects completed diagnostically accurate eFAST in a timely manner. On average, combat medics completed the eFAST in less than 6.5 minutes with either device. This finding is consistent with the results of a previous, similarly designed study that also incorporated the novel transducer (but not GUI) [14]. The time in our study, however, is almost double that reported for out-of-hospital eFAST performed by physicians (3.5 minutes) [20]. The longer time we observed is likely explained by the difference in subjects between studies. Brun, et al.’s study incorporated emergency medicine physicians with US training and experience, while we enrolled US naïve combat medics who underwent a brief training intervention [20]. This difference in time may be of little clinical significance in the prehospital, combat setting where a positive eFAST may significantly reduce time to surgical intervention by expediting medical evacuation directly from the point-of-injury. Future studies with combat medics performing eFAST in a simulated combat environment may be beneficial. Combat medic eFAST diagnostic accuracy was approximately 90% with both devices, despite technical adequacies of roughly 74%. Both of these findings are consistent with the results of a previous study for combat medic performed eFAST [14]. Previously published studies evaluating combat medic performance of soft tissue and pneumothorax US exams also demonstrated high diagnostic accuracies [10,11,13]. Our findings coupled with published data on combat medic US performance suggest combat medics possess the capacity to learn and perform clinically useful eFAST despite less-than-thorough technical evaluations. This, in turn, indicates sustained eFAST utility despite technical skill degradation [21]. Currently, the U.S. military does not offer US training and/or US skill sustainment for combat medics. Future studies assessing eFAST retention and knowledge gaps may enable development of training and sustainment programs for combat medics.
Novel medical devices may offer design benefits over existing options; however, the end-user’s operating environment, technical expertise, and clinical experience may negate these apparent advantages. We found that medics did not prefer the novel to the conventional device, except for image quality. Despite the unique design of the novel finger-worn, dual array transducer, medics reported that the conventional transducer was easier to use. During training and testing, we observed several medics remove the novel transducer from their right index finger and hold it in their hand instead. This most commonly occurred at the outset of the eFAST when the medic, standing on the patient’s right, began scanning the right upper quadrant of the abdomen because in order to place the high-frequency array in proper position the medic had to internally rotate their right upper extremity and direct the palm of their hand towards the ceiling. This awkward position could have been avoided by donning the transducer on the left index finger or standing on the patient’s left side or at the head of the bed; however, virtually all medics opted to hold the device with their right hand and remain in the same position relative to the patient throughout the eFAST. We suspect this awkward positioning while using the novel transducer donned on the finger partially explains the lack of preference for the novel device. Our findings suggest that either device may be employed by combat medics for eFAST.
Our study has several important limitations. First, the Blue PhantomTM model used to assess diagnostic accuracy does not replicate normal human physiology, such as respiration, diaphragmatic movement of the liver and spleen, and cardiac activity. Of particular importance, all lung examinations were abnormal since lung sliding was not possible and many medics likely recognized this. However, we required all subjects to visualize the pleural lining at three separate intercostal spaces on each side of the thorax and vocalize their findings, which may have reduced any artificial impact on time for eFAST completion. Furthermore, we incorporated living human models to overcome these simulation model shortfalls to measure time for eFAST completion. Second, our subjects performed eFASTs in a simulated aid station that does not mimic the far forward battlefield environment. Therefore, our findings likely do not translate to point-of-injury eFAST performance. However, we chose this setting to eliminate as many potential confounders as possible. Third, we did not require subjects to wear the novel transducer on the finger during its use. This limits the findings of our study as it pertains to its intended use as a finger-worn device; however, the manufacturer explicitly states the novel transducer can be held (not worn) based on user preference. Finally, subjects in our study came from a single U.S. Army installation and comprised entirely of combat medics. Consequently, our findings are not generalizable to the all medics in the US Army and sister services.
Conclusion
Combat medic eFAST performance across devices did not differ with respect to time to completion, diagnostic accuracy, and technical adequacy. Medics with limited US experience performed diagnostically accurate eFAST after a brief training intervention. Future research should assess learning gaps and skill retention in order to guide development of U.S. military US training programs for combat medics.
Disclaimer
The views expressed in this article are those of the authors and do not reflect the official policy or position of the Department of the Army, Department of Defense, or the U.S. Government.
Funding
This study was supported by a grant awarded from the Congressionally Directed Medical Research Program’s Joint Warfighter Medical Research Program to Sonivate Medical.
Conflict of Interest
Dr. Jonathan Monti was employed by the Henry M. Jackson Foundation for the Advancement of Military Medicine, a subcontractor of Sonivate Medical, during execution of this research. All of the remaining authors have no conflicts of interest to report.
References
- Eastridge B J, Robert L Mabry, Peter Seguin, Joyce Cantrell, Terrill Tops, et al (2012) Death on the battlefield (2001-2011): implications for the future of combat casualty care. Journal of trauma and acute care surgery 73(6): S431-S437.
- Morrison J J, Adam Stannard, Todd E Rasmussen, Jan O Jansen, Nigel R M Tai, et al (2013) Injury pattern and mortality of noncompressible torso hemorrhage in UK combat casualties. Journal of Trauma and Acute Care Surgery 75(2): S263-S268.
- Stannard A, Jonathan J Morrison, Daniel J Scott, Rebecca A Ivatury, James D Ross, et al (2013) The epidemiology of noncompressible torso hemorrhage in the wars in Iraq and Afghanistan. Journal of trauma and acute care surgery 74(3): 830-834.
- Eastridge B J, Mark Hardin, Joyce Cantrell, Lynne Oetjen Gerdes, Tamara Zubko, et al (2011) Died of wounds on the battlefield: causation and implications for improving combat casualty care. Journal of Trauma and Acute Care Surgery 71(1): S4-S8.
- McGahan JP, J Richards, M Gillen (2002) The focused abdominal sonography for trauma scan: pearls and pitfalls. Journal of ultrasound in medicine 21(7): 789-800.
- Bhat S R, David A Johnson, Jessica E Pierog, Brita E Zaia, Sarah R Williams, et al (2015) Prehospital Evaluation of Effusion, Pneumothorax, and Standstill (PEEPS): Point-of-care Ultrasound in Emergency Medical Services. West J Emerg Med 16(4): 503-509.
- Gharahbaghian L, Kenton L Anderson, Viveta Lobo, Rwo-Wen Huang, Cori McClure Poffenberger, et al (2017) Point-of-Care Ultrasound in Austere Environments: A Complete Review of Its Utilization, Pitfalls, and Technique for Common Applications in Austere Settings. Emerg Med Clin North Am 35(2): 409-441.
- Savatmongkorngul S, S Wongwaisayawan, R Kaewlai (2017) Focused assessment with sonography for trauma: current perspectives. Open access emergency medicine 9: 57-62.
- Smith I M, David N Naumann, Max E R Marsden, Mark Ballard, Douglas M Bowley (2015) Scanning and War: Utility of FAST and CT in the Assessment of Battlefield Abdominal Trauma. Annals of surgery 262(2): 389-396.
- LaDuke M, Jon Monti, Aaron Cronin, Bart Gillum (2017) Ultrasound Detection of Soft Tissue Abscesses Performed by Non-Physician US Army Medical Providers Naïve to Diagnostic Sonography. Military medicine 182(3): e1825-e1830.
- Driskell D L, J Barton Gillum, Jonathan D Monti, Aaron Croni 2018) Ultrasound evaluation of soft-tissue foreign bodies by US army medics. Journal of Medical Ultrasound 26(3): 147-152.
- Hile D C, Andrew R Morgan, Brooks T Laselle, Jason D Bothwell (2012) Is point-of-care ultrasound accurate and useful in the hands of military medical technicians? A review of the literature. Military medicine 177(8): 983-987.
- Monti J D, B Younggren, R Blankenship (2009) Ultrasound detection of pneumothorax with minimally trained sonographers: a preliminary study. Journal of special operations medicine 9(1): 43-46.
- Ahern B, Jonathan D Monti, Jason F Naylor, Aaron J Cronin, Michael D Perreault (2019) U.S. Army Combat Medic eFAST Performance with a Novel Versus Conventional Transducers: A Randomized, Crossover Trial. Military Medicine 185(1): 19-24.
- Nelson B P, E R Melnick, J Li (2011) Portable ultrasound for remote environments, part I: Feasibility of field deployment. Journal of Emergency Medicine 40(2): 190-197.
- Russell TC, P F Crawford (2013) Ultrasound in the austere environment: a review of the history, indications, and specifications. Military Medicine 178(1): 21-28.
- Gharahbaghian L, Kenton L Anderson, Viveta Lobo, Rwo-Wen Huang, Cori McClure Poffenberger, et al (2017) Point-of-Care Ultrasound in Austere Environments: A Complete Review of Its Utilization, Pitfalls, and Technique for Common Applications in Austere Settings. Emerg Med Clin North Am 35(2): 409-441.
- Shearer JL P K, Gendron BC, Cronin AJ, Jason Naylor (2020) U.S. Army Medic Performance Employing Tele-Ultrasound in the Assessment of Trauma: A Prospective, Observational, Pilot Study. Military Medicine 185(1): 19-24.
- Ziesmann M T, Jason Park, Bertram J Unger, Andrew W Kirkpatrick, Ashley Vergis, et al (2015) Validation of the quality of ultrasound imaging and competence (QUICk) score as an objective assessment tool for the FAST examination. Journal of Trauma and Acute Care Surgery 78(5): 1008-1013.
- Brun P M, Jacques Bessereau, Hichem Chenaitia, Anne Lise Pradel,Cecile Deniel, et al (2014) Stay and play eFAST or scoop and run eFAST? That is the question!. The American journal of emergency medicine 32(2): 166-170.
- Stolz L A, Krithika M Muruganandan, Mark C Bisanzo, Mugisha J Sebikali, Bradley A Dreifuss, et al (2015) Point‐of‐care ultrasound education for non‐physician clinicians in a resource‐limited emergency department. Tropical Medicine & International Health 20(8): 1067-1072.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...