
Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2643-6760
Case Report(ISSN: 2643-6760) 
Giant Mesenchymal Hamartoma in Pediatric Patients Volume 5 - Issue 3
Abdelhak Lamara1, Karim Boujema2, Amel Soualmia3, Badreddine Nini1, Sid Ahmed Medjahdi1, Samira Blel4, Adlene Saadna5 and Hafida Menzer6
- 1General Surgery. Regional Military University Hospital of Constantine /5RM Algeria
- 2Surgery Pontchaillou Hospital. Rennes University France
- 3National Transplant Coordination. Algeria
- 4Intensive Care. Regional Military University Hospital of Constantine /5RM Algeria
- 5Patholgy Regional Military Hospital of Constantine /5RM Algeria
- 6Pediatric and infant unit. Regional Military University Hospital of Constantine /5RM Algeria
Received: July 04, 2020; Published: July 24, 2020
Corresponding author: Abdelhak Lamara, Head of General Surgery Regional Military Hospital Abdelaali BENBAATOUCHE - Constantine / 5 RM, BP: 61 C, 25001 - Constantine - Algeria
DOI: 10.32474/SCSOAJ.2020.05.000213
Abstract
Mesenchymal hamartoma of the liver is a benign tumor with good prognosis that usually manifests in childhood before the age of two. Abdominal distension and the presence of a hepatic mass are the most common symptoms; anorexia, vomiting and signs of compression have been reported. The diagnosis is based on radiological data but above all on the results of the pathology. Only surgical resection can prevent recurrences and in some cases, liver transplantation is indicated. We report the case of a 16-monthold girl who was referred to as for surgical assessment of cystic mass in the right liver, with a respiratory recurrent symptomology. Imagery (CT scan MRI) has showed a large hepatic mass which compresses the hepatic pedicle and the vena cava. We performed a right hepatectomy after clamping the hepatic pedicle and the inferior vena cava. The postoperative follow-up were simple and disappearance of the preoperative symptoms.
Keywords:Mesenchymal hamartoma, Infant, Right Hepatectomy
Case Report
A 16 month old infant was referred to us for a surgical
management of a large abdominal mass. The parents revealed that
this infant was born from an uneventful pregnancy, weighing 3 kg
at birth, breastfed up to the 4th month. The newborn was under
inhaled treatment for bronchial asthma. At the age of 12 months
and when hospitalized in a pediatric ward for an unexplained
fever and abdominal distension, the ultrasound showed a
heterogeneous cystic mass of the liver. The blood tests revealed
anemia, hemoglobin electrophoresis showed heterozygous sickle
cell anemia, hemoglobin S level at 38%. Clinical examination
on admission, found a eutrophic infant, body weight and height
were 10kg and 75cm. The patient had slight skin-mucous pallor,
significant abdominal distension with, collateral circulation,
palpable mass reaching the right flank and crossing the middle line,
no splenomegaly, and no jaundice or haemorrhagic syndrome.
The initial blood tests was normal: White blood cells: 12,000,
Hemoglobin: 9.4g / dl, Haematocrit: 28.7%, Platelets: 42000UL:
aspartate transaminase 52U / L; alanine transaminase: 30U /
L, GGT: 49U / L // Total Bilirubin 0.35 mg / dl, direct bilirubin
0.15mg / dl). Abdominal CT scan showed a heterogeneous mass
of the liver (segments V, VI, VII and VIII). Multiple partitions
enhanced by the contrast agent, oval with regular contours and
sharp limits measuring 95X66X100 mm, compression the hepatic
veins, the portal vein and the right kidney. MRI showed a large
multilocular cystic mass involving the right liver, without signs
of vascular invasion (Figure 1). Doubtful hydatid serology is an
absolute contraindication for a biopsy puncture. In the presence
of a large cystic mass of the right liver, compressing the hepatic
pedicle, and the inferior vena cava, the strategy was to perform a
right hepatectomy.
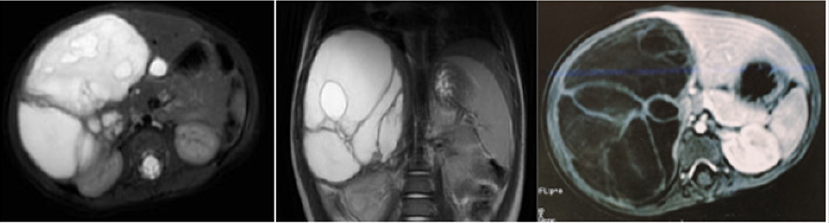
Figure 1: A. Abdominal CT scan showed a heterogeneous mass of the liver (segments V, VI, VII and VIII). Multiple partitions
enhanced by the contrast agent, oval with regular contours and sharp limits measuring 95X66X100 mm, compression the
hepatic veins, the portal vein and the right kidney.
B. MRI showed a large multilocular cystic mass involving the right liver, without signs of vascular invasion.
After the Pringle maneuver, we have done the following: approach to the inferior vena cava below the liver, individualization of the bile ducts, clamping of the hepatic pedicle and inferior vena cava (total clamping time 08 minutes). A right hepatectomy was performed after ligation of the artery right hepatic, right portal vein, right bile duct, and right hepatic vein. (Figure2) The duration of the surgery was 45 minutes; the loss of blood was estimated at 30ml, no transfusion, wall closure without drainage. The post-operative in the hepatobiliary unit was simple; the patient was released after 72 hours. The hepatic enzymes were high in the immediate postoperative phase and became stable in two weeks. The pathology confirmed the diagnosis of mesenchymal hamartoma (Figures 2B & 3). The last checkup was carried out at 09 months and the evolution of the patient was favorable. Respiratory signs disappeared, and there was a significant improvement in the patient’s behavior who has become more active to the satisfaction of her parents.
Figure 2A: Operative Procedure: Right Hepatectomy.
Figure 2B: Hepatic Tumors 16x11x7cm Giant Pseudocystic Mesenchymal Tumors (14x10 cm).
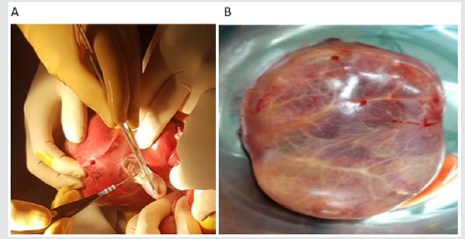
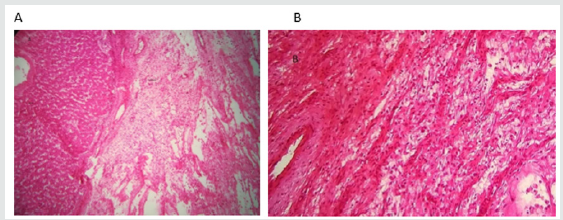
Figure 3: A. Hepatic parenchyma sits from a well-separated tumor formation by a fibrous pseudo-capsule.
B. HM: Low-atypical fusiform cell ranges settle on a myxoid background punctuated by mononucleous inflammatory
elements and congestive blood vessels. The hepatocytic epithelial contingent is minimal.
Discussion
Mesenchymal hamartomas (HM; also known as pseudocystic
mesenchymal tumor, giant cell lymphangioma, bile cell
fibroadenoma, and cavernous lymphagiomatoid tumor) of the liver
represent the second common benign tumor in children, especially
in girls, and is often discovered during the first two years [1,2]. The
etiology of MH has not yet been determined. One theory suggests a
developmental abnormality that stems from a malformation of the
ductal plate, while another theory that is gaining ground, proposes
that MH is a true neoplasm. Recent cytogenetic studies have shown
balanced translocations involving chromosomes 11, 13, 15 and
19; flow cytometry studies have revealed aneuploidy [3-5]. MH
are often asymptomatic, discovered during a routine radiological
examination or for other pathology in the form of a cystic mass.
Abdominal distension and the presence of hepatic mass are the most
common symptoms [6,7]; cases of anorexia, vomiting and signs of
compression such as jaundice and even in some cases compression
of the vena cava have been reported [2]. Sometimes the mass will
quickly increase in volume due to the rapid accumulation of fluid in
the cystic spaces. Rare but often serious respiratory complications
are responsible for asthma attacks and respiratory distress. About
75% of MH in the liver develops in the right lobe. Liver function
and Alpha Foetoprotein (AFP) are only useful for excluding other
diseases such as hepatoblastom [6-9].
The imagery highlights a large, well-circumscribed mass that
may contain cysts of different sizes. Ultrasound and computed
tomography show a multicystic mass with septations as in the
case of our patient [10-14]. It is often question of very large liver
lesions either from the inside or from the growth of the normal
parenchyma, in most cases associating several cysts of 4 to 7 cm
each, containing clear to yellow liquid and solid areas , consisting of
loose myxoid stroma containing irregularly dilated ducts bordered
by cuboid epithelia.
Often there are irregular islets separated from hepatocytes.
The cysts are surrounded by a layer of loose mesenchyme with
no identifiable epithelial lining and appear to be linked to the
mesenchyme itself [15]. The myxomatous stroma contains doubled
epithelial and non-epithelial cysts [1]. If there is any doubt about
the possibility of a malignant tumor, a biopsy can be performed, as
such, a diagnosis can lead to a change in the management of larger
tumors which might otherwise be difficult to resect [9,10].
The practice of observation alone while awaiting spontaneous
regression should be discouraged in view of reports of malignant
transformation into undifferentiated embryonic sarcoma [15-18].
The risk of malignant transformation into indifferent sarcoma
implies a systematic surgical resection [15,16]. Only surgical
resection can prevent recurrences. The treatment consists of
complete surgical resection with clear margins which may require
a lobectomy or a non-anatomical resection [6,7]. If the tumor
is considered unrespectable, some authors have recommended
nucleation or marsupialization [7,19,20]. The insufficiency of the
remaining liver is considered to be the main reason for the failure of
the surgery [19]. Some authors have demonstrated the feasibility of
ALPPS in infants with a huge hepatic tumor, especially mesenchymal
hepatic hamartoma, due to the rapid regeneration of the future
remaining liver [21]. Others believe that liver transplantation is a
safe surgical option for pediatric patients with unresectable giant
mesenchymal hamartoma, who generally have no other therapeutic
alternative [22].
References
- Stringer MD, Alizai NK (2005) Mesenchymal hamartoma of the liver: a systematic review. J Pediatr Surg 40: 1681-1690.
- Meyers RL (2007) Tumors of the liver in children. Surg Oncol 16: 195-203. Bove KE, Blough RI, Soukup S. Third report of t (19q) (13.4) in mesenchymal hamartoma of liver with comments on link to embryonal sarcoma. Pediatr Dev Pathol 1: 438-42.
- Murthi GV, Paterson L, Azmy A (2003) Chromosomal translocation in mesenchymal hamartoma of liver: what is its significance? J Pediatr Surg 38: 1543-1545.
- Rajaram V, Knezevich S, Bove KE (2007) DNA sequence of the translocation breakpoints in undifferentiated embryonal sarcoma arising in mesenchymal hamartoma of the liver harboring the t(11;19) (q11;q13.4) translocation. Genes Chromosomes Cancer 46: 508-513.
- Murray JD, Ricketts RR (1998) Mesenchymal hamartoma of the liver. Am Surg 64: 1097-1103.
- Yen JB, Kong MS, Lin JN (2003) Hepatic mesenchymal hamartoma. J Paediatr Child Health 39: 632-634.
- Unal E, Koksal Y, Akcoren Z (2008) Mesenchymal hamartoma of the liver mimicking hepatoblastoma. J Pediatr Hematol Oncol 30: 458-460.
- Al-Rikabi AC, Buckai A, al-Sumayer S (2000) Fine needle aspiration cytology of mesenchymal hamartoma of the liver. À case report. Acta Cytol 44: 449-453.
- Jimenez-Heffernan JA, Vicandi B, Lopez-Ferrer P (2000) Fine-needle aspiration cytology of mesenchymal hamartoma of the liver. Diagn Cytopathol 22: 250-253.
- Ros PR, Goodman ZD, Ishak KG (1986) Mesenchymal hamartoma of the liver: radiologic-pathologic correlation. Radiology 158: 619-624.
- Stanley P, Hall TR, Woolley MM (1986) Mesenchymal hamartomas of the liver in childhood: sonographic and CT findings. AJR Am J Roentgenol 147: 1035-1039.
- Koumanidou C, Vakaki M, Papadaki M (1999) New sonographic appearance of hepatic mesenchymal hamartoma in childhood. J Clin Ultrasound 27:164-167.
- Ye BB, Hu B, Wang LJ (2005) Mesenchymal hamartoma of liver: magnetic resonance imaging and histopathologic correlation. World J Gastroenterol 11: 5807-5810.
- Stocker JT, Ishak KG (1983) Mesenchymal hamartoma of the liver: report of 30 cases and review of the literature. Pediatr Pathol 1: 245-267.
- De Chadarevian JP, Pawel BR, Faerber EN (1994) Undifferentiated (embryonal) sarcoma arising in conjunction with mesenchymal hamartoma of the liver. Mod Pathol 7: 490-493.
- Ramanujam TM, Ramesh JC, Goh DW (1999) Malignant transforma- tion of mesenchymal hamartoma of the liver: case report and review of the literature. J Pediatr Surg 34: 1684-1686.
- Barnhart DC, Hirschl RB, Garver KA (1997) Conservative management of mesenchymal hamartoma of the liver. J Pediatr Surg 32: 1495-1498.
- Narasimhan KL, Radotra BD, Harish J (2004) Conservative management of giant hepatic mesenchymal hamartoma. Indian J Gastroenterol 23: 26.
- Alvarez FA, Ardiles V, de Santibañes M, Pe- kolj J, de Santibañes E (2018) ALPPS for hepatic mesenchymal hamartoma in an infant. Journal of Pediatric Surgery Case Reports 37: 70-73.
- Karpelowsky JS, Pansini A, Lazarus C (2008) Difficulties in the management of mesenchymal hamartomas. Pediatr Surg Int 24(10): 1171-1175.
- Zhi-Lin Xu, Long Wang, Wei Fan, Zeng-Hui Hao, Chao Li (2018) ALPPS for hepatic mesenchymal hamartoma in an infant Journal of Pediatric Surgery Case Reports 37: 70-73.
- Selby R, Webb M (1995) Orthotopic liver transplantation for benign hepatic neoplasms. Arch Surg 130: 153-156.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...




