Lupine Publishers Group
Lupine Publishers
Menu
Review Article(ISSN: 2641-6875) 
Corona and Other Virus: Their Useful and Harmful Aspects Volume 2 - Issue 1
Birhanu Gizaw*
- Microbial Biodiversity Directorate, Ethiopian Biodiversity Institute, Ethiopia
Received: June 15, 2020; Published: September 22, 2020
*Corresponding author: Birhanu Gizaw, Microbial Biodiversity Directorate, Ethiopian Biodiversity Institute, P.O. Box 30726, Addis Ababa, Ethiopia
DOI: 10.32474/CTBM.2020.02.000129
Abstract
How the single virus is forceful and shakes the world is eyewitness during this contemporary COVID 19 pandemic time. People primarily think of viruses such as HIV, Ebola, Zika, Influenza, Tobacco mosaic virus or whatever new outbreak like SARS, Corona are all viruses worst and non-beneficial. However, not all viruses are detrimental and influential to human, animal and plant health. In fact, some viruses have beneficial properties for their hosts in a symbiotic relationship and scientific research in many disciplines. Understanding the microbial world is very critical and crucial thing that they are driving force and governing the physical world and biosphere at all. The virus and other microbial life those of bacteria, fungi, prion, viroid, virion are requiring great attention and research to enhance their utilization from majority of useful aspects of microbial genetic resource. The secret behind of every industry, agriculture, health and environment are the application of microbes and their products are too high for human being and environment. Without microbes all life would be cease on earth. However, some microbes are very dangerous like Corona virus, HIV, Ebola, Mycobacterium and others that destroy human life, but majority of microorganisms are too useful to promote development. Through building and strengthening microbial culture collection centers and through strong conservation strategy, it is possible to exploit more from the unseen microbial genetic resource. This paper will review the harmful and use full aspect of virus in respect with health, environment, agriculture and biotechnological application during this Covid19 pandemic time to raise awareness about virus at all.
Introduction
Viruses are obligate intracellular infectious agents that require the host cell machinery to complete their replication cycle and propagate. In fact, the word “virus” is derived from the Latin word for poison. The Russian scientist Dmitry Ivanosky first discovered viruses in 1892 by demonstrating that tobacco mosaic virus was capable of passing through a filter that was known to be impenetrable to bacteria and that this filtrate caused plant disease. Initially, he thought that the plant disease may have been caused by a bacterial toxin, but later work by Martinus Bejerinck, Wendall Stanley and Friedrich Loeffler demonstrated that viruses were filterable infectious agents that replicated within cells. Viruses are found in numerous different shapes and sizes and have different levels of complexity. Structurally, viruses are quite simple; often consisting of only DNA or RNA (the viral genome) surrounded by a simple protein coat having either a helical or icosahedral morphology. Most viruses are too small to be seen with even the best light microscopes, and because of their tiny size and dependence on host cells, their genomes are typically quite small, in some cases consisting of only two genes [1]. No one knows exactly when viruses emerged or from where they came, since viruses do not leave historical footprints such as fossils. Modern viruses are thought to be a mosaic of bits and pieces of nucleic acids picked up from various sources along their respective evolutionary paths. Viruses infect organisms as diverse as bacteria, plants, and animals. They exist in a nether world between a living organism and a nonliving entity. Viruses replicate, but to do not so like living things, they do not metabolize or grow, but are assembled in their mature form. They are entirely dependent on their host cells. The second half of the twentieth century was the golden age of virus discovery, and most of the recognized species of animal, plant and bacterial viruses were discovered during these years. Today there are over 5,000 known viruses that are grouped into different families are known. Viruses have evolved over millions of years to be highly effective at infecting cells and transferring their genetic material to the nucleus, where it is expressed.
Harmful Aspects of Virus
Human disease causing virus
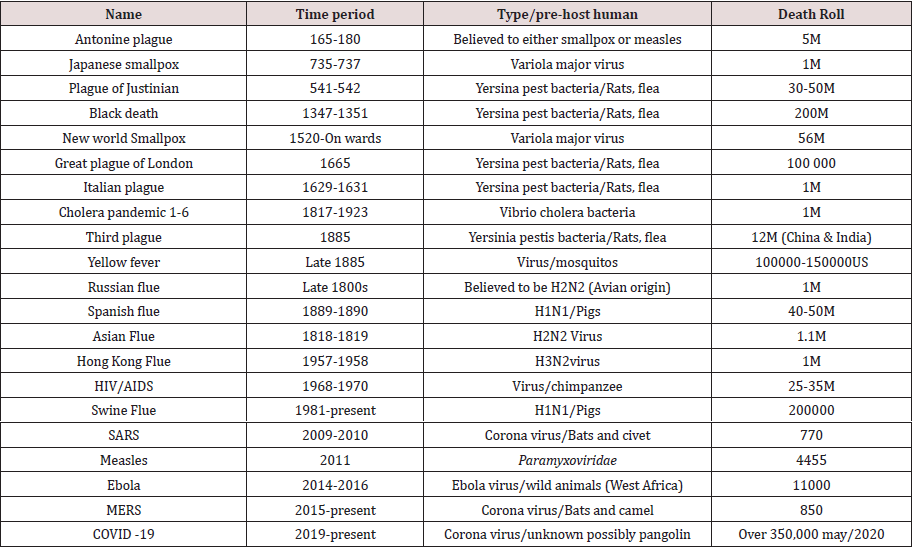
Virus affects all forms of life, from single cellular bacteria and single cellular animals (Protozoa) and to the highest forms of plants and animals including man. Following on from the discovery of tobacco mosaic virus in 1892 and foot-and-mouth disease virus in 1898, the first ‘filterable agent’ to be discovered in humans was yellow fever virus in 1901 [2], at a rate of three or four per year viruses make up over two thirds of all new human pathogens [3]. Disease and illnesses have plagued humanity since the earliest days, our mortal flaw. However, it was not until the marked shift to agrarian communities that the scale and spread of these diseases increased dramatically. Widespread trade created new opportunities for human and animal interactions that sped up such epidemics. Malaria, tuberculosis, leprosy, influenza, smallpox, and others first appeared during these early years. Dengue fever, zika fever and chikungunya fever viruses are recent viral diseases that target poor populations worldwide. In 2016, there were 37 million people living with HIV, 1 million HIV deaths and 1.8 million new infections worldwide. Approximately 70% of HIV-infected people live in Africa [4]. The more civilized humans became with larger cities, more exotic trade routes, and increased contact with different populations of people, animals, and ecosystems, the more likely pandemics would occur. Evola virus, other severe diseases have occurred in Central and West Africa, with 90% high lethality rates aggravating the health situation in these zones and others pandemic still occurring in different part of the world [5,6]. As humans have spread across the world, so have infectious diseases. Even in this modern era, outbreaks are nearly constant, though not every outbreak reaches pandemic level as the Novel Coronavirus (COVID-19). Through different period human beings and animals were diseased by different viral infection and there is certain outbreak of pandemic history (Table 1 & 2) (Figure 1).
Figure 1: Symptom of some human viral disease A. Herpes Simplex Virus, B. Epstien Bar Virus C. Papovavirus, D. Variola Major Virus (A color Atlas of infectious disease R. T. D. Emnd, Ed. Wolf Medical Book London) (1974).
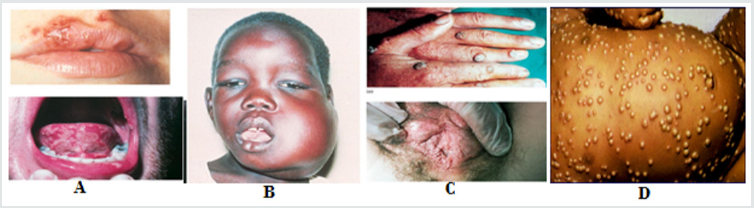
Table 2: Some human disease-causing virus.
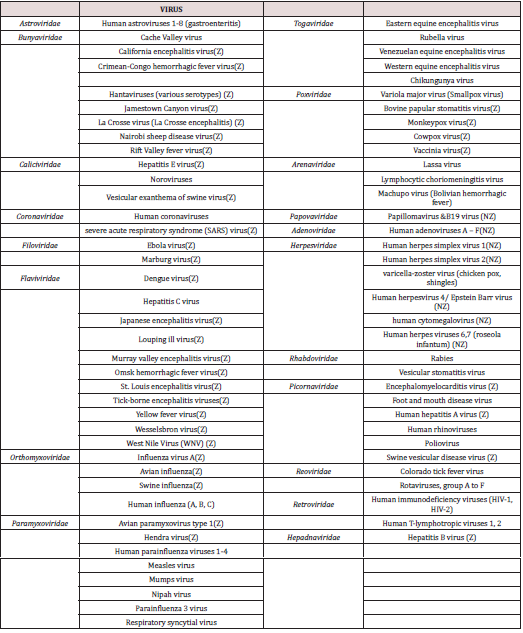
Lists of viruses affect human health and having some zoonotic origin.
Note: Z. (Zoonotic origin), NZ (Non zoonotic).
The corona virus
The name “Coronavirus,” coined in 1968, is derived from the “corona”-like or crown-like morphology observed for these viruses in the electron microscope [7]. Coronaviruses have been described for more than 50 years. The Coronaviridae family is divided into two subfamilies, the Coronaviruses and the Toro viruses, the latter of which cause enteric diseases in cattle and possibly in humans. The Coronaviridae family, along with the Arteviridae and Roniviridae families, forms the Nidovirales order. The Arteviridae family includes swine and equine pathogens; the Roniviridae family is composed of invertebrate viruses [8,9]. Coronaviruses are divided into three genera (I to III), usually referred to as groups and based on serological cross-reactivity [10]. Group I coronaviruses include animal pathogens, such as TGEV of the pig, Porcine Epidemic Diarrhea Virus (PEDV), and Feline Infectious Peritonitis Virus (FIPV), as well as the human coronaviruses HCoV229E and HKU1, which cause respiratory infections. Group II also includes pathogens of veterinary relevance, such as BCoV, porcine hemagglutinating encephalomyelitis virus, and equine coronavirus, as well as human coronaviruses viruses OC43 and NL63, which, like HCoV-229E, also cause respiratory infections. Group II also includes viruses that infect both mice and rats. MHV is often studied as a prototype coronavirus; MHV is a group of highly related strains causing a variety of diseases, such as enteric disease, hepatitis, and respiratory disease, as well as encephalitis and chronic demyelination. Rat sialodacryoadenitis coronavirus also belongs to group II. Group III thus far includes only avian coronaviruses, such as IBV, turkey coronavirus, and pheasant coronavirus [11]. Coronaviruses cause acute and chronic respiratory, enteric, and Central Nervous System (CNS) diseases in many species of animals, including humans [12]. Human coronavirus Previous to the emergence of SARSCoV, there were two prototype human coronaviruses, OC43 and 229E, both etiologic agents of the common cold [12].
The recently identified SARS-CoV, which was shown to cause a severe acute respiratory syndrome was the first example of serious illness in humans caused by a coronavirus [13]. Murine coronavirus there are many strains of murine coronavirus, or MHV, exhibiting different tropisms and levels of virulence. Porcine coronavirus, it is a major cause of viral enteritis and fetal diarrhea in swine; it is most severe in neonates, with mortality resulting in significant economic loss [14]. Avian coronavirus causes a highly contagious disease in chickens; it is spread by aerosol and thus is of considerable economic importance to the poultry industry. The feline coronaviruses are composed of two biotypes. Feline enteric coronavirus (FeCoV), commonly found in multi cat environments in an asymptomatic carrier state, causes seroconversion. FIPV, a less common variant of FeCoV, has the ability to replicate in macrophages, causing a severe and lethal disease [15,16]. Bovine Coronavirus (BCoV) is a ubiquitous virus worldwide as measured by serology. BCoV causes both respiratory and enteric disease, including calf diarrhea, winter dysentery in adults, and respiratory infections in cattle of all ages, including those with shipping fever [15,16].Coronaviruses make up a large family of viruses that can infect birds and mammals, including humans, according to World Health Organization (WHO). These viruses have been responsible for several outbreaks around the world, including the Severe Acute Respiratory Syndrome (SARS) pandemic of 2002-2003 and the Middle East Respiratory Syndrome (MERS) outbreak in South Korea in 2015. Most recently, a novel coronavirus (SARS-CoV-2, also known as COVID-19) triggered an outbreak in China in December 2019, sparking international concern. Coronaviruses belong to the subfamily Coronavirinae in the family Coronaviridae. Different types of human coronaviruses vary in how severe the resulting disease becomes, and how far they can spread. Doctors currently recognize seven types of coronavirus that can infect humans. Common types are 229E (alpha coronavirus), NL63 (alpha coronavirus), OC43 (beta coronavirus), HKU1 (beta coronavirus), Rarer strains that cause more severe complications include MERS-CoV, which causes Middle East Respiratory Syndrome (MERS), and SARS-CoV, the virus responsible for severe acute respiratory syndrome (SARS). In 2019, a dangerous new strain called SARSCoV-2 started circulating, causing the disease COVID19 [17].
Plant disease causing virus
Plants viruses affect plant health & like all other viruses are obligate intracellular Parasites that do not have the molecular machinery to replicate without a host. Bacteria and fungi pathogen reduce crop yields by about 15% and viruses reduce 3% [18]. The first virus to be discovered was Tobacco Mosaic Virus (TMV). Viruses cause an estimated US $60 billion loss in crop yields worldwide each year. Plant viruses are grouped into 73 genera and 49 families. However, these figures relate only to cultivated plants that represent only a tiny fraction of the total number of plant species. Viruses in wild plants have been poorly studied [19]. Plant viruses must use strategies that are usually different from animal viruses because plants do not move, and so plantto- plant transmission usually involves vectors (such as insects). Plant cells are surrounded by solid cell walls; therefore, transport through plasmodesmata is the preferred path for virions to move between plant cells. In 1939 Holmes published a classification list of 129 plant viruses. This was expanded and in 1999 there were 977officially recognized (Table 3). Over 50% of known plant viruses are rod-shaped (flexuous or rigid) (Figure 2 & 3). The length of the particle is normally dependent on the genome, but it is usually between 300-500nm with a diameter of 15-20nm [20]. 75% of plant viruses have genomes that consist of single stranded RNA (ssRNA), Plant viruses have had to evolve special techniques to allow the production of viral proteins by plant cells.
Table 3: Some common pant disease causing virus Taken. From Comparative Plant Virology, by Roger Hull.2009, Elsevier Inc.).
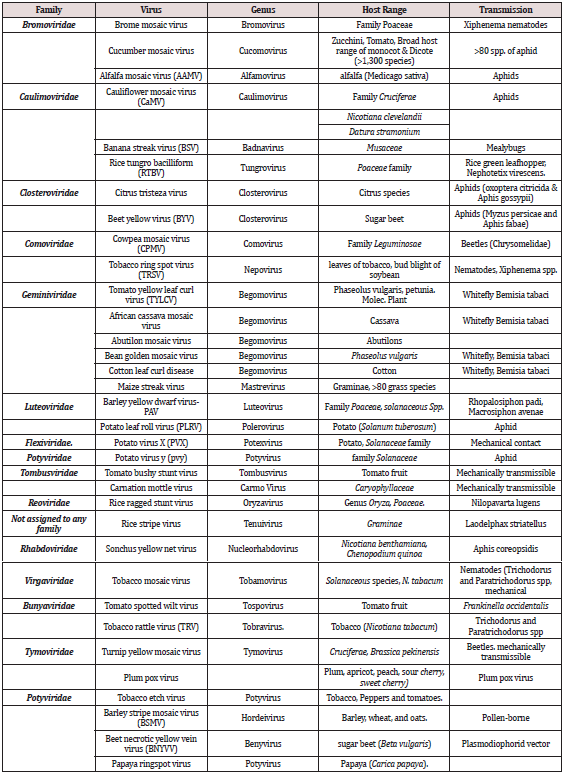
Figure 2: Morphology of some plant virus disease symptom A. BYV cause vein clearing and vein yellowing symptom, B. Tobacco Ring Spot Virus (TRSV). C. Abutilon Mosaic Virus (ABMV), Pavonchus Yellow Netvirus, E. Barley dwarf virus-PAV, F. Rice Tungro Bacilliform Virus (RTBV), G. Rice aged Stunt Virus (RRSV), H. Tomato Spotted Wilt Virus (TSWV). (Photo Taken. From Comparative Plant Virology, by Roger Hull.2009, Elsevier Inc.)

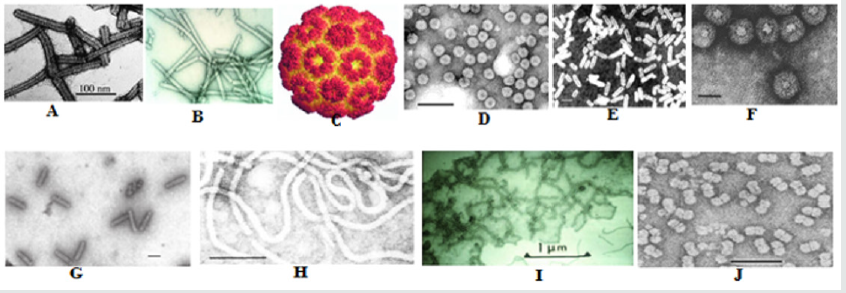
Figure 3: Microscopic image of some plant virus A. Barley Stripe Mosaic Virus, B. Beet Necrotic Yellow Vein Virus, C. Bromo Virus. D. Cucumber Mosaic Virus. E. Alfalfa Mosaic Virus, F. Cauliflower Mosaic Virus, G. Banana Streak Virus, H. Citrus Tristerza Virus I. Beet Yellows Virus, J. Cowpea Mosaic Virus (CPMV). From Comparative Plant Virology, by Roger Hull.2009, Elsevier Inc.)
Emerging zoonotic virus
Zoonotic diseases have potentially serious human health and economic impacts and their current upwards trend is likely to continue. Approximately 75% of viruses and 50% of bacteria known to cause disease in humans are zoonotic and can be transmitted between animals and people [21]. The last 30 years have seen a rise in emerging infectious diseases in humans and of these over 70% are zoonotic [22,23]. Of the 1415 species recorded as pathogenic for humans in 2001, 868 (a staggering 61%) could be characterized as zoonotic [21]. As the vast majority of novel species pathogenic for humans recognized since 2001 are also of a zoonotic nature, including SARS coronavirus and novel H1N1 influenza virus (pandemic influenza of swine origin) [24]. Zoonotic infections are not new. They have always featured among the wide range of human diseases and most, Chikungunya virus, Zika virus, Ebola virus , Hantaan virus, HIV, Hanta virus, Hendra virus, H5N1, Nipah virus SARS, MERS, H1N1, Anthrax, Tuberculosis, Plague, Yellow fever and influenza, have come from domestic animals, poultry and livestock as well as wild animal origin [25]. The next human pandemic is likely to be zoonotic, and wildlife is emerging as the primary source. Many zoonosis from wildlife, including avian influenza and rabies, are well established, while others have only recently emerged or have only recently been linked to wildlife reservoir species. One example of the latter is the Ebola virus, which, after decades of research, was recently linked to cave-dwelling bats in Africa. Similarly, the Severe Acute Respiratory Syndrome (SARS) coronavirus [26,27]. Today over 500 animal viruses are classified as arboviruses, i.e., viruses able to replicate in a blood-feeding arthropod and to infect a vertebrate host whenever the arthropod feeds on that host. Additionally, numerous vertebrate-infecting viruses are transmitted by arthropod vectors but do not replicate in the vector [28].
Viruses in bioweapons and biological warfare
The use of biological agents as war weapons is not a modern era novelty. Although it is not easy to identify a definite time when the use of bioweapons began, ancient evidence reported that in pre- Christian era, around 300 B.C., the Greeks used animal cadavers to contaminate water wells of enemies [29]. Several biological warfare actions carried out during the World War are not sufficiently confirmed in the literature. However, it is frequently reported that the Germans inoculated cattle with Bacillus anthracis and Pseudomonas mallei, responsible to cause severe diseases such as anthrax and glanders, before sending them into enemy states [29- 31]. The event of bioterrorism-related inhalation of anthrax in the United States towards the end of 2001 has brought into stark reality the possibility of biological weapons as a tool of mass destruction and renewed fear of the re-introduction of biological warfare. Bioterrorism attack as “the deliberate release of viruses, bacteria or other germs (agents) used to cause illness or death in people, animals, or plants” [32]. In the last few decades, the development of innovative biotechnology techniques has provided the knowledge to create more aggressive bioweapons. These new organisms cause great concern, because they can produce devastating and completely unexpected effects, of the same level or even higher than the most dangerous wild type biological agents. During the past century, more than500 million people died of infectious diseases. Several tens of thousands of these deaths were due to the deliberate release of pathogens or toxins. Haemorrhagic fever causing like Marburg virus (V), Ebola virus (V), Arenaviruses (V), Lassa virus, Alphaviruses (V), Variola major (V) were considered as bioweapon agent. Nipah virus, a newly emerging deadly paramyxovirus isolated during a large outbreak of viral encephalitis in Malaysia, has many of the physical attributes to serve as a potential agent of bioterrorism. Pathogens that are made more pathogenic by genetic engineering, including hantavirus, Nipah virus, tick-borne encephalitis and hemorrhagic fever viruses, yellow fever virus [33].
Use Full Aspect of Virus
Viruses in nanotechnology
Most virus capsids measure between 20-500nm in diameter. Because of their nanoscale size, symmetrical structural organization, load capacity, controllable self-assembly, and ease of modification. Viruses are essentially naturally occurring nanomaterials capable of self-assembly with a high degree of precision and nanoparticles have been subject to the nano science and nano engineering disciplines [34]. Many different viruses have been studied for various applications in nanotechnology. Mammalian viruses are being developed as vectors for gene delivery, and bacteriophages and plant viruses have been used in drug delivery and imaging applications as well as in vaccines and immunotherapy intervention [23].Viral capsid nanoparticle hybrid structures, which combine the bio-activities of virus capsids with the functions of nanoparticles, are a new class of bio Nano materials that have many potential applications as therapeutic and diagnostic vectors, imaging agents, and advanced nanomaterial synthesis reactors. Plant virus nanoparticles are non-infectious to mammalian cells [35]. Plant viruses have a size particularly suitable for nanoscale applications and can offer several advantages. In fact, they are structurally uniform, robust, biodegradable and easy to produce [34]. Moreover, many are the examples regarding functionalization of plant virus-based nanoparticles by means of modification of their external surface and by loading cargo molecules into their internal cavity. This plasticity in terms of nanoparticles engineering is the ground on which multi valency, payload containment and targeted delivery can be fully exploited [36]. The most popular vectors are derived from Tobacco Mosaic Virus (TMV), Potato Virus X (PVX) and Cowpea Mosaic Virus (CPMV) [37]. More recently, plant viruses have been utilized as nanoparticles or Virus-Like Particles (VLP) to transport drugs and active molecules into cancer cells, thus offering a new and potent arsenal for the fight against cancer.
Viral vectors for vaccine applications
Viral vectors are deemed as an attractive alternative to the traditional platforms to deliver vaccine antigens as well as to specifically target and kill tumor cells. With potential targets ranging from cancers to a vast number of infectious diseases. Adenovirus has become one of the most exploited vectors for vaccine development. Their ability to infect broad range of hosts and to induce high levels of transgene expression without the potential of viral genes being integrated into the host genome. Adenoviral vectors can inherently stimulate innate immune responses via Toll-like receptor-dependent and Toll like receptorindependent pathways. The use of recombinant adenoviral vectors has been explored in the area of HIV vaccine development as well. Alpha viral vaccine vectors have been explored in myriad of vaccine applications for cancers, HIV, and human parainfluenza virus [38- 40]. Alphavirus vectors can create a proper environment for the cross-priming of vaccine antigen by inducing apoptosis in some cells [41]. Due to the success of vaccinia virus-vectored vaccine in the eradication of smallpox; poxviruses have been widely evaluated for their use in vaccine applications.
Viruses as bio control agents
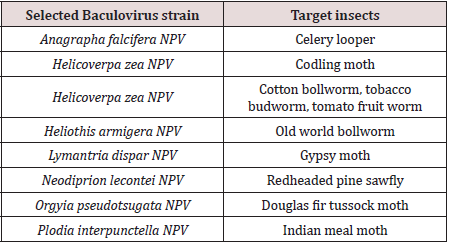
Viruses have been used to control insect pests in the classical way of biological control by inoculative release of a virus into the population or by inundate application as a biological insecticide [42]. Many baculoviruses have been reported from insect pests of economic importance in agriculture and forests [43]. In field trials at least 40 different viruses have given control efficacy comparable to that of chemical insecticides [44,45]. Over a 1000 baculovirus insect associations are known [43]. The majority (800) of these involves Polyhedrosis Viruses (NPVs). Among the species infected by NPVs many important lepidopteran pests of cultivated crops are found. Baculoviruses predominantly infect the immature (larval) forms of moth species but they also infect sawflies, mosquitoes, and shrimps [46-49]. The mechanism of action of Baculoviruses against wide arrays of insect pest is due to their ability to multiply and replicate rapidly in different developmental forms of insects (larva or adult form) and cause multiple cytotoxic effect in their hosts [50-52]. Most lepidopteran larvae show increased mobility and food consumption as they age, even though their susceptibility to infection to some pathogens, such as baculoviruses, seems to decline considerably as larvae develop [53]. Recently, Baculoviruses have been subdivided into four genera these include the Alpha Baculoviruses (the Lepidopteran specific Nucleopolyhedroviruses), Beta Baculoviruses (the Lepidopteran specific Granuloviruses), Gamma Baculoviruses (the Hymnopteran specific Nucleo polyhedroviruses), and Delta Baculoviruses (the Dipteran specific Nucleo polyhedroviruses) [50,54,55]. The use of Baculoviruses as viral biopesticides presents a relatively cheap, practicable, environmentally friendly and potent tool for biocontrol of insect pests of agricultural importance of farm crops and stored products. The viruses have been known to be capable of entering mammalian cells, but they do not replicate in vertebrate animal tissues [50,56]. Several Nuclear Polyhedrosis Viruses (NPVs) are potential candidates as biological control agents for beet armyworm. From Californian populations of beet armyworm, a multiply-enveloped NPV specific for S. exigua (SeMNPV) has been collected and described [57-59]. Another baculovirus, Autographa californica MNPV (AcMNPV) has a broad host range among noctuid pests of agricultural crops and is also infectious for beet armyworm [60,61] (Table 4).
Table 4: Bio control agent virus. N.B. Strain names follow the naming conventions for Baculoviruses, where each virus is named for the target insect followed by general type (genus) of the infecting Baculoviruses; Nucleopolyhedroviruses (NPV) or Granuloviruses (GV).
MYXV and RHDV Virus have been released in Australia to control the abundance and spread of the European rabbit (Oryctolagus cuniculus). The release of MYXV, starting in the 1950s , and of RHDV which began the mid-1990s are the most successful accomplishments in the biological control of vertebrate populations, and together have reduced the size of the invasive rabbit population by approximately 85% [60]. Virus being successfully used as a vertebrate biocontrol was the deliberate deployment of feline parvovirus (also known as feline panleukopenia virus) against cats on Marion Island [61].This small and remote island in the Southern Indian Ocean had an estimated feral cat population of over 2,000 in 1975, and which posed a major threat to the island’s bird population, with 450,000 killed each year. Following the release of parvovirus, the cat population declined to approximately 600 animals within a few years, rather which were then successfully eradicated using traps and hunting [62].
Viruses in bacteriophage therapy
A bacteriophage also known informally as a phage is a virus that infects and replicates within bacteria and archaea. The term was derived from “bacteria” and the Greek (phagein), meaning “to devour”. Bacteriophages are composed of proteins that encapsulate a DNA or RNA genome and may have structures that are either simple or elaborate. Their genomes may encode as few as four genes (e.g. MS2) and as many as hundreds of genes. Phages replicate within the bacterium following the injection of their genome into its cytoplasm. Bacteriophages are among the most common and diverse entities in the biosphere [63]. Bacteriophages are ubiquitous viruses, found wherever bacteria exist. It is estimated there are more than 1031 bacteriophages on the planet, more than every other organism on Earth, including bacteria, combined [64]. Bacteriophages are the most abundant biological entity on this planet, and it is estimated that phages kill half of all bacteria each day and influence most of earth’s biogeochemistry through their bacterial host [65]. Since their discovery in 1915 and 1917 by Herelle and Twort [65,66]. These are highly specific viruses that can target, infect, and (if correctly selected) destroy pathogenic bacteria. Bacteriophages are believed to be the most numerous types of viruses accounting for the majority of the viruses present on Earth. These are basic tools in molecular biology. They have been researched for their use in therapy. Phages have been used since the late 19th century as an alternative to antibiotics in the former Soviet Union and Central Europe, as well as in France [67,68]. They are seen as a possible therapy against multi-drug-resistant strains of many bacteria [69]. Phages of in oviridae have been shown to complicate biofilms involved in pneumonia and cystic fibrosis and to shelter the bacteria from drugs meant to eradicate disease, thus promoting persistent infection.
Viruses in cancer prevention and control
Cancers are the second most common cause of death
worldwide. A number of different viruses have been adapted as
vectors for cancer gene therapy. Retroviruses have been employed
predominantly for gene transfer ex vivo, which involves a timeconsuming
and complex procedure in which the target cells are
removed from the patient, genetically modified, and then re
implanted into the same recipient, whereas the attributes of other
viruses favor their use for in vivo gene transfer. Several mammalian
and avian C-type retroviruses have been exploited as vectors
for cancer gene therapy, including Moloney Murine Leukemia
Virus (MuLV), gibbon ape leukemia virus, spleen necrosis virus,
and avian sarcoma and leukosis viruses. The retroviral DNA then
integrates into the host DNA to form the provirus. Oncolytic viruses
can replicate specifically in tumor cells and induce toxic effects
leading to cell lysis and apoptosis. Recombinant viruses can be
used to genetically modify cells, triggering the expression of tumorspecific
antigens. These viruses can also be used to directly infect
antigen-presenting cells to boost anti-cancer immune responses
[70]. Several viruses have been used in attempts to develop optimal strategies for transgene delivery for a range of cancers.
These include, retroviruses, Lentiviruses (including HIV-1) [71].
Adenoviruses [72]. Adeno-associated viruses [73] Poxviruses [74]
and Herpes viruses [75] Poxviruses, which are able to induce both
CD4+ and CD8+ mediated immune response against heterologous
antigens, are the most widely used viruses for anti-tumor vaccine
production.
To enhance the efficacy of oncolytic virotherapy, several
combination therapies with GM-CSF, chemotherapy or radiation
therapy have been studied [75]. Another breakthrough in virusmediated
cancer therapy is the use of oncolytic viruses, which have
a direct cytotoxic effect on tumor cells, and additionally stimulate
the immune response by releasing tumor-specific antigens and
inducing local inflammation. Oncolytic virus therapy has recently
been recognized as a promising new therapeutic approach for
cancer treatment. Oncolytic viruses are defined as genetically
engineered or naturally occurring viruses that selectively replicate
in and kill cancer cells without harming the normal tissues. T-Vec
(Talimogene laherparepvec), a second-generation oncolytic herpes
simplex virus type 1 (HSV-1) armed with GM-CSF, was recently
approved as the first oncolytic virus drug in the USA and Europe
[76].
Viral Mutualistic Symbioses
Viruses of endoparasitoid wasps
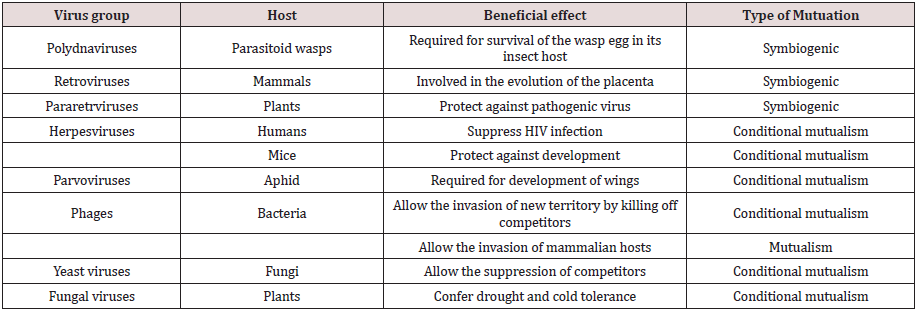
The polydnaviruses (poly-DNA) which consist many DNA segments that are the best studied mutualistic viruses. There are thousands of these viruses; an estimated 30,000 species of endoparasitoid braconid and ichneumonid wasps probably have their own mutualistic viral species (called bracoviruses and ichnoviruses, respectively. The relationship between polydnaviruses, wasps and caterpillars. Many parasitoid wasps lay their eggs inside a living insect larva. When a female wasp deposits her eggs inside a lepitdopteran caterpillar, she also deposits her symbiogenic polydnavirus virions, which only express wasp genes. These genes are expressed in the caterpillar, where they prevent the encapsulation process that would otherwise wall off and kill the wasp egg. The polydnaviruses of parasitoid wasps, for example are required for the survival of their hosts [77]. Another A three-way mutualistic symbiosis seen in the panic grass Dichanthelium lanuginosum is found in geothermal soils where it can grow at soil temperatures >50 °C. The plant requires a fungal endophyte, Curvularia protuberata, to survive at this temperature. In turn, the fungus requires a virus, Curvularia Thermal Tolerance Virus (CThTV), to confer this thermotolerance effect [78]. There are many symbiotic virus with different host which has different vital role for survival (Table 5).
Virus induced gene silencing (VIGS)
Gene Silencing (GS) is a molecular process involved in the down regulation of specific genes, and probably evolved as a genetic defense system against viruses and invading nucleic acids [79-82]. It plays an important role at the cellular level, affecting differentiation, gene regulation [83] and protection against viruses and transposons [83]. This powerful biotechnological tool has provided plant breeders and researchers with great opportunity to accelerate breeding programs and developmental studies in woody plants. Virus induced gene silencing is a technique designed to suppress gene expression and study gene function in plants [84] VIGS used silencing of endogenous plant genes initiated by recombinant viral vectors [85].The model includes viral dependent initiation and viral independent maintenance of VIGS. The approach consists of cloning and inserting plant endogenous gene sequences in recombinant viral vectors, which are then inoculated in plants, triggering PTGS/RNAi. This ends up silencing plant genes with homologous sequences to those contained in the viral vectors [86]. VIGS is a powerful tool due to its fast initiation of silencing in intact wild-type or transgenic plants [84].
Virus-Plant Interaction Role
Virus-plant interactions are obligate, symbiotic interactions that exist along a spectrum from parasitism to commensalism to mutualism. In a mutualistic relationship net effects are positive with enhanced survival and/or reproduction for both the plant and virus, thus as with all mutualisms the benefits outweigh the costs of the relationship. Cucumber Mosaic Virus (CMV) benefits Arabidopsis thaliana by increasing seed production in comparison to plants without virus though this effect depends upon environmental conditions. Cucumber mosaic virus alters volatiles in Solanum lycopersicum making it more attractive to pollinators [87].
Virus-based pharmaceutical production in plants
Viral expression vectors based on plant viruses have greatly enhanced plant-made therapeutic production and have been exploited to produce a variety of proteins of industrial, pharmaceutical and agribusiness interest. Plant viruses have been engineered to either express subunit vaccines or act as epitope presentation systems. Many plant viruses have been used for these purposes [88]. Among them, the most popular vectors are derived from Tobacco mosaic virus (TMV), Potato virus X (PVX) and Cowpea mosaic virus (CPMV) [89]. Several viral expression vectors based on viruses belonging to some families, including Potexviruses, Comoviruses, Geminiviruses and Tobamoviruses. Tobacco Mosaic Virus (TMV) was the first virus engineered as a deconstructed vector system because of its high number of Capsid Protein (CP) copies (2130 copies) and the relatively simple organization of its genome. TMV has been used successfully for the production of several proteins of interest. The expression of vectors has been improved by genetic engineering with modifications of transcript splicing sites, codon usage patterns and insertion of introns into coding sequences [90]. Human papillomavirus (HPV) E7 protein has been generated in plants with TMV vectors [91]. Influenza M2e epitope has also been produced in plants thanks to TMV [92].
Conclusion
Virus and other microbial life have both useful and harmful aspects, since they are tiny and unseen, their role and value are not magnified rather they are expected as disease causing and problematic. In fact, some are too dangerous in history of pandemic human history in affecting human life, plant and animals. The virus like Corena, SARS, MERS, Variola major, H2N2 Virus, Evola, HIV are some commonly known killer virus. But also, useful virus in vaccine development, bio control agent etc. In general, the microbial life occupies greatest diversity on earth and majority are consisting of several useful application for human and environment. Through building and strengthening microbial culture collection centers and through strong conservation strategy, it is possible to exploit more from the unseen microbial genetic resource.
References
- Faurez F, Dory D, Grasland B, Jestin A (2009) Replication of porcine circoviruses. Virology Journal 6: 60.
- Levine AJ, Enquist LW (2007) History of virology. In: BN Fields, DM Knipe, PM Howley (Eds.), 5th (edn.), Fields virology, Pp: 565-604.
- Woolhouse MEJ, Gaunt E (2007) Ecological origins of novel human pathogens. Crit. Rev. Microbiol 33: 1-12.
- UNAIDS DATA (2017).
- Feldmann H, Jones S, Klenk HD, Schnittler HJ (2003) Timeline: Ebola virus: From discovery to vaccine. Nat Rev Immunol 3: 677-685.
- WHO (2019) Ebola virus disease? World Health Organization.
- Tyrrel DAJ, Almedia JD, Berry DM, Cunningham CH, Hamre D, et al. (1968) Coronavirus. Nature 220: 650.
- Cowley JA, Dimmock CM, Spann KM, Walker P J (2000) Gill-associated virus of Penaeus monodon prawns: an invertebrate virus with ORF1a and ORF1b genes related to arteri- and coronaviruses. J Gen Virol 81(6): 1473-1484.
- Enjuanes LDC, Holmes K, Lai MMC, Laude H, Masters P Rottier P, et al. (2000) Coronaviridae, Virus taxonomy. Classification and nomenclature of viruses. Wickner (Edt.), Academic Press, San Diego, California Pp: 835-849.
- McIntosh K (1974) Coronaviruses: A comparative review. Curr Top Microbiol Immunol 63: 85-129.
- Cavanagh DK, Mawditt B, Welchman D, Britton PR, Gough RE (2002) Coronaviruses from pheasants (Phasianus colchicus) are genetically closely related to coronaviruses of domestic fowl (infectious bronchitisvirus) and turkeys. Avian Pathol 31(1): 81-93.
- Roger Hull (2009) Comparative Plant Virology. Second edition. Academic Press is an imprint of Elsevier Pp: 303-363.
- Rota PA, Oberste MS, Monroe SS, Nix WA, Campagnoli Icenogle RJ, et al. (2003) Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science 300(5624): 1394-1399.
- Enjuanes LC, Smerdou J, Castilla IM, Anton JM, Torres I, et al. (1995) Development of protection against coronavirus induced diseases. Adv Exp Med Biol 380: 197-211.
- Saif LJ (2004) Animal coronaviruses: What can they teach us about the severe acute respiratory syndrome? Rev. Sci. Technol 23(2): 643-660.
- Saif LJ (2004) Animal coronavirus vaccines: lessons for SARS. Dev. Biol 119: 129-140.
- Shrikrushna Subhash Unhale, Quazi Bilal Ansar, Shubham Sanap, Suraj Thakhre Shreya Wadatkar, Rohit Bairagi, et al. (2020) A review on corona virus (COVID-19). World Journal of Pharmaceutical 6(4): 109-115.
- Oerke EC, Dehne HW (2004) Safeguarding production: Losses in major crops and the role of crop protection. Crop Prot 23(4): 275-285.
- Roossinck MJ (2011) The good viruses: viral mutualistic symbioses. Nature Reviews Microbiology 9: 99-108 Tech Vert. Evidence of First Virus That Moves from Plants to Humans. Tech Vert.
- Gray SM, Banerjee N (1999) Mechanisms of Arthropod Transmission of Plant and Animal Viruses. Microbiology and Molecular Biology Reviews 63(1): 128-148.
- Taylor LH, Latham SM, Wool house ME (2001) Risk factors for human disease emergence. Philos Trans R Soc Lond B Biol Sci 356(1411): 983-989.
- Jones KE, Patel NG, Levy MA, Storeygard Niagro FD, Forsthoefel AN, et al. (1998) Beak and feather disease virus and porcine circovirus genomes: intermediates between the geminiviruses and plant circoviruses. Archives of Virology 143: 1723-1744.
- Woolhouse ME, Haydon DT, Antia R (2005) Emerging pathogens: the epidemiology and evolution of species jumps. Trends Ecol. Evol 20(5): 238-244.
- Cutler SJ, Fooks AR, Van der Poel WH (2010) Public health threat of new, reemerging, and neglected zoonoses in the industrialized world. Emerg Infect Dis 16(1): 1-7.
- Benfield E, Treat J (2014) As Ebola death toll rises, remembering history’s worst epidemics. National Geographic, Deadly companions: How microbes shaped our history. Oxford University Press.
- Leroy EM, Kumulungui B, Pourrut X, Rouquet P, Hassanin A, et al. (2005) Fruit bats as reservoirs of Ebola virus. Nature 438: 575-576.
- Li W, Shi Z, Yu M, Ren W, Smith C, et al. (2005) Bats are natural reservoirs of SARS-like coronaviruses. Science 310(5748): 676-679.
- Carn VM (1996) The role of dipterous insects in the mechanical transmission of animal Viruses. Br. Vet. J 152(4): 377-393.
- Stockholm international peace research institute (SIPRI) (1971b) The problem of chemical and biological Warfare. Technical Aspects of Early Warning and Verification. Humanity press. New York, Vol. 2.
- Poupard JA, Miller LA (1992) History of biological warfare: Catapults to capsomers. Ann N Y Aca Sci 666: 9-20.
- Hugh Hones M (1992) Wickham Steed and German biological warfare research. Intell. Nat. Secur 7: 379-402.
- Centers for Diseases Control and Prevention (CDC) (2013) Bioterrorism Agents/Diseases.
- Friedrich Frischknecht (2003) The history of biological warfare EMBO reports. science & society 4: 47-51.
- Krishnan, Kathiravan, Thangavelu, Raja Muthuramalingam, Sundarajan, et al. (2018) Developing a Programmable, Self-Assembling Squash Leaf Curl China Virus (SLCCNV) Capsid Proteins into "Nano-Cargo"-Like Architecture: A Next-Generation "Nanotool" for Biomedical Applications.
- Thangavelu Raja Muthuramalingam, Ganapathy Rajendran, Ramasamy Pandian, Krishnan Kathiravan (2020) Fabrication of virus metal hybrid nanomaterials: An ideal reference for bio semiconductor. Arabian Journal of Chemistry 13(1): 2750-2765.
- Lico C, Schoubben A, Baschieri S, BlasiP, Santi L (2013) Nanoparticles in Biomedicine: New Insights from Plant Viruses. Current Medicinal Chemistry 20(28): 3471-3487.
- Daniell H, Singh ND, Mason H, Streatfield SJ (2009) Plant-made vaccine antigens and biopharmaceuticals. Trends Plant Sci 14(12): 669-679.
- Lundstrom K (2005) Biology and application of alphaviruses in gene therapy. Gene Ther 12(1): 92-97.
- Nancy L Davis, Ande West, Elizabeth Reap, Gene MacDonald, Martha Collier, et al. (2002) Alphavirus replicon particles as candidate HIV vaccines. IUBMB Life 53(4): 209-211.
- Greer CE, Zhou F, Legg HS (2007) A chimeric alpha virus RNA replicon gene based vaccine for human para influenza virus type 3 induces protective immunity against intranasal virus challenge. Vaccine; 25(3): 481-489.
- Levine B, Huang Q, Isaacs JT, Reed JC, Griffin DE, et al. (1993) Conversion of lytic to persistent alphavirus infection by the bcl-2 cellular oncogene. Nature 361(6414): 739-742.
- Payne CC (1986) The control of insect pests by pathogens and insect-parasitic nematodes. Proc. preliminary report of efficacy. Clinical Otolaryngol 34: 349-357.
- Martignoni ME, Iwai PJ (1981) Catalogue of viral diseases of insects, mites and ticks. Pp: 897-911. In: H.D. Burges (Ed) Microbial control of pests and plant diseases 1970-1980.
- Cunningham JC (1982) Field trials with baculoviruses: Control of forest insect pests. In: E Kurstak (Edt.), Microbial and viral pesticides. Marcel Dekker, New York, Pp: 335-386.
- Yearian UC, Young SY (1982) Control of insect pests of agricultural importance by viral insecticides. In: E Kurstak (Edt.), Microbial and uiral pesticides. Marcel Dekker, New York, Pp: 387-424.
- Jeremy A, Bryong C, Robert H (2015) Expression, delivery and function of insecticidal proteins expressed by recombinant baculoviruses. Viruses 7(1): 422-455.
- Ignoffo M, Conch T (1981) The nucleopolyhedrasisvirus of Heliothis species as a microbial insecticide. Academic Press, London 92-94.
- Jehle J, Blissard G, Bonning B, Cory J, Herniou E, et al. (2006) On the classification and nomenclature of baculoviruses: A proposal for revision. Journal of Virology 151(7): 1257-1266.
- Templeton N (2008) Gene and cell therapy: Therapeutic mechanisms and strategies. 3rd (edn.), CRC Press, Boca Raton. 1267-1289.
- Theze J, Bezier A, Periquet G, Drezen J, Herniou A (2011) Paleozoic origin of insect large dsDNA viruses. Proceedings of the National Academy of Sciences 108(38): 15931-15935. Three way symbiosis required for thermal tolerance. Science 315: 513-515.
- Wright A, Hawkins H, Änggård E, Harper D (2009) A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; a preliminary report of efficacy. Clinical Otolaryngol 34(4): 349-357.
- Harrison RL, Hoover K (2012) Baculoviruses and other occluded insect viruses. Academic Press: Boston, MA 73-131.
- Evans HF (1983) The influence of larval maturation on responses of Mamestra brassicae (Lepidoptera: Noctuidae) to nuclear polyhedrosis virus infection. Arch Virol 75: 163-170.
- Rohrmann G (2013) Baculovirus Molecular Biology. (3rd ), Bethesda MD: NCBI Pp: 46-57.
- Di Lelio I, Varricchio P, Di Prisco G, Marinelli A, Lasco V, et al. (2014). Functional analysis of an immune gene of Spodoptera littoralis by RNAi. J Insect Physiol 64: 90-97.
- Xue J, Qiao N, Zhang W, Cheng R, Bao Y, et al. (2012) Dynamic interactions between Bombyx mori nucleo-polyhedrovirus and its host cells revealed by transcriptome analysis. J. Virol 86(13): 7345-7359.
- Hunter DK, Hall S IPI (1968) Pathogenicity of a nucleopolyhedrosis virus of the beet armyworm. Spodoptera exiqua. J. Invert. Pathol 12(1): 83-85.
- Smith GE, Summers MD (1978) Analysis of baculovirus genomes with restriction endonucleases. Virology 89(2): 517-527.
- Gelernter D, Toscano NC, Kido K Federici BA (1986) Comparison of a nuclear polyhedrosis virus and chemical insecticides for control of beet armyworm, Spodoptera exiqua (Lepidoptera: Noctuidae) on head lettuce. J. Econ. Entomol 79(3): 714-717.
- CoxT et al (2013) PetSmart Toolkit publication. Invasive Animals Cooperative Research Centre Canberra, Australia. Benefits of Rabbit Biocontrol in Australia.
- Van Rensburg PJJ, JD Skinner, RJ Van Aarde (1987) Effects of feline panleucopaenia on the population characteristics of feral cats on marion island. J Appl Ecol 24(1): 63-73.
- Marthán N Bester, JP Bloomer, Rudi van Aarde, BH Erasmus, PJJ van Rensburg, et al. (2002) A review of the successful eradication of feral cats from sub-Antarctic Marion Island, Southern Indian Ocean. S Afr J Wildl Res 32(1): 65-73.
- Abedon ST (2009) Kinetics of phage mediated bio control of bacteria. Food born pathog Dis 6(7): 807-815.
- Burand J, Hunter B (2013) RNAi: Future in insect management. J. Invertebr Pathol 112(1): 68-74.
- Dherelle F (1917) Sur un microbe invisible antagonist desbacilles dysenteries. CR Hebd Seances AcadS 165: 373-375.
- Twort F (1915) An investigation on the nature of ultramicroscopic viruses. Lancet 186(4814): 1241-1243.
- BBC Horizon (1997) The virus that cures. Documentary about the history of phage medicine in Russia and the west.
- Borrell Brendan (2012) Science talk: Phage factor. Scientific American 307(2): 80-83.
- Keen EC (2012) Phage Therapy: Concept to Cure. Frontiers in microbiology 3: 238.
- Lizee G, Gonzales MI, Topalian SL (2004) Lentivirus vector-mediated expression of tumor-associated epitopes by human antigen presenting cells. Hum. Gene Ther 15(4): 393-404.
- Yu H, Zhu Z, Chang J, Wang J, Shen X (2015) Lentivirus-mediated silencing of myosin VI inhibits Proliferation and cell cycle progression in human lung cancer cells. Chem. Biol. Drug Des 86(4): 606-613.
- Li L, Zhang X, LY Zhou, L Ke, et al. (2013) Antitumor efficacy of a recombinant adenovirus encoding endostatin combined with an E1B55KD-deficient adenovirus in gastric cancer cells. J. Transl. Med 11: 257.
- Vermeij J, Zeinoun Z, Neyns B, Teugels E, Bourgain C, et al. (2001) Transduction of ovarian cancer cells: A recombinant adeno-associated viral vector compared to an adenoviral vector. Br. J. Cancer 85(10): 1592-1599.
- Parato KA, Breitbach CJ, Le Boeuf F, Wang J, Storbeck C et al. (2012) The oncolytic poxvirus JX-594 selectively replicates in and destroys cancer cells driven by genetic pathways commonly activated in cancers. Mol Ther 20(4): 749-758.
- Goshima F, Esaki S, Luo C, Kamakura M, Kimura H, et al. (2014) Oncolytic viral therapy with a combination of HF10, a herpes simplex virus type 1 variant and granulocyte-macrophage colony-stimulating factor for murine ovarian cancer. Int. J. Cancer 134(12): 2865-2877.
- Smith RR, Huebner RJ, Rowe WP, Schatten WE, Thomas LB (1956) Studies on the use of viruses in the treatment of carcinoma of the cervix. Cancer 9(6): 1211-1218.
- Webb BA, Michael R Strand, Stephanie E Dickey, Markus H Beck, Roland S Hilgarth, et al. (2006) Polydnavirus genomes reflect their dula roles as mutualists and pathogens. Virology 347(1): 160-174.
- Márquez LM, Redman RS, Rodriguez RJ, Roossinck MJA (2007) Virus in a fungus in a plant: Three way symbiosis required for thermal tolerance. Science 315(5811): 513-515.
- Brigneti G, Voinnet O, WX Li, LH Ji, Ding S, et al. (1998) Viral pathogenicity determinants are suppressors of transgene silencing in Nicotiana benthamiana. The EMBO Journal 17(22): 6739-6746.
- Voinnet O, Lederer C, Baulcombe DC (2000) Viral movement protein prevents spread of the gene silencing signal in Nicotiana benthamiana. Cell 103(1): 157-167.
- Waterhouse PM, Wang MB, Lough T (2001) Gene silencing as an adaptative defense against viruses. Nature 411(6839): 834-842.
- Wassenegger M (2002) Gene Silencing. International Review of Cytology 219: 61-113.
- Bartel DP (2004) MicroRNAs: Genomics, biogenesis, mechanism and function. Cell 116(2): 281-297.
- Robertson D (2004) VIGS Vectors for gene silencing: many targets, many tools. Annual Review in Plant Biology 55: 495-519.
- Ruiz MT, Voinnet O, Baulcombe DC (1998) Initiation and Maintenance of Virus-Induced Gene Silencing. The Plant Cell 10: 937-946.
- Urch Smith TM, Anderson JC, Martin GB, Dinesh Kumar SP (2004) Applications and advantages of Virus induced gene silencing for gene function studies in plants. The Plant Journal 39(5): 734-746.
- Groen SC, Jiang S, Murphy AM, Cunniye NJ, Westwood JH, et al. (2016) Virus infection of plants alters pollinator preference: a payback for susceptible hosts. PLOS Pathog 12(9).
- Hefferon K (2014) Plant virus expression vector development: new perspectives. Biomed Res Int.
- Massa S, Franconi R, Brandi R, Muller A, Mett V, et al. (2007) Anti-cancer activity of plant-produced HPV16 E7 vaccine. Vaccine 25(16): 3018-3021.
- Noris E, Poli A, Cojoca R, Rittà M, Cavallo F, et al. (2011) A human papillomavirus 8 E7 protein produced in plants is able to trigger the mouse immune system and delay the development of skin lesions. Arch Virol 156(4): 587-595.
- Wen Amy, Steinmetz M, Nicole F (2016) "Design of virus-based nanomaterials for medicine, biotechnology, and energy". chemical society reviews 45(15): 4074-4126.
- Vail PM, DL Jay DL, Hunter OK (1971) Cross infectivity of a nuclear polyhedrosis Virus isolated from the alfalfa looper, Autoqrapha californica. Proc. 4th Coll. Insect Pathol, Brighton Pp: 297-304.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...