Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4676
Research Article(ISSN: 2637-4676) 
Effect of Triflumuron and Diflubenzuron on Survivability and Developmental Period of Life Stages of Tribolium castaneum (Herbst) Under Laboratory Conditions Volume 3 - Issue 5
Jannatul Ferdous, W Islam* and Selina Parween
- Department of Zoology, Institute of Biological Sciences, University of Rajshahi, Bangladesh
Received: July 10, 2018; Published: July 20, 2018
Corresponding author: W Islam, Department of Zoology, Institute of Biological Sciences, University of Rajshahi, Bangladesh
DOI: 10.32474/CIACR.2018.03.000172
Abstract
The contact action of triflumuron (TFM) and diflubenzuron (DFB) on development and survivability of Tribolium castaneum (Herbst) were studied. The mean developmental period was recorded in different concentrations of both BPUs. Lower percentage of hatching was recorded on TFM compared to DFB and control. TFM was more effective than DFB on the survivability of different life stages. TFM at 0.05ppm provided the highest duration (49 days) of developmental period, and the lowest duration was 32.33 days (near to control) in DFB at 0.005ppm. Percent reduction of adult emergence was highest in 0.1TFM as 85.95 and at 0.05 DFB as 62. 50.
Introduction
The red flour beetle, Tribolium castaneum (Herbst) has had a long association with human’s stored food. It is a major pest in anthropogenic structures used for the processing and storage of grain-based products and a wide range of other commodities [1]. The tropical climate encourages rapid growth of this cosmopolitan species, and the population increases rapidly from a small residue. Due to the development of resistance against almost all synthetic insecticides it is very difficult to control T. castaneum with the traditionally used insecticides. Among the alternative reduced-risk insecticides, the benzoylphenyl urea’s (BPUs) proved themselves as potent candidate in controlling both susceptible and resistant strains of T. castaneum [2]. BPUs act as stomach poisons [3] at longer exposures to larval instars of the beetle.
Development and growth of the life stages of the treated insects have been reported to be inhibited by BPUs [2-4]. Have reported that triflumuron, a BPUs compound possesses some contact activity against the eggs of holometabolan insects. Most of the early reports on the biological activities of triflumuron, diflubenzuron or other BPUs were reported to show gustatory effects on the treated [2]. As moult inhibitors both triflumuron and diflubenzuron have been reported to disturb metamorphosis in a number of stored product insect species [5-11] and death may occur either during or immediately after the subsequent moult [12]. Some reports showed that BPUs also show contact toxicity towards insects but at a lower degree than the gustatory effect [13]. Reports on the effects of contact action of triflumuron and diflubenzuron against the growth and development of stored product coleopterans are scanty. So, the present experiment was designed to observe the contact activities of these two BPU compounds on the survivability of life stages and their developmental time in T. castaneum under laboratory conditions.
Materials and Methods
BPUs used
Triflumuron was used as a liquid product of Bayar AG, commercially known as Starycide 480 SC, containing mg/ml active ingredient. Dimilin with 25% wp of diflubenzuron was the source of diflubenzuron.
Pilot experiments
Pilot experiments were conducted with newly laid eggs of T. castaneum, released on filter papers treated with different concentrations of either triflumuron or diflubenzuron. The concentrations for each BPU compound were determined where egg mortality was nearly zero percent. Zero egg mortality was obtained at >0.1ppm of TFM and >0.05ppm of DFB. Depending on those concentrations, a series of three concentrations for each compound were selected. The concentrations of triflumuron (TFM) were used as 0.1, 0.05 and 0.01 ppm; and those of diflubenzuron (DFB) were used as 0.05, 0.01 and 0.005ppm. The abbreviations used here for the BPUs, were suggested by Grosscurt [14].
Preparations of concentrations
Required quantities of liquid triflumuron and powder diflubenzuron were diluted separately with such volume of distilled water, which gave 5ppm of TFM and DFB. These 5ppm concentrated solutions were used as stock solution. The solutions were shaked vigorously. Then by serial dilution of the stock solution of each compound was made by adding required amount of distilled water. At every step shaking of the solutions were continued to ensure thorough mixing of compounds with water. Thus, ultimately the required concentrations of TFM and DFB were obtained, kept in glass bottles separately keeping the mouth of the bottles made air tight and refrigerated at 40C.
Bioassays
Separate filter papers (9cm) were soaked in different concentration of treated solutions and dried at room temperature for an hour. The treated filter paper was then set in a petri dish of same diameter. Previously collected 24-h old eggs of T. castaneum were released on the filter paper in the petri dish. The petri dish was covered and kept for until the eggs began to hatch. Eggs on the treated filter paper were checked for hatching up to 10 days. The neonates were collected carefully and released in standard food. A treated filter paper with same concentration and compound was placed under the food in the petri dish. The larvae were reared up to the emergence of adults. After every three days the food was changed with a fresh one, but the filter paper was not changed. A similar set of experiment was carried on filter soaked with distilled water only, as a control. All the experiments were conducted at 30±10C in the Control Temperature (CT) room and replicated three times.
Parameters observed
Effect of TFM and DFB were observed on the following biological parameters of T. castaneum:
a) Survivability of life stages of T. castaneum
i. Hatching percentage
ii. Larval survivability up to pupation
iii. Pupal survivability
iv. Adult recovery/emergence
b) Developmental period of life stages of T. castaneum
i. Hatching period
ii. Larval period
iii. Pupal period
Statistical analysis
The effect of TFM and DFB on the survivability of the life stages and their developmental periods were tested by ANOVA and Tukey’s test. The percent reduction of adult emergence in treatments compared to control (PRC) was calculated by the formula provided by Mian [5] as follows:
PRC = 1–Average no. of adult emergence (treatment)
Average no. of adult emergence (control)x 100
Results and Observations
Effects on the survivability of life stages
a) Effect on hatching: TFM showed a concentration related effect on the percentage of average larval hatching (Table 1). At concentration 0.1 and 0.05ppm the percent larval hatching was significantly less than the control (Table 1). Whereas, at 0.01ppm the result was not differed from control. In the contrary DFB concentrations showed a different trend. At lowest concentration (0.05ppm) the larval hatching significantly differed from the control, but at higher two concentrations the effects were similar to the control (Table 1).
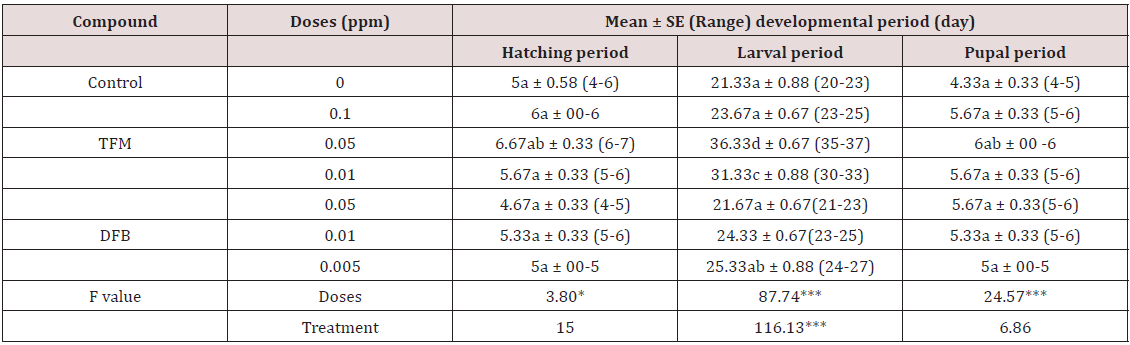
Table 1: Effect of TFM and DFB on survivability at different stages and adult emergence of T. castaneum.
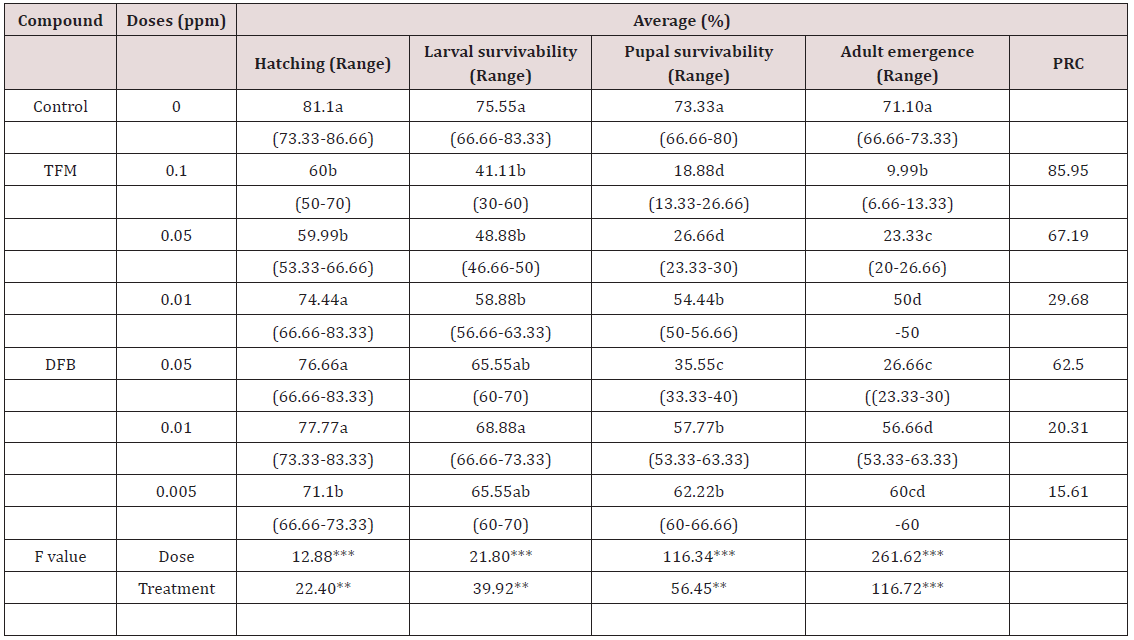
Note: PRC= Percent reduction compared to control in adult emergence.
***Significant at P<0.001, means with same letter do not significantly differed from each other (Tukey’s Test).
**Significant at P<0.05 and ***P<0.001 levels by ANOVA test.
b) Effect on larval survivability: Larval survivability was calculated from the difference between number of larvae hatched and the number obtained before pupation. The cumulative mortality decreased the larval survivability as 75.55% in control. The results showed that both TFM and DFB treatment reduced the percent survivability significantly differently (p<0.00, F=39.92) (Table 1). TFM had a greater mortal effect on the larvae than DFB (Table 1). TFM showed a concentration related larval survivability. Whereas, DFB at highest and lowest concentrations (0.05 and 0.005 ppm respectively) slightly affected larval survivability compared to control, and at concentration 0.01ppm the effect was similar to control (Table 1).
c) Effect on pupal survivability: In control batch out of 75.33% of larvae successfully transformed into pupae. At the end of pupal period 73.33% of the pupae succeeded to survive (Table 1). Both the compound significantly reduced pupal survivability at all concentrations compare to control. TFM was more potential in inhibiting pupal survivability than DFB.
d) Effect on adult emergence: Most of the survived pupae were succeeded to metamorphose into adult. In control the percentage of emergence was 71.10, which was significantly reduced up to 50% at 0.1 and 0.05ppm of TFM and 0.05ppm of DFB. Adult emergence was significantly reduced at all doses of both compounds compared to control. The PRC value ranged from 29.68–85.96 in TFM treatment and from 15.61–62.50 in DFB treatment.
Effects on the developmental periods of the life stages
a) Effect on hatching period: Untreated eggs of T. castaneum took 4-5 (5±0.58) days to hatch. Neither TFM nor DFB produced any on the hatching time difference of the eggs compared to the control (Table 2).
b) Effect on larval period: Both the compounds produced slight increase in larval duration. Interestingly, in the present study the highest concentration of both TFM and DFB produced no significant effect on the larval period (Table 2). TFM at 0.05ppm increased the larval period as 36.33±0.67) days and at 0.01ppm the increment was 31.33±0.88 days; whereas, in untreated control batch the duration was 21.33±0.88 days (Table 2). DFB at higher two concentrations did not produce any significant effect on the larval duration, but at 0.005ppm the period was observed as 25.33±0.88 days (Table 2). TFM produced more severe effect on the developmental time of the larvae than produced by DFM (Table 2).
c) Effect on pupal period: Like the hatching period, TFM at 0.05ppm increased the pupal period more; otherwise in all the treatments the pupae took the same time to develop as took in control batch.
***Significant at P<0.001, means with same letter do not significantly differed from each other (Tukey’s Test)
**Significant at P<0.05 and ***P<0.001 levels by ANOVA test
Discussion
BPU compounds are well known for their larvicidal activity against the young age larvae [2,5,8]. The larvicidal effect of either TFM or DFB as reported by different workers in coleopteran species, were the result of gustatory action of the compounds. Grosscurt [14] reported DFB showed that contact action against the lepidopteran eggs. Fox [3] found slight effect of contact action of TFM and DFB against the eggs of other insects. These observations proved the potentiality of these compounds as avicides. In the present study, TFM at concentration levels of 0.1 and 0.05 ppm resulted in about 40% mortal effects against the egg stage, in untreated eggs 20% of the eggs were failed to hatch. Hatching of eggs of Tribolium in untreated medium may often cause due to the effect of temperature, light and humidity [15,16]. Similarly, exogenous materials also affect growth, survivability and developmental time of life stages of Tribolium [17]. Penetrations of treatment of any type into the eggs are rapid during the first 24 hour. TFM and DFB have been reported to cause embryogenesis if they pass within eggs before blastokinesis stage [14]. So, in the present study inhibition of egg hatching in presence of TFM and DFB is quite possible as a function of their contact action.
BPUs are known to act as larvicides against the early larvae. The mature larvae of Tribolium require higher doses to kill [18]. But, the cumulative mortality in the larvae was obtained at significantly low doses in the present experiment. The latent effect of both TFM and DFB reduced the survivability rate at pupal and adult stages of T. castaneum. Longer exposures to BPU compounds have been reported to decrease the emergence of adult Tribolium species [6,19-21,22] and in other species [5,23,24]. In the present study adult emergence was reduced up to 85% in treatment compared to control.
As BPUs inhibit developmental process in the treated insects, longer exposure to these compounds therefore increased effectively the developmental period in insects. In the present experiments the embryonic developmental time and adult developmental were not much affected due to the BPU treatment, whereas the larval developmental time was significantly increased, especially with TFM treatment. Interstingly, highest doses of both TFM and DFB affected the larval period at lesser degree than the other two lower doses. As TFM and DFB are hormone analogues so, they need a definite amount at definite time of the insects’ life to produce effective results. This might be the reason, why the higher concentrations failed to effectively inhibit development in the larval stages of T. castaneum. Such information is lacking in the published reports, so this finding cannot be explained.
Conclusion
The results of this experiment can be concluded as both TFM and DFB are potent candidate as alternative reduced-risk insecticides against T. castaneum. So, the compounds will provide effective control against other stored-products insects pests. Because these compounds inhibit effectively the development and survivability at egg to pupal stages of the holometabolan insects [3].
References
- Campbell JF, Runnion C (2003) Patch exploitation by female red flour beetles, Tribolium castaneum. J Insect Sci 3(1): 20.
- Mondal KAMSH, S Parween (2000) Insect growth regulators and their potential in the management of stored product insect pests. Integrated Pest Manage Rev 5: 255-295.
- Fox D (1990) Insect Growth Regulators. PJB Publ Ltd. Richmond, UK, pp. 108.
- Hamman I, Sirrenberg W (1980) Laboratory evaluation of SIR 8514, a new chitin synthesis inhibitor of the benzoylated urea class. Pflanzenschutz-Nachrichten Bayer 33(1): 1-34.
- Mian LS, MS Mulla (1982) Biological activity of IGRs against four stored product coleopterans. J econ entomol 75(1): 80-85.
- Eisa AA, Ammar IMA, Radwan HSH, El Nabawi A (1984) Larvicide activity and Tribolium castaneum (Herbst) feed on insect growth regulators treated wheat flour. Minnfiya J Agric Res 8: 405-414.
- Ishaaya I,Yablonski S (1987) Toxicity of two benzyolphenyl ureas against insecticide resistant mealworms. In Chitin and benzyolphenylureas (JE Wright, Retnakaran A eds.). Dr W Junk Publ The Netherlands, pp. 1330140.
- Parween S (1996) The effcet of Triflumuron on Malathion-susceptible (FSS II) and malathion-resistant (CTC 12) strains of Tribolium castaneum (Herbst), Ph D Thesis, University of Newcastle upon Tyne, UK, pp. 229.
- Mazid MA (2000) Effect of Triflumuron on the confused flour beetle, Tribolium confusum Duval (Coleoptera: Tenebrionidae). Unpublished Ph D Thesis, University of Rajshahi, Bangladesh, pp. 279.
- Hasnat H (2003) Combined action of Dimilin and deltamethrin on Tribolium castaneum (Herbst). Ph D Thesis, University of Rajshahi, Bangladesh, pp. 273.
- Hasnat H, MA Mazid, KAMSH Mondal, W Islam, S Parween (2003) Effect of insect growth regulators on the population growth of Tribolium species. J Sci Foundation 1(2): 93-98.
- Retnakaran A, Granett J, Ennis T (1985) Insect growth regulators. In Comprehensive insect physiology, Biochemistry and Pharmacology (GA Kerkut, Gilbert LI eds). 15: 529-601.
- Mulder R, Gijiswijt M J (1973) The laboratory evaluation of two promising new insecticides which interfere with cuticle deposition. Pestic Sci 4(5): 737-745.
- Grosscurt AC (1978) Diflubenzuron: some aspects of its ovicidal and larval mode of action and an evaluation of its practical possibilities. Pestic Sci 9(5): 373-386.
- Howe RW (1956) The effects of temperature and humidity on the rate of development and mortality of Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Ann Appl Bio 44(2): 356-368.
- Khalifa A, Badawy A (1955) Biological studies on Tribolium confusum Duval, Tribolium castaneum Herbst. and Latheticus oryzae Waterh (Coleoptera: Tenebrionidae). Bull Roy Soc Ent Egypte 39: 351-373.
- Sokoloff A (1972) The biology of Tribolium. Vol. 1 Oxford University Press, UK, pp. 300.
- Kamaruzzaman AHM, Mazid MA, Parween S, Mondal KAMS, Islam W (1999) Dose-mortality responses of the flour beetles to triflumuron and cyromazine. Tribolium Inf Bull 39: 298-307.
- Mondal KAMSH, Parween S, Reichmuth Ch, Akter N (1999) Effect of triflumuron on the development of the red flour beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). 7th Inter Work Conf on Stored Product Protection, Beijing, China, 1: 933-939.
- Begum M, Faruki SI, Mondal KAMSH, Parween S, Islam W, et al. (2000) Effect of egg treatment with Triflumuron on the development of Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). J bio sci 8: 7-12.
- Parween S (2003) Embryocidal and larvicidal effect of triflumuron against the red flour beetle, Tribolium castaneum Herbst (Coleoptera:Tenebrionidae). Int Pest Contr 45(6): 329-332.
- Salokhe SG, Pal JK, Mukherjee SN (2003) Effect of sublethal concentrations of flufenoxuron on growth, development and reproductive performance of Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Invertebrate Reproduction & Development 43(2): 141-150.
- Rup PJ, Chopra PK (1984) Effect of hydroprene on Callosobruchus maculatus (F.) (Coleoptera: Bruchidae). J Stored Prod Res 20(4): 229- 232.
- Parween S (2000) Baycidal: effect on Tribolium castaneum Herbst (Coleoptera: Tenebrionidae) population. Entomon 25(1): 25-28.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...