Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4579
Research Article(ISSN: 2637-4579) 
Implications of EEG and Speech Signal in the Analysis of Neurological Disorders-A Survey Volume 3 - Issue 3
Chetan Balaji2 and D S Suresh2*
- 1Research Scholar, Channabasaveshwara Institute of Technology, Research Centre, Karnataka, India
- 2Professor, Channabasaveshwara Institute of Technology, Gubbi, Tumkur, Karnataka, India
Received: July 31, 2019; Published: August 08, 2019
*Corresponding author: D S Suresh, Research Scholar, Channabasaveshwara Institute of Technology, Gubbi, India
DOI: 10.32474/OAJBEB.2019.03.000165
Abstract
This paper presents the need of diagnosing Neurological disorders related to Alzheimer’s disease (AD) as well as Parkinson Disease (PD) by analyzing both EEG and Speech Signal. Research papers discuss about the various approaches in the analysis of Alzheimer’s disease by extracting the electrical activity of brain signals. EEG systems are widely used to extract brain signals as they are quite capable of measuring Electrical activities of the brain but possess very poor spatial resolution and will be contaminated with various artifacts. Current Surveys indicate that about 70% - 90% of patients suffering with Parkinson’s disease exhibit dysphonic (Impairment in speaking ability) symptoms; thereby speech signal analysis is widely used to diagnose PD. Due to ambient environment, motion artifacts the speech quality may be varied which may result in wrong diagnosis. Even though researchers propose various risk factors for effective analysis of AD and PD using EEG and Speech signals the above stated limitations of EEG and Speech Signal can lead to a fault diagnosis of the diseases and may lead to subtle manifestation of defects. An efficient diagnostic system is therefore necessary to diagnose these Neurological disorders more effectively analyzing EEG and speech signal. This paper addresses the different techniques and methodologies incorporated in diagnosing the Neurological disorders such as AD and PD using EEG and Speech signal which provides a better insight to carry out the further research by developing an efficient integrated algorithm for diagnosing the above stated Neurological disorders using both EEG and Speech Signal.
Keywords: Parkinson’s disease; Alzheimer’s disease; EEG; Speech Signal; FOG
Introduction on Neurological Disorders
Neurodegenerative diseases like Alzheimer’s disease (common type of Dementia), Parkinson’s and Epilepsy disease are generally a group of disorders that are progressive and create serious impairment of the nervous system. These are considered as most disabling disease and it has high prevalence in developed countries and at a high rate in elderly people. It is expected to quadruple by 2020 [1-3]. The most common type of dementia is Alzheimer’s disease which can be found in 50%-75% of all cases of dementia. The brain cells and the nerves will be destroyed which causes the problems in the transmitters which contributes to carry messages in the brain. The symptoms start with lapses in memory, problems in identifying the correct words, swing in the daily mood etc. As EEG systems are quite capable of measuring brain waves of an individual, it is used as the main tool in the diagnosis of Alzheimer’s disease. The EEG waves that represent the different mental states of the human are categorized by their frequency, shape, amplitude as well as position of electrode.
They are named as Alpha waves (8- 13 Hz) that are commonly observed in adults who is awake but in relaxed state with eyes closed. The Beta waves (13- 30 Hz) are concerned with actions and behaviors. Gamma waves (30-100 Hz) are associated with consciousness and perception. The waves associated to subconscious activity are Theta waves (4-8 Hz). These are observed in the meditation state and deep relaxation. The slowest brain activity found in all stages of sleep is Delta waves (0-4 Hz). These EEG signals/waves are of very small amplitude, and highly susceptible to myoelectrical, motion and environmental artifacts, hence it has made the analysis of EEG signal more complex. The removal of artifacts by retaining the required is the most complex task in EEG signals. Though many research works have been carried out in removing the artifacts, validation is still needed to ensure that only artifacts are removed from EEG signals. Parkinson’s disease is a chronic neurodegenerative disorder characterized by the progressive deterioration of motor function and the emergence of considerable non-motor problems. The loss in dopaminergic neurons in the mid part of the brain is one of the characteristics of PD. Patients with PD usually suffer from hypo kinetic dysarthria, which associates articulation, impairment of phonation, speech and prosody.
Approximately 70% to 90% of patients affected with PD undergo problems with Speech. Therefore, Speech Signal analysis is liable for diagnosing Parkinson’s disease. The different features such as energy, speaking rate, pitch, harmonic ratio, shimmer, jitter is taken into considerations for detecting the Parkinson’s disease using Speech signal. Due to any unavoidable circumstances, If the voice changes, it may lead to faulty diagnosis. Even though the advances in the technology propels an increasing number of studies related to the diagnosis of neurological disorders, still there remains a few unaddressed problems and issues that occur in EEG and Speech Signals which may lead to faulty analysis of the signals and results in the wrong diagnosis. Hence there is a need to develop effective methodology and algorithm in the diagnosis AD and PD. This paper provides an overview on various methodologies used in the diagnosing AD and PD considering both EEG and Speech Signals respectively and the necessity of an integrated Algorithm for the detection of AD and PD. This paper consists of four sections; this part of introduction will be followed by the section II which discusses the basic frame work for the analysis of EEG Signal and Speech Signal. Section III highlights the literature review on various modes of diagnosing PD and AD using EEG and Speech Signal and finally Section IV presents the proposed methodology.
Generalized Methodology to Analyze EEG and Speech Signals
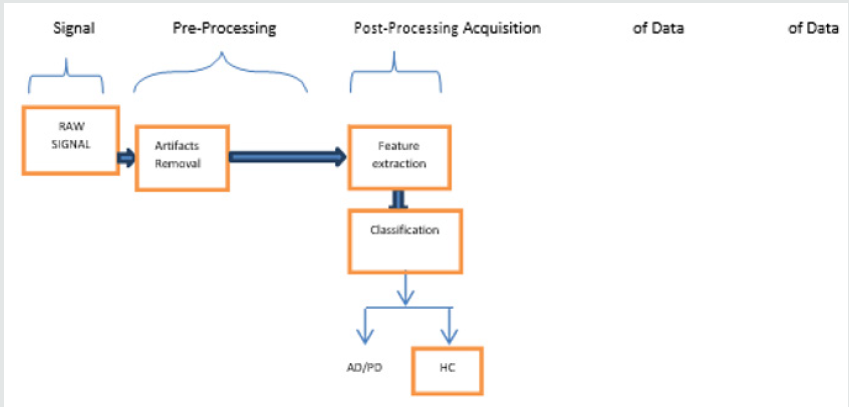
The millions of neurons in the human brain play a vital role in controlling the behavior of a human body. These neurons act as the information carrier between brain and human body. EEG is a modality which is efficient to acquire the brain signals that corresponds to various state of the scalp surface area. Hence the EEG signal analysis is considered as a primary focus to analyze the brain signals which can be used to diagnose various brain disorders. Speech is a very complex process that involves a wide range of Bio signals. A successful speech recognition system requires the knowledge and expertise from a wide range of disciplines such as computer science, physiology, communication and Information theory, Signal processing and many more. The modules and the frameworks that have been used frequently to analyze the EEG and speech signal are shown in (Figure 1). During the first phase raw EEG/Speech signals are collected. In the Pre-Processing Stage, which is a second phase, the artifacts removal, signal averaging, enhancement of resulting signal, thresholding of the output and finally edge detection will be handled.
Artifacts may occur due to several factors which include head motion, Physical Problems and connectivity issues. The artifact removal is quite challenging task as it creates signals with abnormal frequency and shapes. During the Post-Processing phase some meaningful signal features in time and frequency domain which highlights the relevant information of the signal like mean, standard deviation, zero crossings, etc. will be extracted using various signal processing techniques like Fourier transform, wavelet decomposition ,Principal Component Analysis, Independent Component analysis, etc. The last stage is the classification stage which is mainly used for the analysis of the extracted EEG and Speech signals and to classify these signals based on the target observations [1]. Even though propelled by technology advances in investigation of EEG and speech signals, due to their interdisciplinary nature the analysis of these random signals still remains a topic of interest and a very active research field and gives a scope for improvement in accuracy and efficiency.
Literature Survey
This section briefs out the related works and outcomes undergone in the field of:
a. Diagnosis of PD using EEG signal
b. Diagnosis of PD using Speech Signal
c. Diagnosis of AD using Speech signal
d. Diagnosis of AD using EEG Signal
a) Diagnosis of Parkinson’s Disease Using EEG Signal
The most common disabling disturbances of PD with respect to the physical activity are the Freezing of Gait (FOG), Turning Freeze (TF), and Gait Initiation Failure (GIF). The PD affected patient’s experiences difficulty in walking as they feel that their feet have been glued to the floor. During this stage the EEG signals can be recorded from the patients given them a TUG tasks. The cognitive process (brain mechanism) can be analyzed for the timing and scaling of movement using EEG signal analysis and could be a useful tool in the detection of above-mentioned disturbances. The below section discusses the related work carried in the analysis of PD using EEG signals. AM Ardi Handojoseno, et.al discusses the method of detection of Freezing of Gait in PD patients using EEG signal based on wavelet decomposition. In this paper the dynamics of FOG was analyzed by extracting the EEG Entropy and Sub-band wavelet energy. 26 Patients around 60-70 years with PD and significant FOG were considered. The author highlights that by using BPNN (Back Propagation Neural Network) classifier the onset of Freezing of PD can be identified with 75% accuracy, specificity and sensitivity using sub bands of EEG.
Also, the statistical analysis indicates that there will be a difference in the Energy sub bands of delta, theta and alpha with Normal and PD affected group. Finally gives a future scope for the usage of different classifiers for still better results [4]. [5] Jonathan Weihenmayer proposes the method of diagnosing of PD patients with a Healthy control by extracting the Raw and Clean EEG data of patients during the quasi resting state for 1 sec of Nine PD patients and ten healthy individuals. Nonlinear delay differential equations (DDE) are applied to classify the PD patients from and Healthy individual. The classification is done upon the plotting of area under the receiver operating characteristic curve (AROC). It is shown that the raw EEG data (AROC = 0.59-0.86) can classify the PD patients with a Healthy individual more robustly than clean EEG data (0.57-0.72).The author states that increased classification of raw EEG time series originates from muscle artifacts and concludes by stating that a non-linear extraction and classification of raw EEG data can be used as biomarker for PD. This paper can be extended for a further scope for research by considering the patients in true resting states and for a larger data sets and sample size.
Quynh Tran Ly et al. [6] discuss the brain mechanism during the Gait Initiation Failure (GIF), which is a subtype of Freezing of Gait (FOG). EEG signals of 61 samples were recorded from Four PD affected patients (3 Male and 1 Female) to study the brain dynamic changes during the occurrence of GIF. EEG samples were analyzed through the Centroid Frequency (CF) and Power Spectral Density (PSD). The results showed that GIF were associated with significant increase in high beta band (21-38 Hz) in the frontal, central, parietal, occipital regions of brain. Using BPNN classifier the analysis of PSD and CF in EEG provided 84.27 % of performance and 84.80% of sensitivity and accuracy respectively. The author concludes the paper by stating that High beta activity was found in EEG with PD affected patients during the analysis of GIF and suggested to use a larger sample size and other multiple electrodes.
Paper [7] by Quynh Tran Ly et al. discusses the best montages (Placement of Electrodes) to detect the FOG in PD patients. The identification of combination of sensors and optimal location are also discussed. EEG measures/parameters, in the form of Power Spectral entropy, Power Spectral density and Centroid Frequency for 343 seconds from 7 patients (6 M and 1 F) with ‘Effective walking’ and PD affected with FOG during a series of TUG were extracted to analyze the important features that were significantly changed during FOG. Feed- Forward Neural networks were applied to classify the EEG signals amongst the ‘Effective walking’ and Freezing. The sensitivity and accuracy from the classification using single channel, two channels, three, Four, Six, Seven and Thirty-two channels were discussed. The performance of C4O2 in a two channel and four locations at F3C4P4O2 provided the best indicator of FOG detection with 72.54% and 72.29% of sensitivity and 69.71% and 70.29% of accuracy respectively. The outcome of the paper is that the best montage is using EEG system with two channels C4-O2. The author concludes by stating that the increase in the number of channels will not increase the accuracy and suggests that the further scope of research can be done by considering larger sample of data and to use of any specific Neural Network which can lead to a better result. The authors [8] discuss a method for the suppression of Artifacts that occur in EEG signals due to Neuro stimulation.
The Neurostimulation is assessed by Advanced Statistical Signal Processing and Machine Learning Methodologies. The unsupervised clustering method is applied to neural recordings in order to suppress Neurostimulation. The author also discusses about the Deep Brain Simulation (D B S) method for the treatment of PD, where electrical simulator is placed into the body and it delivers the electrical current to thalamus in order to reduce the severity of PD. This Simulator can send the EEG signals which indicates the treatment and possible DBS side effects. Time Frequency features in EEG are extracted in patients undergoing DBS treatment of PD using matching pursuit decomposition (MPD). This paper concludes by stating that the behavioral task clustering for PD patients with DBS using Time frequency features gives the differences in the band activity (delta, beta and gamma) of EEG. The authors Quynh Tran Lya et al. [9] briefs about technique of S-transform decomposition of EEG Signals in detection of Turning freeze in PD patients. Turning Freeze (TF) is the most common subtype of FOG and is causing the majority rate of falls in PD patients.
The brain signals were captured to using EEG signal to measure the feet getting stuck while making a turn (TF). Data was collected from six PD affected patients during TUG tasks. The main discussion in this paper is the extraction of EEG features using time frequency technique S-Transform decomposition (Stock well transform). Independent Component analysis by entropy bound minimization technique was used to separate EEG sources. Bayesian neural networks (BNN) classification algorithm was implemented to improve the classification results in detecting TF in PD patients. The main findings in this paper are the inclination in the high beta activity in the region of cortex near occipital O2 and frontal F4 locations. The author concludes the paper by projecting 84.24% sensitivity, 88.0% of specificity and 86.2% accuracy in detecting the TF. The scope for the future work is to increase the accuracy. AM Ardi Handojoseno et al. [10] proposes a methodology for the prediction of FOG in PD using EEG spatial correlation, wavelet coefficient and cross frequency energy techniques. Integration of spatial, spectral and temporal features of EEG signal with the use of Neural Network and K-NN classifiers is discussed. EEG signals from 26 PD affected patents with significant FOG were recorded during TUG tasks. The individual EEG data is extracted using Wavelet transform. The statistical features like mean, SD, Skewness, Kurtosis, maximum and minimum were applied to extract information from the wavelet coefficient of and EEG epoch. Multilayer perceptron neural network (MLP) analysis was used to classify data samples based on selected features. The obtained features were also compared with K-NN classifiers. MLP-NN provided the best results of 83.37% of sensitivity with input parameter as WCS (Wavelet Cross Spectrum) and 71.47%, 73.47% of specificity and accuracy respectively
K-NN provided best results with 87.25% of sensitivity by the combination of all features and 61.50% specificity using centroid frequency wavelet cross spectrum (CFWCS) and highest accuracy of 72.00% using WCS. The author concludes by stating that the novel approach combining the spectral, spatial and temporal features of surface EEG recordings may prove effective method for the prediction of FOG in PD affected patients and gives a scope for further research by considering a better classifier which can improve the utility of this approach. This study of paper [11] by AM Ardi Handojoseno et al. highlights upon the requirement of ambulatory EEG to record the underlying brain dynamic changes associated with FOG and also suggests the most suitable EEG sensor location that can detect FOG effectively. The EEG recordings from four PD affected patients and features of the EEG recordings were extracted using PSD. This study indicates that the Occipital area of the brain is more affected during TF. Visual Cortex region can be considered as optimal reference location for the detection of TF. This paper concludes with the result that proved with significant alterations in the high beta and theta power spectral densities across the occipital and parietal areas during TF and gives scope to use of more advanced feature extraction and classifier systems that can provide a better performance.
b) Diagnosis of Parkinson’s Disease using Speech Signal
The PD is a kind of Degenerative disease of the nervous system and is the second major neurodegenerative disease after the AD. The PD is characterized by a group of certain conditions which causes the disorders in the motor system due to loss of dopamineproducing brain cells. The recent estimates indicate that 90% of patients with PD typically will exhibit the symptoms in vocal impairment, which is known as dysphonia. Hence the reliable tool to detect and monitor PD is to collect the voice samples and to measure the dysphonic indicators [12]. The following literature survey will give a brief overview of analysis of Speech Signal to detect PD. Max A Little and coauthors [12] discriminates healthy people from PD affected people by using a new measure of dysphonia called PPE (Pitch Period entropy) that observes the changes in speech specific to PD.. The author with 195 sustained phonations from 31 people with 23 people affected with PD achieved an overall correct classification performance of 91.4 % using Kernel Support vector machine as classifier.
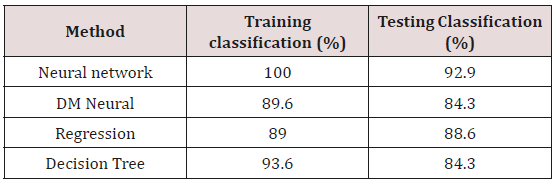
The comparison of different types of classification methods for the effective diagnosis of PD is highlighted in paper [13] by Rasul Das. Four independent classifiers viz Neural Network, DM Neural, Regression and Decision tree were applied to diagnose the PD. A detailed comparison suggests that the Neural Network classifier yields the best result with 92.9 % and is much better than Kernel Support vector machine classifier. Table 1 from paper [11] indicates the different model classification rates. Hui-Ling Chen et al. [14] discuss about the detection of PD using Fuzzy K Nearest Neighbor approach which is the improved version of non- parametric classification method K-NN and incorporates PCA for feature reduction to eliminate redundant features. The comparison is made between F-KNN classifier with SVM classifier and concludes that F-KNN approach performs advantageously and achieves better results in terms of specificity, sensitivity, accuracy and AUC. Michal Vadovsky et al. [15] cover the option of determining which category of audio data shall be utilized to create the most accurate classification model.
The data sets were recorded from the PD affected patients covering the records in the form of sentences, permanent vowels pronunciation, numbers and certain words. It was highlighted that the precise classification decision trees can be constructed using the speech signals of the patients’ pronouncing the numbers which produced an accuracy of 66.5 %. The Parkinson disease data sets were investigated by using machine learning tools by pronouncing the sustained vowels. The authors Betul Erdogdu Sakar et al. [16] claims that the sustained vowels will carry more discriminative information of PD than the short sentences and isolated words do. Hananel Hazan et al. [17] applies Machine learning tools for the early diagnosis of PD. Support Vector Machine (SVM) along with Radial Basis Function (RBF) kernel was used as the Machine learning tool can be used for the speech data in the classification of PD from Healthy controls. The accuracy in the range of 80%-88% was achieved by using only vowel formants. The authors [18] discuss the method of selecting meaningful and dominant features from the speech signal to predict the PD from Healthy controls using the factor analysis approach.
Along with shimmer, jitter, PPE the Recurrence period density entropy (RPDE) and spread parameters are also the features which can be considered for detecting PD. An average accuracy, specificity and sensitivity was achieved by using SVM as classifier and gives an insight that even sustained phonations will carry the necessary information of PD. The analysis of four tasks of speech in the form of sustained vowels, free speech, reading and fast syllable repetitions were carried out by extracting the features by MFCC (Mel Frequency Cepstral Coefficients) with GMM (Gaussian Mixture Model) [19] to detect the early stages of PD. The average accuracy using these techniques was in the range of 60 % to 91 % with 91 % as the best accuracy in men during reading the task. The conventional and non-conventional dysphonic features were collected [20] as data sets for the PD diagnosis. The model based clustering using GMM based was used to discard the irrelevant information (Feature weighing).Feature reduction/selection were carried out using principal component analysis (PCA), sequential backward selection (SBS), sequential forward selection (SFS) and linear discriminant analysis (LDA)techniques , and classification using three supervised classifiers such as probabilistic neural network (PNN) ,least-square support vector machine (LS-SVM), and general regression neural network (GRNN). The author claims that the combination of the feature pre-processing, above feature reduction/ selection methods and classification gives a maximum classification accuracy of 100% for the Parkinson’s dataset. Zhennao Cai et al. [21] establishes an optimal support vector machine (SVM) based bacterial foraging optimization (BFO) to predict PD effectively. This mode of approach is compared with SVM based on particle swarm optimization (PSOSVM) and SVM based grid search method (Grid-SVM) methods. A classification accuracy of 97.42% was achieved by using this (BFOSVM) method and proved to be the best method compared to the other two (PSO-SVM and Grid SVM).
c) Diagnosis of Alzheimer’s Disease using Speech Signal
The second major problem after the loss of memory in AD patients is the language. This loss of language will affect the people suffering from AD in speaking, understanding, recognizing, naming things, finding the proper word in the spontaneous/continuous speech, usage of vocabulary in daily needs and very limited answers to the questions .The below literature briefs the related works carried out in diagnosis of AD using speech signal analysis. [22], the early detection of AD has been improvised by applying the non-invasive methods viz. Automatic Spontaneous Speech Analysis (ASSA) and Emotional Temperature (ET). The spontaneous speech fluency by the measures of pause length, segment length of voice, and short time energy will be used as bio markers in early diagnosing of AD. The voice samples from the people were collected and measured to analyze the degree of severity of the AD patients using SVM classifier and radial basis kernel function. The results showed that there will be a significant loss in fluency with regard to voiced/unvoiced percentage in the speech and also in the length of voiced/unvoiced segments and lower short time energy, decrease in the length of voiced segments and increase in the unvoiced segment numbers.
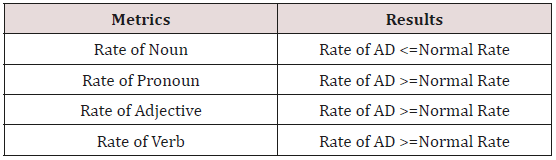
The cognitive status of a person is analyzed by the continuous monitoring of the speech to decide whether the person is suffering from AD. The voice samples are collected through microphone and these sequences of characters are converted into a sequence of tokens (Verb, Noun, Pronoun, Adjective etc...) through lexical analyzer and are further processed for decision making [23,24]. Table 2 indicates the results of the sequence of tokens in AD and HC (Healthy Controls). People affected with AD may face difficulty in understanding the speech in the existence of the Background noise. They have CAPD (Central Auditory Processing Disorders) problems. This may lead to the deterioration of Speech, processing the emotional qualities of words as well as in prosody recognition. Speech output gets deteriorated, as patients with Alzheimer’s disease have issues in properly vesting their speech with emotions [25]. Alexandra Keonig et al. [26] the short cognitive vocal tasks the first vocal markers during the voice recordings were extracted. These vocal markers were tested and classified using Mann- Whitney U test to differentiate between AD and healthy controls.
d) Diagnosis of Alzheimer’s Disease using EEG Signal
The most common type of Dementia is Alzheimer’s disease which is degenerative brain syndrome that affects memory, thinking, behavior and emotion. People suffering from AD face issues in performing the previously routine tasks, personality changes, loss of memory, and difficulty in finding right words etc. [ardsi.org]. The recording of the brain signals and its analysis shall be made to diagnose the AD from healthy controls. EEG provides high temporal resolution systems and is efficient tools in recording the brain signals. The literature discussed below gives an overview of different techniques and classifiers used for the effective diagnosis of AD by analyzing the EEG signals. Patients with AD have less complexity in EEG signals compared to healthy controls. The features such as spectral entropy (SE), spectral roll-off (SR), zero-crossing rate (ZCR), spectral centroid (SC) and Higuchi fractal dimension (HFD) in EEG signals were extracted and analyzed. These features carry relevant information in the primary and temporal lobes and are less complex and lower in patients with AD. The SE and ZCR values are found less in frontal and temporal lobes. The SVM classifier achieved an accuracy of 78% [27].
The analysis of coherence and frequency of qEEG were studied to attain greater accuracy in diagnosing AD. This analysis indicates that AD patients will present increase in absolute power values in delta band and theta band [28]. The accuracy is around 72.3 % which is very limited [28]. The relative power in various EEG frequency bands were used as attributes in distinguishing AD and Healthy controls [29]. The frequency bands in the range of 4-30 Hz are tested using linear discriminant analysis, Mann- Whitney U test which spotlights that the frequency bands between 4 Hz and 7 Hz is optimal for the detection of AD. EEGs were recorded and the relative power were reckoned for alpha 1, alpha 2, alpha 3, delta and theta which indicates that there will be an raise in the delta power and fall-off in the alpha 2 power in patients with CVD (Cerebrovascular damage). The pathological changes in the various EEG markers will play a prominent role in the segregation of AD patients [30].
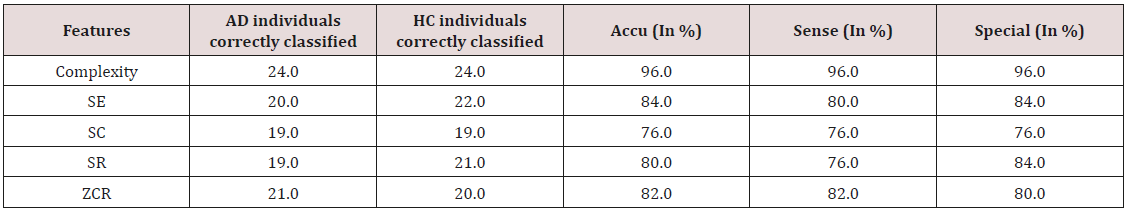
The role of EEG as Biomarkers to detect the AD is discussed in [31]. The Neural changes that are associated with AD can be detected with biomarkers such as EEG and qEEG. The author in his vast survey proves that the patients suffering from AD will have the change in the rhythmic activity of the brain. The power in the delta and Theta frequencies will be increased with the decrease in the alpha and Beta power. Hence to identify these changes EEG biomarkers are most suitable. Based on the different complexity features EEG signals were analyzed in AD patients [32] and obtained the result indicating the complexity loss in EEG signals of Alzheimer’s disease patients. Zero Crossing Rate (ZCR) along with Spectral Entropy (SE), Spectral Roll-off (SR), and Spectral Centroid (SC) were the features analyzed to diagnose AD. The accuracy, sensitivity, specificity percentage were calculated. (Table 3) highlights the accuracy, specificity and sensitivity values for different features of AD patients and HC. The EEG activity in the analysis of AD was carried out by analyzing Cross Sample Entropy (CSE) and graph theory measures. The characteristic path length (PL) and clustering coefficient (CC) were calculated. The result after the analysis proved that the CSE values were lower. CC values were declined and found inclination in PL values in AD patients [33].
Table 3: Highlights the accuracy, specificity and sensitivity values for different features of AD patients and HC.
Discussions and Proposed Methodology
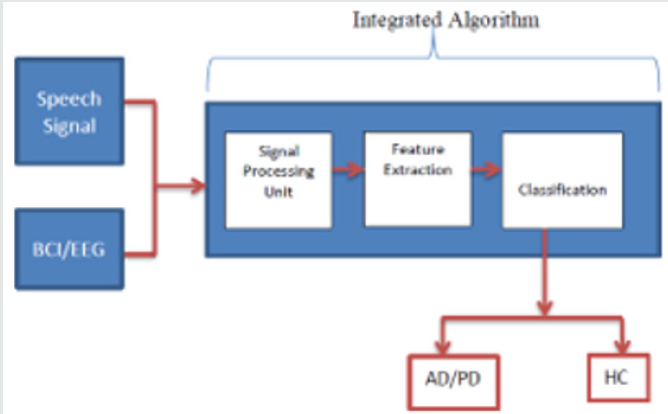
This paper gives an insight to various methodologies and techniques used to diagnose AD and PD using EEG and Speech signals. Though EEG systems are quite capable of measuring electrical activities of the brain, but they exhibit poor spatial resolution and will be contaminated with various artifacts. In addition, speech signal experiences deterioration due to ambient environment and motion artifacts. These limitations of EEG and Speech signal can give a way to fault diagnosis of the diseases and may lead to subtle manifestation of defects. Though recent research has proposed various methods to overcome the above-mentioned drawbacks in EEG and Speech signal, validation is still needed to ensure the accuracy and early diagnosis of these Neurological disorders. In our research work we propose a unified/Integrated algorithm that can diagnose Parkinson’s disease and Alzheimer’s disease by EEG as well as Speech signal which contributes in improvising the existing techniques. The framework of the proposed approach/design is shown in Figure 2.
Acknowledgement
Authors gratefully acknowledged to Trivedi Global, Inc., Trivedi Science, and Trivedi Master Wellness for their support. In addition, authors are thankful for the support of Dabur Research Foundation for conducting this study.
References
- J Satheesh Kumar, P Bhuvaneswari (2012) Analysis of Electroencephalography (EEG) and its categorization A Study. Procedia Engineering 38: 2525-2536.
- Tanja Schultz (2017) Bio signal Based Spoken Communication: A Survey. ACM Transactions on audio, speech, and language processing IEEE 25(12).
- Sandhya Joshi, Deepa Shenoy (2010) Classification of Alzheimer's disease and Parkinson's Disease by Using Machine Learning and Neural Network Methods. Second International Conference on Machine Learning and Computing.
- A M Ardi Handojoseno, James M Shine (2012) The detection of Freezing of Gait in Parkinson’s disease patients using EEG signals based on Wavelet Decomposition. 34th Annual International Conference.
- Jonathan Weyhenmeyer (2014) Muscle artifacts in single trail EEG data distinguish patients with Parkinson’s disease from healthy individuals. 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society.
- Quynh Tran Lya (2016) Detection of gait initiation failure in Parkinson’s disease patients using EEG signals. 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society.
- Quynh Tran Ly (2016) Identifying montages that best detect the electroencephalogram power spectrum alteration during freezing of gait in Parkinson's disease patients. 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) pp 6094-6097.
- A Maurer, S Hanrahan, J Nedrud (2016) Suppression of Neurostimulation Artifacts and Adaptive Clustering of Parkinson’s Patients Behavioral Tasks using EEG. 50th Asilomar Conference on Signals, Systems and Computers.
- Quynh Tran Lya, AM Ardi Handojoseno (2017) Detection of Turning Freeze in Parkinson’s Disease based on S-transform Decomposition of EEG signals. Annual International Conference of the IEEE Engineering in Medicine and Biology Society pp3044-3047.
- AM Ardi Handojoseno, James M Shine (2013) Using EEG Spatial Correlation, Cross Frequency Energy and Wavelet Coefficients for the prediction of Freezing of Gait in Parkinson’s disease patients. 35th Annual International Conference EMBS IEEE pp 4263-4266.
- AM Ardi Handojoseno (2015) An EEG Study of Turning Freeze in Parkinson’s disease Patients: The Alteration of Brain Dynamic on the Motor and Visual Cortex. Annual International Conference of the IEEE Engineering in Medicine and Biology Society, pp. 6618-6621.
- Max A Little (2009) Suitability of Dysphonia Measurements for Telemonitoring of Parkinson’s disease. Transactions on Biomedical Engineering.
- Resul Das (2009) A comparison of multiple classification methods for Diagnosis of Parkinson disease. Expert Systems with Applications 37(2): 1568-1572.
- Hui Ling Chen (2012) An efficient diagnosis system for Detection of Parkinson’s disease using fuzzy k nearest neighbor approach. Expert Systems with Applications 40(10): 263-271.
- Michal Vadovský, JánParali (2017) Parkinson´s Disease patients Classification based on the speech signals 15th International Symposium on Applied Machine Intelligence and Informatics (SAMI).
- Betul Erdogdu Sakar, M Erdem Isenkul (2013 ) Collection and Analysis of a Parkinson Speech Dataset with Multiple Types of Sound Recordings IEEE- Hananel Hazan et al. Early diagnosis of Parkinson's disease via machine learning on speech data. 27th Convention of Electrical and Electronics Engineers-IEEE 17(4): 828-834.
- Hananel Hazan (2012) Early diagnosis of Parkinson's disease via machine learning on speech data- 27th Convention of Electrical and Electronics Engineers- IEEE.
- Spriha Chandrayan (2017) Selection of Dominant Voice Features for Accurate Detection of Parkinson’s disease. 3rd International Conference on Bio signals, images and instrumentation-IEEE.
- Laetitia Jeancolas (2017) Automatic Detection of Early Stages of Parkinson’s Disease through Acoustic Voice Analysis with Mel-Frequency Cepstral Coefficients. International Conference on Advanced Technologies for Signal and Image Processing (ATSIP).
- M Hariharan (2014) A new hybrid intelligent system for accurate detection of Parkinson’s disease. Comput Methods Programs Biomed 113(3): 904-913.
- ZhennaoCai (2017) A New Hybrid Intelligent Framework for Predicting Parkinson's Disease. IEEE.
- K Lopez de Ipna (2012) New approaches for Alzheimer Disease Diagnosis based on Automatic Spontaneous Speech Analysis and Emotional Temperature. 4th International work shop pp 407-414.
- Vassilis Baldas (2011) Early Diagnosis of Alzheimer’s Type Dementia Using Continuous Speech Recognition.
- Ali Khodabakhsh (2014) Detection of Alzheimer's disease using prosodic cues in conversational speech. 22nd Signal Processing and Communications Applications Conference (SIU).
- Vassili kiIliadou (2004) Clinical psychoacoustics in Alzheimer's disease central auditory processing disorders and speech deterioration. A Review. Annals of General Hospital Psychiatry 2(1): 12.
- Alxendra Koing (2015) Automatic speech analysis for the assessment of patients with pre-dementia and Alzheimer’s disease. Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring 1(1): 112-124.
- Tyler Staudinger (2011) Analysis of Complexity Based EEG Features for the Diagnosis of Alzheimer’s Disease. Conference of the IEEE Engineering in Medicine and Biology Society pp 2033-2036.
- Lineu Corrêa Fonseca (2011) Quantitative electroencephalography power and coherence measurements in the diagnosis of mild and moderate Alzheimer's disease. Neuro Psiquiatr. 69: 297-303.
- Mohamed Elgendi (2011) Optimization of EEG frequency bands for improved diagnosis of Alzheimer disease. Conference of the IEEE Engineering in Medicine and Biology Society. Pp. 6087-6091.
- Davide Vito Moretti (2014) The Contribution of EEG to the Diagnosis of Dementia. J. Biomedical Science and Engineering, pp. 546-562.
- Noor Kamal Al Qazzaz (2014) Role of EEG as Biomarker in the Early Detection and Classification of Dementia. The Scientific World Journal, pp. 16.
- Nilesh N Kulkarni (2017) Use of Non-linear Complexity features for EEG based dementia and Alzheimer’s disease Diagnosis
- Carlos Gómez (2016) Analysis of spontaneous EEG activity in Alzheimer's disease using cross-sample entropy and graph theory. Conference of the IEEE Engineering in Medicine and Biology Society, pp. 2830-2833.
Editorial Manager:
Email:
biomedicalengineering@lupinepublishers.com

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...