
Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1709
Research Article(ISSN: 2641-1709) 
Negative Pathology Report Following Salivary Gland Surgery for Suspected Primary Tumor– What Went Wrong? Volume 7 - Issue 2
Ohad Cohen1*, Chanan Shaul1, Victoria Doviner2, Yoav Sherman2 and Jean-Yves Sichel1
- 1Department of Otolaryngology, Head and Neck Surgery, Shaare Zedek Medical Center, Israel
- 2Department of Pathology, Shaare Zedek Medical Center, Hebrew University Medical School, Israel
Received:September 01, 2021; Published:September 09, 2021
Corresponding author: Ohad Cohen, Department of Otolaryngology, Head and Neck Surgery Shaare Zedek Medical Center, Shmu’el Bait St 12, Jerusalem, Israel
DOI: 10.32474/SJO.2021.07.000262
Abstract
Objective: For patients undergoing an oncologic surgery, postoperative pathological diagnosis negative for a tumor is a confusing outcome. Additionally, it may carry clinical and medicolegal consequences. The study defines the causes of such discrepancies in order to prevent such instances in the future.
Methods: A retrospective cohort study of patients who had undergone resection of a major salivary gland for a suspected or diagnosed primary tumor but had no tumor on surgical pathology.
Results: Eight patients (2.5%) had negative pathology. Causes for negative pathology were A) Surgical pathology error (n=3) B) Surgical management error (n=1) C) Surgery for definite diagnosis (n=2) D) Unexplained (n=2).
Conclusions: Negative pathology in salivary gland surgery is not rare. Negative pathology should raise the suspicions of both the surgeon and the pathologist. An immediate multidisciplinary review of all data will find the cause in most cases Keywords: Negative pathology; no tumor on pathology; salivary gland tumor; parotid gland tumor
Introduction
Salivary gland neoplasms constitute 3-4% of all head and neck tumors. The benign or malignant nature of the tumor may influence the patient consultation and decision-making process. It could also determine the extent of surgical resection and the ability to preserve the facial nerve. Fine needle aspiration biopsy (FNAB) is often used for preoperative cytological diagnosis. However, Although FNAB is highly specific for malignancy (93%-100%), its sensitivity is not as good (83%-92%) [1-3]. While reviewing our database on patients following salivary gland resection, we noticed a small subgroup (2.5%) who were operated for diagnosed or suspected primary salivary gland tumor, but in whom no tumor was found on surgical pathology (negative pathology). The goals of this study are to review cases of negative surgical pathology in patients who undergo salivary gland resection for suspected salivary tumor, and to define the causes of such discrepancies in order to prevent such instances in the future.
Material and Methods
A retrospective review was performed of patients who had undergone resection of a major salivary gland in the otolaryngology department at Shaare Zedek medical center between 2005 and 2020. Data included: demographics, preoperative FNAB, surgical treatment and complications, surgical pathology results and followup. Cases with negative pathology were reviewed thoroughly, including investigation of the perioperative decision making and follow-up by telephone call when needed. Preoperative cytology and postoperative pathology samples were reviewed by an experienced pathologist (V.D, Y.S). Some patients were referred to us following cytology samples elsewhere; therefore, not all samples were available for reevaluation. This work was approved by the hospital’s ethics committee.
Results
Overall, 312 patients underwent salivary gland resection in this period. FNAB sensitivity and specificity for malignancy were 76% and 93%, respectively. Ten patients (3.2%) had no tumor on surgical pathology. Two were referred to us for a completion surgery following a previous excisional biopsy of a lump in the parotid area by plastic surgeons. Negative pathology in this situation is predictable hence they were excluded from this review. A review of the remaining eight patients revealed the causes for the negative pathology, which can be categorized as follows:
Surgical Pathology Errors
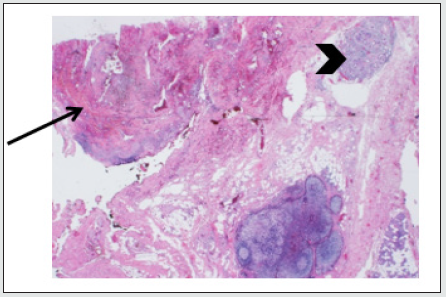
a) Patient no. 1: An 18-year-old woman who had had a preauricular lump for the past year. Ultrasound (US) showed a 2.5 cm hyper-vascularized parotid mass. Computed tomography (CT) and magnetic resonance imaging (MRI) demonstrated “a similar solid mass” (based on records on admission: the original scans were not available). The two FNAB samples showed only blood and foam cells. During surgery the mass was identified and excised completely. Although no tumor was found postoperatively, pathological review of the specimen revealed an arteriovenous malformation (Figure 1) in only one of the original slides.
Figure 1: Arteriovenous malformation (arrow) and adjacent normal parotid gland tissue (arrowhead), H&E stain, original magnification ×12.5.
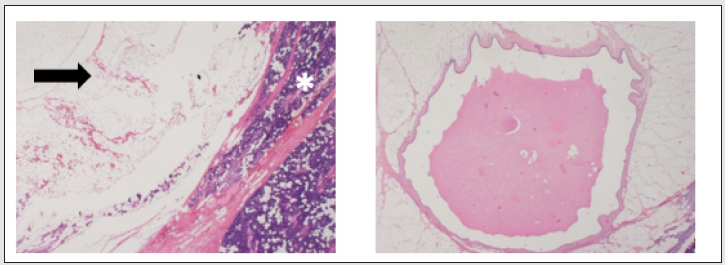
b) Patient no. 2: A 50-year-old man with a parotid swelling. US and CT showed a well-defined, mostly cystic mass. FNAB demonstrated only acinar cells with no tumor. The mass had grown on follow-up visits and was therefore excised. The pathology report noted dilatation of the parotid ducts and “lipomatous infiltrations”, but no tumor. However, review of the histology found an intraparotid sialolipoma with secondary obstructed and dilated ducts (Figure 2).
Figure 2: Intra-parotid sialolipoma. Left – lipoma (arrow) within normal parotid (asterisk), H&E stain, original magnification ×12.5; right – dilated excretory duct entrapped in the lipoma, H&E stain, original magnification ×40.
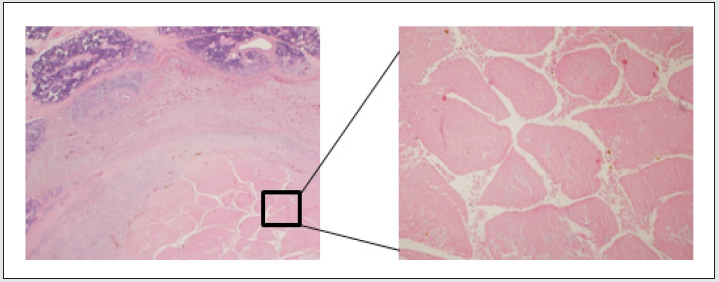
c) Patient no. 3: A 77-year-old man who had had a progressively growing firm mass in the left parotid gland. CT demonstrated a well-defined mass, and FNAB led to a diagnosis of Warthin’s tumor. The mass was palpable during a superficial parotidectomy, but pathological examination showed only necrotic tissue and inflammation. Meticulous review of the slides revealed a necrotic Warthin’s tumor (Figure 3), with the necrosis attributed to the preoperative needle biopsy.
Figure 3: Warthin’s tumor showing extensive ischemic necrosis (resulting from fine needle aspiration), but still preserving its general papillary-cystic architecture. Left - H&E stain, original magnification ×12.5; right - H&E stain, original magnification ×40.
Surgical Management Error
a) Patient no. 4: A 47-year-old man with swelling of the submandibular area. US and CT demonstrated enlarged lymph nodes and an only mildly enlarged submandibular gland. Two sequential FNABs were inconclusive. He was diagnosed with suspicious submandibular gland mass and underwent resection with negative postoperative pathology. Retrospectively, the lymph nodes were the source of swelling, and resection of the salivary gland was probably not indicated. In any case, no recurrence of swelling was reported at the 12-year follow up.
“Rule Out” Surgery - cases in which it was hard to rule out a tumor without surgery
a. Patient no. 5: A 58-year-old man had temporary left facial nerve paresis followed by facial spasm. CT and MRI demonstrated a small, multi-cystic parotid mass, suggesting the differential diagnosis of neoplasm versus lymphatic malformation. FNAB showed “suspicious features” of Warthin’s tumor. In light of the inconclusive findings, it was decided to perform parotidectomy for both diagnostic and therapeutic purposes. The pathology findings were parotid tissue with fibrosis and chronic inflammation. Followup, including US and MRI imaging, was normal, although facial spasm remained unchanged.
b. Patient no. 6: A 45-year-old man with a history of chemoradiation therapy for neck lymphoma had had a parotid mass for the previous 6 months. US and CT demonstrated a 1 cm mass in the superficial parotid lobe, with no neck lymphadenopathy. FNAB demonstrated a group of atypical cells and could not rule out neoplasm, due to the lack of architecture. The pathologic diagnosis was normal parotid tissue. Two years later, the patient had recurrent swelling in the parotid gland area, but there were only a few cells in the FNAB, and the swelling resolved spontaneously. In the 8 years since then, he has been free of symptoms.
Unexplained
a. Patient no. 7: A 66-year-old woman who had had a mass in the parotid gland for some years. US and CT revealed a solid, well defined parotid lesion. Two FNABs led to a diagnosis of pleomorphic adenoma. The pathologic diagnosis was parotid tissue with a few intraglandular reactive lymph nodes; no tumor was identified. Histologic review verified this finding. The FNA cytology was not available for review. She is free of symptoms in the past 8 years.
b. Patient no. 8: A 67-year-old man with submandibular swelling. US and CT showed a mixed cystic-solid mass in the submandibular gland. The first FNAB only revealed “suspected squamous cell carcinoma,” but a second FNAB led to a diagnosis of Warthin’s tumor. Review of the first FNAB led to a conclusive diagnosis of Warthin’s tumor, but surgical pathology found an unremarkable submandibular gland. The patient was lost to followup soon after the operation in 2007.
Discussion
Parotidectomy is a delicate surgical procedure. It is usually done under general anesthesia and involves a significant risk of complications [4]. The failure to find any tumor on surgery might be devastating for both patient and surgeon and could have medicolegal implications, yet no literature was found on this specific scenario. This led us to investigate the possible explanations for such an outcome. The specificity and sensitivity of FNAB in salivary glands has been extensively investigated. A metaanalysis by Schmidt and coworkers found 79% sensitivity and 96% specificity [5]. We found the sensitivity and specificity of our FNAB to be similar to those reported in the literature. Surgical pathology errors, which are not uncommon, usually significantly influence postoperative management. Kronz et al. advocated the systematic review of the pathologic diagnosis of patients referred to a tertiary center, after finding 1%-2% diagnoses to be inaccurate [6]. Peck et al. showed that head and neck pathology have a significantly higher rate of inaccurate pathological diagnosis in comparison to other anatomical sites [7]. Westra at al. found that a second opinion of head and neck surgical pathology changed the diagnosis and resulted in major therapeutic and prognostic modification in 7% of cases, rising to 9% for the salivary gland (and 11% in major glands) [8].
In our study, 3 cases of negative pathology were explained by an inaccurate pathological diagnosis. Patient no. 1 had a vascular malformation, and review of the pathology only found the lesion in one slide. If more slides had been done, it is reasonable to assume that the malformation would have been detected. Nonetheless, the young age, female gender and two FNABs containing only blood cells should have raised suspicions in the patient’s preoperative management. One case of necrotic Warthin’s tumor (patient no. 3) posed a real diagnostic challenge. Although described in the literature [9,10], widespread necrosis of Warthin’s tumor is extremely rare. This diagnosis should be considered in the event of negative surgical pathology after diagnosis of Warthin’s tumor from the FNAB and the presence of classic clinical features (e.g., bilateral parotid mass, smoking history). These two cases also demonstrate the possibility that cystic masses may shrink after excision, as well as the importance of performing more sections on the specimen if the pathology findings are negative. Salivary gland fatty neoplasms (patient no. 2) are rare [11], however familiarity of the expert pathologist with this group of neoplasms may prevent inaccurate diagnosis. Patients 5 and 6 had an unusual course of events prior to their operation. The FNAB for patient 5 was suspicious for Warthin’s, and patient 6 had a history of lymphoma; these were the main indications for surgical resection, even though the FNAB did not definitively diagnose a tumor. But should have we pursued a definitive diagnosis? A study by Romano et al. demonstrated a high diagnostic value with selective use of core needle biopsy as an adjunct to FNAB in specific cases (sensitivity 100% and specificity 92%) [12].
Complications of core needle biopsy are uncommon [13], therefore its utility in cases of uncertainty and its potential to obviate unnecessary surgery may outweigh its risks. Nevertheless, a definite preoperative diagnosis may not always be achievable, and clinical judgment is needed. It is of paramount importance to discuss this issue with the patient beforehand, and it should be completely understood that no tumor may be found postoperatively. In two cases no explanation was found for the negative pathology, and the patients were subsequently lost to follow-up, including the unavailability of their imaging data. Limits of this review include its retrospective design, lack of full medical records and the variability in preoperative evaluation, in particular the imaging modality. Magnetic resonance imaging (MRI) has shown good accuracy in differentiating benign from malignant tumors and is now an accepted method in the preoperative evaluation of salivary gland masses [14]. Currently, almost every such patient in our center is referred to MRI, nevertheless, it was not as common in the early years of this study’s period. would have an MRI changed the outcome in our series? Patients 1 and 5 had an MRI preoperatively. Although MRI may have demonstrated nicely the lipomatous content of the tumor in patient no. 2, it could not prevent the histological misdiagnosis. The same is true in the event of accurate preoperative FNA diagnosis as in the case of patient no. 3. The advantage of MRI in delineating soft tissue may have prevent the wrongful surgery for patient no. 4 and may add useful information in the case of patients 6 and 8. Pleomorphic adenoma has a classic hyperintense signal on T2 sequence, hence, lack of hyperintensity might have raised suspicion even though the FNA diagnosis was unequivocal.
Conclusions
Negative pathology following salivary gland resection should raise the suspicions of both the surgeon and the pathologist. After thorough retrospective investigation, only 0.6% of cases (2/312) remained unexplained. In both unexplained cases, the retrospective investigation was not completed due to lack of medical records and unavailable histology for review of past years. We assume that immediate review would probably have found the cause in these cases too, and therefore stress the need for an immediate multidisciplinary review of all data in the event of negative surgical pathology.
Disclosure of Interest
The Authors declare that there is no conflict of interest.
References
- Stein AP, Britt CJ, Hartig GK (2014) The diagnostic accuracy of fine-needle aspiration for parotid malignancy. Otolaryngol Head Neck Surg 151(1 suppl): 431-436.
- Liu CC, Jethwa AR, Khariwala SS (2016) Sensitivity, specificity, and posttest probability of parotid fine-needle aspiration: a systematic review and meta-analysis. Otolaryngol Head Neck Surg 154(1): 9-23.
- Stewart CJ, MacKenzie K, McGarry GW (2000) Fine-needle aspiration cytology of salivary gland: a review of 341 cases. Diagn Cytopathol 22(3): 139-146.
- Marchese-Ragona R, De Filippis C, Marioni G (2005) Treatment of complications of parotid gland surgery. Acta Otorhinolaryngol Ital 25(3): 174-178.
- Schmidt RL, Hall BJ, Wilson AR (2011) A systematic review and meta-analysis of the diagnostic accuracy of fine-needle aspiration cytology for parotid gland lesions. Am J Clin Pathol 136(1): 45-59.
- Kronz JD, Westra WH, Epstein JI (1999) Mandatory second opinion surgical pathology at a large referral hospital. Cancer 86(11): 2426-2435.
- Peck M, Moffat D, Latham B (2018) Review of diagnostic error in anatomical pathology and the role and value of second opinions in error prevention. J Clin Pathol 71(11): 995-1000.
- Westra WH, Kronz JD, Eisele DW (2002) The impact of second opinion surgical pathology on the practice of head and neck surgery: a decade experience at a large referral hospital. Head Neck 24(7): 684-693.
- Eveson JW, Cawson RA (1986) Warthin's tumor (cystadenolymphoma) of salivary glands. A clinicopathologic investigation of 278 cases. Oral Surg Oral Med Oral Pathol 61(3): 256-262.
- Kern SB (1988) Necrosis of a Warthin's tumor following fine needle aspiration. Acta Cytol 32(2): 207-208.
- Leyva Huerta E, Quezada Rivera D, Tenorio Rocha F (2015) Sialolipoma of minor salivary glands: presentation of five cases and review of the literature with an epidemiological analyze. Indian J Otolaryngol Head Neck Surg 67(Suppl 1): 105-109.
- Romano EB, Wagner JM, Alleman AM (2017) Fine-needle aspiration with selective use of core needle biopsy of major salivary gland tumors. Laryngoscope 127(11): 2522-2527.
- Robertson EG, Baxter G (2011) Tumour seeding following percutaneous needle biopsy: the real story! Clin Radiol 66(11): 1007-1014.
- Stoia S, Băciuț G, Lenghel M (2021) Cross-sectional imaging and cytologic investigations in the preoperative diagnosis of parotid gland tumors - An updated literature review. Bosn J Basic Med Sci 21(1): 19-32.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...




