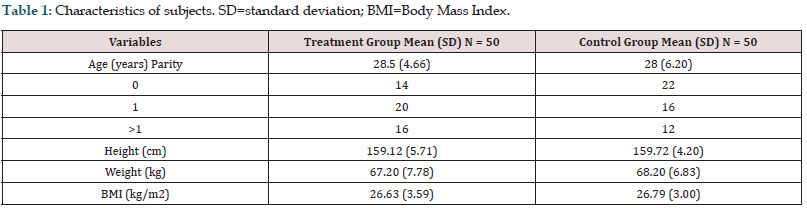Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2644-1403
Research Article(ISSN: 2644-1403) 
Effect Of Intrathecal Labor Analgesia On Labor-Induced Endothelial Nitric Oxide Synthase, Tumor Necrosis Factor- Alpha, Interleukin-6, And Neonatal Lactate Levels: A Randomized Controlled Trial Volume 3 - Issue 5
Raden Theodorus Supraptomo*, Suroto, Bambang Purwanto and Ari Natalia Probandari
- Department of Anesthesiology and Intensive Care, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia
Received: November 13, 2020; Published: December 09, 2020
Corresponding author: Raden Theodorus Supraptomo, Department of Anesthesiology and Intensive Care, Dr. Moewardi Hospital, Jalan Kolonel Sutarto No.132, Jebres, Surakarta, Central Java 57126, Indonesia
DOI: 10.32474/GJAPM.2020.03.000175
Abstract
Background: Labor pain is the most painful experience. The use of neuraxial intrathecal labor analgesia (ILA) in standard delivery is increasing with lower maternal pain scores and higher levels of satisfaction. This study was aimed to demonstrate that neuraxial ILA mechanisms decrease endothelial nitric oxide synthase (eNOS), and decrease interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α).
Methods: This randomized controlled trial was conducted in 100 pregnant patients, aged <35 years old, who will be in labor within 2017 and were a patient in obstetrics and gynecology at six hospitals and six primary health centers in Surakarta. Subjects were randomized and divided into two groups. Group ILA (n = 50) received 2.5 mg bupivacaine + 50 μg clonidine +0.1 mg morphine and the control group (n = 50) did not receive any treatment. Laboratory findings of eNOS, TNF-α, IL-6, and neonatal lactate levels were recorded.
Results: There was a decrease in TNF-α levels by 44.12%, and in IL-6 levels post-treatment by 74.85% in the treatment group (p<0.05). However, eNOS levels also decreased by 22.87% in the treatment group. There was a difference of 23.40% in lactate levels between the treatment and control groups.
Conclusions: Neuraxial ILA can cause a reduction in TNF-α, IL-6, and neonatal lactate levels and decrease eNOS levels.
Keywords: Analgesia; endothelial nitric oxide synthase; interleukin-6; labor pain; regional anesthesia; TNF-alpha
Introduction
Labor pain is described as the most painful experience. It is a painful experience that results from some physiological causes. Labor pain grew in its importance since its first mention about 1.5 centuries ago. However, along with developments in the field of medicine, anesthesia began to be used to treat labor pain, with ether as the first agent. Since the discovery in 1987 by Moncada et al. 1 nitric oxide (NO) has become one of the mediators of pain in labor. In this study, NO was identified as an important biological mediator, which is involved in diverse activities, such as smooth muscle relaxation, neurotransmission, inflammation, and regulation of the immune response [1] Previous research by Bullarbo et al [2] showed that the use of isosorbide dinitrate as an NO donor to ripen the cervix and induce labor showed promising and safe results. The mechanism of the influence of neuraxial analgesia on NO levels is not fully known. It is suspected that, in labor, there is a decrease in NO levels due to the influence of pain [2] Some studies, such as those conducted by Chen et al. [3] showed that the administration of neuraxial analgesia could prevent a decrease in NO levels during labor. The use of neuraxial intrathecal labor analgesia (ILA) techniques in standard delivery is increasing in use (e.g., 22% in 1981, 61% in 2001, and 75–80% in 2013) [4] There are physiological benefits for both the mother and fetus through improvements in the cardiovascular and respiratory status of the mother and fetal outcomes, so that this technique has the potential to become the gold standard for labor analgesia [5] This study intends to prove that neuraxial ILA mechanisms relieve pain and shorten labor duration, by reducing eNOS and aerobic metabolism as well as lactate levels, and serve as predictors of neonatal outcome.
Methods
This randomized controlled trial was conducted in 100 pregnant patients, aged <35 years old, who will be in labor within 2017, and were a patient in obstetrics and gynecology at any of the six hospitals and six primary health centers in Surakarta. Ethical clearance was obtained from the Health Research Ethics Committee of Faculty of Medicine, Universitas Sebelas Maret, Dr. Moewardi Hospital (No: 827/VIII/HREC/2017). The sample size was 100 women calculated using Open-Source Epidemiologic Statistics for Public Health software version 3.0, with the formula for the sample size for comparing two means. The value of the confidence interval (two-sided) was 99%, power 90%, and the ratio of sample size (treatment group/control group) was 1. Subjects received an envelope containing the number 1 or 2, there was blinding method and the health center staff assisted this [6]. The number 1 meant the treatment group, whereas the number 2 meant the control group. There were 110 envelopes in total. Eligible research subjects, who had signed the informed consent form, picked up the envelope that was distributed randomly. Each group consisted of 50 patients. Group ILA (n = 50) received 2.5 mg bupivacaine + 50μg clonidine +0.1 mg morphine and the control group (n = 50) did not receive any treatment. Neuraxial ILA was performed by anesthesiologist at Moewardi hospital, while the control group is at health centre because of limited basic monitoring device. In the treatment group, 5 cc of blood was drawn from each subject before ILA (2 cm dilatation), then subjects were given minimal ILA at the time of 4 cm dilatation using 0.1 mg morphine + 2.5 mg bupivacaine + 50 μg clonidine via the spinal anesthesia procedure with a 27G spinal needle. After 5 min, about 5 cc of blood was taken after ILA (8 cm dilatation). Samples for neonatal lactate levels were taken from the umbilical artery. In the control group, the 5 cc of the subjects’ blood was drawn at 2 cm and 8 cm dilatation. Blood samples were put into a tube containing ethylenediaminetetraacetic acid (EDTA), were centrifuged, and stored in serum/plasma form. The sample tubes were stored at −20°C in a thermos filled with ice gel, and then sent to the Clinical Pathology Laboratory Dr. Moewardi Hospital, Surakarta to determine the levels of eNOS, TNF-α, IL-6, and neonatal lactate using Wilcoxon test and Mann–Whitney test.
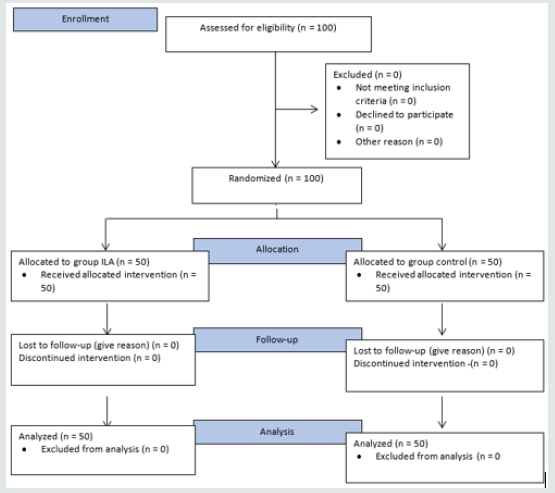
The inclusion criteria in this study were pregnant women who wish to give birth normally in the predetermined hospital and Puskesmas, willing to be a research participant, and sign the informed consent, primigravida or multigravida under gravida 4, and have no history of cesarean section. The exclusion criteria of this study were pregnant women who were not expected to be able to undergo standard delivery, have a history of exposure to and/ or are undergoing cytostatic/cytotoxic treatment (chemotherapy, radiation), have a previous concomitant metabolic disorder (hypertension, diabetes mellitus, etc.), and have contraindications to regional anesthesia. The drop out criteria for this study included the following: pregnant women in shock with general weakness; weak perfusion: pale skin, cold, wet, capillary refill time >2 sec, tachycardia, hyperventilation; hypotension: systolic blood pressure <80 mmHg or mean arterial pressure <60 mmHg, cyanosis, oliguria, peripheral vasodilation, anxiety, decreased consciousness; hyperemesis and dehydration; allergic reactions to medication; and apnea (Figure 1).
Results
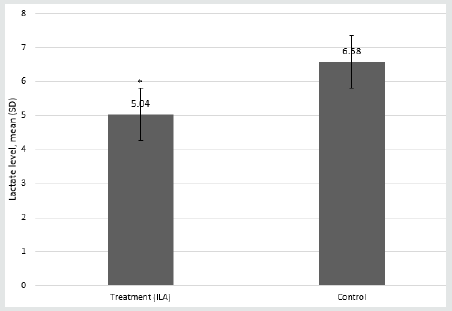
The characteristics of the subject are listed in Table 1 below. In this table, it is shown that the data are homogeneous and have no significant difference that will affect the results of this study. Based on the results of the above study, the pre- and post- levels indicated that there was a significant difference in the change in TNF-α, eNOS, and IL-6 levels. All three parameters decreased in the treatment group but increased in the control group (Figure 2). The lactate levels in the ILA group were lower than those of the control group (Figure 3). The Mann–Whitney statistical test results obtained p = 0.001.
Figure 2: Pre- and post-levels of: (a) tumor necrosis factor-alpha (TNF-α); (b) endothelial nitric oxide synthase (eNOS); (c) interleukin-6 (IL-6). *Wilcoxon test; p = 0.01.
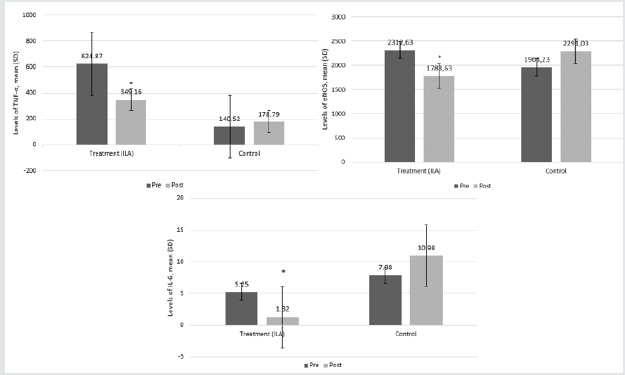
Figure 3: Pre- and post-levels of neonatal lactate. *Mann–Whitney test (p<0.001). ILA = intrathecal labor analgesia
Discussion
The use of ILA causes NOS synthesized through arginine to increase. In pregnant women, there is an increased expression of eNOS compared with non-pregnant women. In chronic hypoxic conditions, there is a decreased expression of eNOS, but increased expression of neuronal NOS (nNOS). This suggests that a normal fetal response to chronic hypoxia can be maladaptive because at least the eNOS-derived NO may prolong the duration of ischemia, whereas an increase in nNOS may be cytotoxic by the increased release of excitatory amino acids [7,8]. Decreased eNOS results in decreased NO production leading to the vasoconstriction of blood vessels. If this vasoconstriction is persistent or long-lasting, it will decrease tissue oxygenation and trigger anaerobic metabolism. Since anaerobic metabolism only produces 2 ATP, uterine contractions are weakened due to the lack of ATP [9,10]. This study showed the decrease of eNOS level, which was caused by eNOS half time has a short (4 sec) half time after extracted from blood, yet the duration between the primary health care and hospital’s laboratory was up to an hour, to analyze the sample.
This increase results in better vascularization and oxygenation of the blood vessels, resulting in increased aerobic metabolism, and increased ATP production. Also, uterine contractions increase in strength, and labor becomes faster. The normal concentration of lactate in the blood ranges from 0.3 to 1.3 mmol/liter. The concentration of lactate measured in the plasma indicates a balance between the production and metabolism of lactate. Neuraxial ILA will block the peripheral nervous system so that pain and stress are reduced. The sympathetic nerves will become vasodilated, so adrenaline and noradrenaline decrease. This decrease will cause aerobic metabolism in the umbilical artery, so that lactate levels in patients with ILA are lower than those in the control group, as seen in (Figure 3) [7]. Tumor necrosis factor-alpha (TNF-α, or cachexin, or cachectin) is a signal cell protein (cytokine) that is involved in systemic inflammation and is one of the cytokines comprising an acute phase reaction. Increased TNF-α will lead to the increased expression of adamantol adhesion intercellular adhesion molecule 1 and vascular cell adhesion molecule 1 in endothelial cells. This increased expression results in a change in the balance between vasodilator and vasoconstrictor molecules resulting in general vasospasm and decreased perfusion of some organs. Pregnant women may experience the infection in urinary tract which fungal and bacterial infection as the source of infection, this may affect the baseline of TNF-α in both groups. The sympathetic nerves will trigger vasodilatation, and both adrenaline and noradrenaline levels will decline, causing a decrease in TNF-α [7,8] ILA will block the pain at the transmission level, but TNF-α and reactive oxygen species (ROS) production will also decline. Asymmetric dimethylarginine, which was initially damaged by ROS, will increase in number, and cause NO production to increase. In addition, the vasculature that originally constricted will return to normal or even dilate. Early anaerobic metabolism will return to aerobic metabolism as perfusion of the uterus improves, and the amount of ATP for uterine muscle contraction increases so that the progress of labor can return to normal [9,10].
The results in (Figure 2c) show that there was no significant difference in IL-6 levels; in the ILA group, levels of IL-6 decreased, whereas, in the control group, IL-6 levels increased. It is known that ILA administration could decrease IL-6 levels in female patients. IL-6 is a driver of the immune system. There is evidence to suggest that IL-6 plays an important role during the transition period from congenital to acquired immunity. Acute inflammation is characterized by the early infiltration of neutrophils. These neutrophils are subsequently replaced by monocytes and T cells after 24 to 48 hours to prevent increased tissue damage from the accumulation of the neutrophil-secreted protease and the ROS at the site of inflammation [11,12] The pain mechanism felt by the patient will increase the stress to the cerebral cortex, then to the hypothalamus. Then, the corticotropin-releasing hormone will be released, increasing adrenocorticotropic hormone, so that cortisol also increases, suppressing the sympathetic nerves, and increasing adrenaline and noradrenaline. This will trigger macrophages to affect the rise of TNF-α, IL-6, and angiotensin II type 1 receptors (AT1R). TNF-α will stimulate endothelial, e-selectin, and polymorphonuclear leukocytes. IL-6 will stimulate hepatocytes, CRP, and endothelial dysfunction. When neuraxial ILA blocks the peripheral nervous system, both pain and stress are reduced. The sympathetic nerves will become vasodilated, so adrenaline and noradrenaline decrease. This decrease will lead to a reduction in macrophages, TNF-α, IL-6, AT1R, and NADPH oxidase enzymes.
Through this study, the authors hope to expand the knowledge about biomolecular changes in pregnancy and what can be altered by anesthetic modalities to lessen the pain of a woman in labor. Clinically, these biomolecular changes were associated with faster cervical dilatation rates, fewer delays in labor progress, and less significant variation in fetomaternal hemodynamics [13] The limitation of this study is its small sample size, with only 50 women in each group, so there is the possibility of a false-negative result. The short duration of the study is also a limitation of the study. It would be better to conduct another study with a larger sample size. From this study, in which the data was homogeneous, the authors concluded that ILA administration could decrease eNOS production in pregnancy. ILA could also decrease the levels of TNF-α, IL-6, and neonatal lactate, which could shorten labor duration (from 522.66 to 363.76 min) [14,15].
Conflicts of Interest
The authors affirm no conflict of interest in this study.
Acknowledgment
None
Funding Sources
None
References
- Moncada S (2006) Adventures in vascular biology: a tale of two mediators. Philos Trans R Soc Lond B Biol Sci 361(1469): 735-759.
- Bullarbo M, Orrskog ME, Andersch B, Granström L, Norström A, et al. (2007) Outpatient vaginal administration of nitric oxide donor isosorbide mononitrate for cervical ripening and labor induction post term: a randomized controlled study. Am J Obstet Gynecol196(1): 1-5.
- Chen X, Qian X, Chen H, Dong M (2008) Serum level of nitric oxide metabolites during labour with or without combined spinal-epidural analgesia. Eur J Anesthesiol 25(9): 708-713.
- Jung H, Kwak KH (2013) Neuraxial analgesia: a review of its effects on the outcome and duration of labor. Korean J Anesthesiol 65(5): 379-384.
- Cambic CR, Wong CA (2010) Labour analgesia and obstetric outcome. Br J Anaesth 105(Suppl 1): 150-160.
- Ranasinghe JS, Birnbach DJ (2009) Progress in analgesia for labor: focus on neuraxial blocks. Int J Womens Health 1: 31-43.
- Darkwa EO, Djagbletey R, Sottie D, Owoo C, Vanderpuye NM, et al. (2018) Serum nitric oxide levels in healthy pregnant women: a case-control study in a tertiary facility in Ghana. Matern Health Neonatol Perinatol 4: 3.
- Ledingham MA, Thomson AJ, Greer IA, Norman JE (2000) Nitric oxide in parturition. BJOG 107(5): 581-593.
- Haider S, Knöfler M (2009) Human tumour necrosis factor: physiological and pathological roles in placenta and endometrium. Placenta 30(2):111-123.
- Trudinger B, Wang J, Athayde N, Beutler L, Wang X (2002) Association of umbilical placental vascular disease with fetal acute inflammatory cytokine responses. J Soc Gynecol Investig 9(3): 152-157.
- Erta M, Quintana A, Hidalgo J (2012) Interleukin-6, a major cytokine in the central nervous system. Int J Biol Sci 8(9): 1254-1266.
- Scheller J, Chalaris A, Schmidt-Arras D, Rose-John S (2011) The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim Biophys Acta 1813(5): 878-888.
- Mathur P, Jain N, Prajapat L, Jain K, Garg D, et al. (2017) Effect of intrathecal labor analgesia using fentanyl 25 μg and bupivacaine 2.5 mg on progress of labor. J Obstet Anaesth Crit Care 7(1): 47-51.
- Ray PD, Huang BW, Tsuji Y (2012) Reactive oxygen species (ROS) homeostais and redox regulation in cellular signaling. Cell Signal 24(5): 981-990.
- Poniedziałek-Czajkowska E, Mierzyński R, Szymula D, Leszczyńska-Gorzelak B, Oleszczuk J (2016) Intercellular adhesion molecule and endogenous NOS inhibitor: asymmetric dimethylarginine in pregnant women with gestational diabetes mellitus. J Diabetes Res 1342643.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...