Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1709
Research Article(ISSN: 2641-1709) 
Epipharyngeal Abrasive Therapy for Patients with Immunoglobulin A Nephropathy: A Retrospective Study Volume 8 - Issue 3
Manabu Mogitate*
- Department of ENT, Mogitate ENT Clinic, Japan
Received: May 16, 2022; Published: May 27, 2022
Corresponding author: Manabu Mogitate, Mogitate ENT Clinic, 1-2-5 Sekiguchi-Daiichi Building 401, Hisamoto, Takatsu-ku, Kawasaki-City, Kanagawa-Prefecture 213-0011, Japan
DOI: 10.32474/SJO.2022.08.000286
Abstract
The incidence of chronic epipharyngitis is high among patients with immunoglobulin A nephropathy (IgAN). The current study aimed to evaluate the efficacy of epipharyngeal abrasive therapy (EAT) for IgAN. This retrospective study assessed the medical records of 21 patients with IgAN who visited my clinic between November 2016 and October 2017. All patients presented with chronic epipharyngitis and received EAT. In total, 19 participants who were treated until the final session were divided into group A (n = 10, those who had combined EAT before and after tonsillectomy and steroid pulse therapy) and group B (n = 4, those whose condition did not improve after tonsillectomy and pulse therapy and n = 2, those with hematuria recurrence after remission). Data about the characteristics of patients and changes in creatinine (Cr) levels, estimated glomerular filtration rate (eGFR), urinary protein levels, and urinary occult blood level before and after EAT were analyzed. There was no significant difference in terms of Cr levels and eGFR before and after EAT. The urinary protein (p < 0.001) and occult blood (p = 0.003) levels significantly differed before and after EAT. Moreover, the duration of urinary occult blood significantly decreased after EAT in group B (p = 0.047). Improvement in hematuria was significantly faster in group B than in group A (p = 0.041). EAT for chronic epipharyngitis can improve hematuria and can be used as additional treatment in patients with IgAN.
Keywords: Chronic epipharyngitis; epipharyngeal abrasive therapy; IgA nephropathy; nasopharynx
Introduction
Immunoglobulin A nephropathy (IgAN) is the most frequent type of primary glomerulonephritis [1]. Moreover, it is currently the second most frequent indication for dialysis after diabetic nephropathy in Japan [2]. Approximately 30%–40% of patients with IgAN who do not receive treatment develop end-stage renal failure [3]. Focal infections of the palatine tonsils are associated with IgAN development, and bilateral palatine tonsillectomy is effective against IgAN. Combined bilateral palatine tonsillectomy and steroid pulse therapy has good clinical outcomes among patients with IgAN [4]. However, despite this treatment, some patients experience residual or relapsing hematuria [5]. Patients with IgAN have a high incidence of chronic epipharyngitis and tonsillitis [6]. Epipharyngeal abrasive therapy (EAT) is effective against IgAN [7,8]. However, only a few studies have reported this notion. Further, which patients should undergo EAT remains unclear. Before EAT can be used as additional treatment for patients with IgAN, its efficacy against IgAN must be validated based on improvement in clinical findings (including hematuria). My outpatient clinic specializes in the treatment of chronic epipharyngitis, and it has a higher number of patients who receive therapy for chronic epipharyngitis than other local clinics and hospitals [9]. About 20 patients with IgA nephropathy chose and receive EAT annually. Acute epipharyngitis is a condition characterized by colds, which is a type of viral infection. It is caused by different viruses, bacteria, and chemicals. Chronic epipharyngitis [10] is a persistent inflammation of the epipharynx, regardless of the cause, which causes different symptoms. Chronic epipharyngitis can be classified as a direct symptom of epipharyngeal inflammation (such as chronic sore throat and throat discomfort), autonomic symptom (including dizziness), or focal disease (such as IgAN and palmoplantar pustulosis) [11].
EAT is a local treatment for chronic epipharyngitis, and it has been used by Horiguchi since the 1960s. EAT has recently been reported as one of the treatment options for patients suffering from long coronavirus disease (COVID) [12,13]. Imai [14] reported that among the symptoms of long COVID, fatigue, headache, and attention disorder relieved by approximately 1 month of EAT. Hagino [15] also reported the effectiveness of EAT on Postural Orthostatic Tachycardia Syndrome (POTS) associated with long COVID symptoms. In addition, Tanaka [16] reported the effectiveness of intranasal sphenopalatine ganglion stimulation (INSPGS), a technique similar to EAT, in treating patients with long COVID. Furthermore, Nishi [17] provided histological evidence of EAT inducing squamous epithelialization of ciliated cells and inhibiting the expression of angiotensin-converting enzyme 2 (ACE2) and transmembrane protease, serine 2 (TMPRSS2), thereby preventing the entry of SARS-CoV-2 into the cells. They explained the mechanism of action of EAT by referring to a report by Hotta [11]. The following three mechanisms of action of EAT were explained:
a) anti-inflammatory (astringent and bactericidal) action of zinc chloride,
b) phlebotomy action, and
c) vagus nerve stimulation action. In 2010, Hotta [18] showed that not only chronic tonsillitis but also chronic epipharyngitis can be a focal infection for IgAN based on the similarity between tonsil and epipharyngeal cells.
Thereafter, EAT was commonly utilized by several otolaryngologists in Japan. In a previous study, Tanaka [19], an otolaryngologist at a clinic, reported a patient treated with zinc chloride abrasive therapy. Further, although tonsillectomy alone was performed, the patient’s urinary findings normalized. Higa [20], an otolaryngologist at a university hospital, showed that EAT had a therapeutic effect in six patients with IgAN with residual hematuria after tonsillectomies who were referred to an otolaryngologist by a nephrologist.
However, in which patients should EAT be performed has not been established yet. The current study investigated the effectiveness of EAT among patients with IgAN who presented with chronic epipharyngitis. Moreover, to evaluate the effectiveness of EAT, patients with IgAN who went to my clinic were further divided into groups according to the treatment provided in the nephrology department. If EAT was found to be effective against IgAN and the urinary findings improved, such treatment can be considered for patients who have no other treatment options and who can only undergo follow-up. Otolaryngologists can facilitate EAT in patients with IgAN, instead of tonsillectomy, and their progress can be monitored. The current study aimed to evaluate the effectiveness of EAT treatment on the blood and urinary findings of patients with IgAN who visited my clinic.
Materials and Methods
Study design/Sample size
In this retrospective study, I evaluated the clinical data of 21 patients with IgAN who visited my clinic for the examination and treatment of chronic epipharyngitis between November 2016 and October 2017. It was conducted in accordance with the Declaration of Helsinki. Since the ethics committee is not located in my clinic, the research plan was submitted to the ethics committee of Ota General Hospital (approval no. 19010). All patients provided informed consent.
Setting/study population
At the initial visit, all patients underwent endoscopic examination. Thereafter, 1% zinc chloride was applied to the epipharynx via the nasal insertion of a cotton swab and the oral insertion of an applicator once per week for at least 12 weeks (Figure 1). The treatment of two patients was discontinued, and the cause was not identified. Therefore, the final analyses included the data of 19 patients. All patients who were evaluated were included, rather than randomly selected, in the analysis. Data about age, sex, presence or absence of referral to my clinic from a nephrologist, previous treatments, chief complaint, and urine occult blood levels were extracted from the medical records. There was no change in urinary occult blood level if it remained at the pretreatment level of 3+. It improved if the level changed with at least one point (e.g., from 3+ to 2+) and negative if the level became negative. If the urinary occult blood level was worse after treatment, it was considered worsening. The estimated glomerular filtration rate (eGFR) and urinary protein and urinary occult blood levels before and after EAT were compared to determine changes in creatinine (Cr) levels in 19 patients who were treated until the final session. Further, the patients were divided into two groups—those who underwent tonsillectomy and received steroid pulse therapy and those who did not received treatments. Then, they were further classified into groups A and B based on the timing of EAT initiation (Table 1). Group A comprised 10 patients with combined EAT before and after tonsillectomy and steroid pulse therapy. Group B included four patients who continually presented with hematuria after tonsillectomy + pulse therapy and two patients with hematuria recurrence after remission.
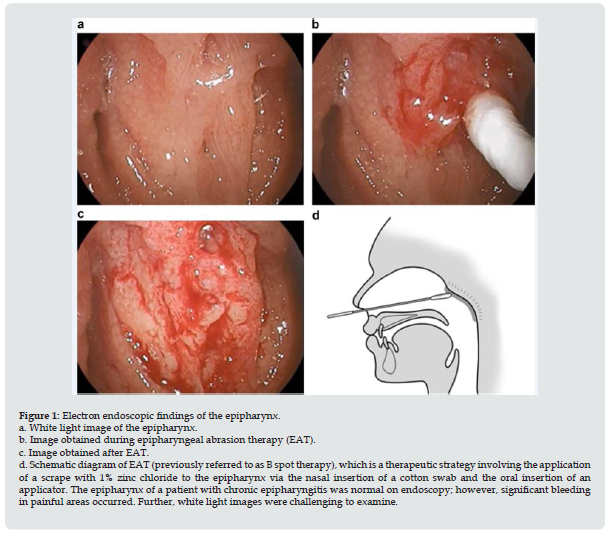
Figure 1: Electron endoscopic findings of the epipharynx. a. White light image of the epipharynx. b. Image obtained during epipharyngeal abrasion therapy (EAT). c. Image obtained after EAT. d. Schematic diagram of EAT (previously referred to as B spot therapy), which is a therapeutic strategy involving the application of a scrape with 1% zinc chloride to the epipharynx via the nasal insertion of a cotton swab and the oral insertion of an applicator. The epipharynx of a patient with chronic epipharyngitis was normal on endoscopy; however, significant bleeding in painful areas occurred. Further, white light images were challenging to examine.
Data collection
The blood and urine test whose data were used in this study were performed by the nephrologist of the facilities where the patients were being followed. The patients then brought their results to my clinic where I analyzed them. A total of 16 and 19 patients underwent blood and urine testing, respectively.
Statistical analysis
Using the Kaplan–Meier method, the changes in urinary protein and occult blood levels were analyzed, and the Wilcoxon signedrank test was utilized to assess changes in eGFR and Cr levels. All analyses were performed using the Statistical Package for the Social Sciences for Windows version 22.0 (IBM Japan, Tokyo, Japan). A p value of <0.05 was considered statistically significant.
Results
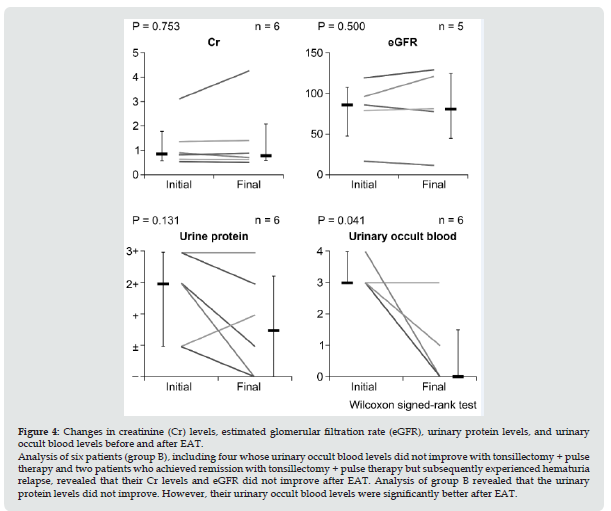
Table 2 shows the characteristics of patients. In total, 19 of 21 patients received treatments until the final session. Two patients dropped out. However, the reasons were unknown. Data were obtained from 16 cases for blood tests and 19 cases for urine tests. The mean age of the patients was 27.8 (range: 16–64) years, and 10 patients were men. All patients were followed-up until the final treatment session. Results showed no statistically significant differences in terms of Cr levels and eGFR before and after EAT (Figure 2). Moreover, based on the statistical analysis, the urinary protein (p < 0.001) and urinary occult blood (p = 0.003) levels significantly improved before and after EAT (Figure 3). The groups were further divided based on the timing of EAT initiation. Table 2 shows the result of the urinary occult blood test. EAT significantly improved urinary occult blood levels in six patients in group B (6.00 ± 3.11) compared with group A (16.9 ± 2.40) (p = 0.047). Of six patients in group B, four had urinary occult blood levels that did not improve with tonsillectomy + pulse therapy. Two achieved remissions with tonsillectomy + pulse therapy. However, they subsequently experienced hematuria relapse. The Cr and urinary protein levels as well as eGFR did not improve after EAT in group B (Figure 4). However, their urinary occult blood levels (p = 0.014) significantly improved.
Figure 2: Analysis based on the Kaplan–Meier method of the negative urinary occult blood. Group A: Ten patients received EAT before and after tonsillectomy + pulse therapy. Group B: Six patients, including four patients with urinary occult blood levels that did not improve with tonsillectomy + pulse therapy and two patients who achieved remission with tonsillectomy + pulse therapy but subsequently experienced hematuria relapse. EAT significantly reduced the duration of urinary occult blood reduction in group B.
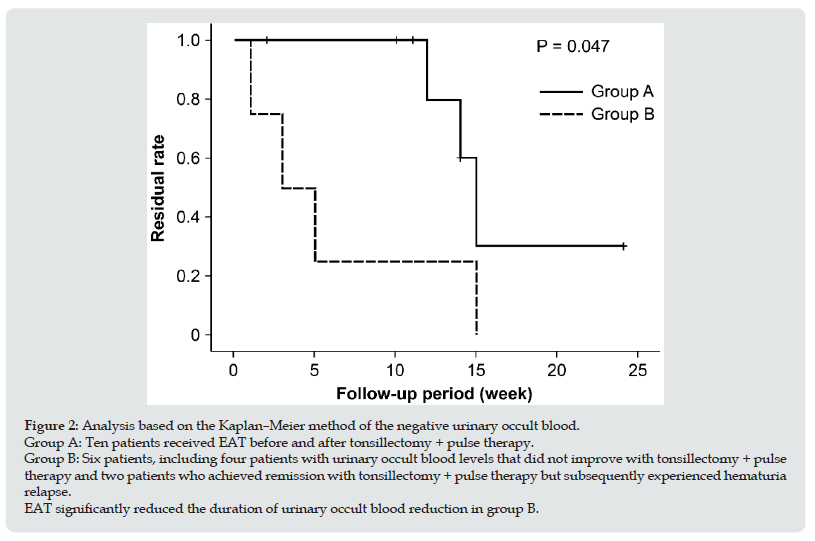
Figure 3: Changes in creatinine (Cr) levels, estimated glomerular filtration rate (eGFR), urinary protein levels, and urinary occult blood levels before and after EAT. Statistical analysis revealed no significant changes in creatinine (Cr) levels or estimated glomerular filtration rate (eGFR) before and after EAT. The urinary protein and occult blood levels improved after EAT, with significant differences observed in 19 patients until the final sessions.
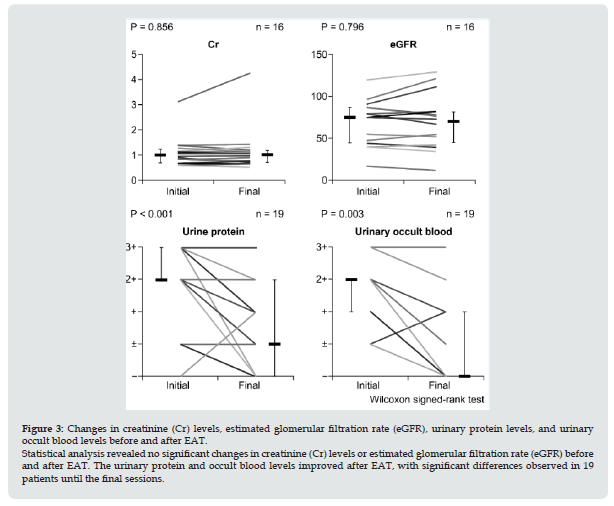
Figure 4: Changes in creatinine (Cr) levels, estimated glomerular filtration rate (eGFR), urinary protein levels, and urinary occult blood levels before and after EAT. Analysis of six patients (group B), including four whose urinary occult blood levels did not improve with tonsillectomy + pulse therapy and two patients who achieved remission with tonsillectomy + pulse therapy but subsequently experienced hematuria relapse, revealed that their Cr levels and eGFR did not improve after EAT. Analysis of group B revealed that the urinary protein levels did not improve. However, their urinary occult blood levels were significantly better after EAT.
Discussion
In November 2016, my clinic developed a specialized outpatient clinic for the diagnosis and treatment of chronic epipharyngitis [9]. Approximately 200 new patients are examined annually, which included 21 with IgAN in the first year. The number of patients with IgAN who chose EAT was higher at my institution than at other local otolaryngological institutions. Therefore, physicians at my clinic have real-world experience in investigating the efficacy of EAT among patients with IgAN. However, to the best of my knowledge, no study about the efficacy of EAT against IgAN has been published in the English literature by otolaryngologists. IgAN is most common in the second to fifth decades of life. According to data from the Japan Renal Biopsy Registry, which was established by the Japanese Society of Nephrology in 2007, the mean ages of patients with IgAN are 37.2 years in men and 35.7 years in women (n = 660) in Japan [21]. The incidence of IgAN does not differ between sexes among Japanese patients 15. These findings are consistent with those of this study. Approximately 40% of patients were referred to my clinic by hospital nephrologists in this study. Nephrologists commonly refer patients with IgAN to otolaryngologists for tonsillectomy. In the future, physicians can make referrals for EAT.
More studies about chronic epipharyngitis are published, and both patients with IgAN and physicians who treat them understand the role of chronic epipharyngitis in IgAN. Several patients present to otolaryngology clinics and choose EAT even without a referral. Therefore, otolaryngologists in hospitals and clinics will manage more patients requesting EAT in the future, and they should be aware of this treatment method. Approximately 80% of patients with IgAN who visited my clinic for EAT were treated with tonsillectomy + steroid pulse therapy. This therapeutic technique is one of the most common treatment options for IgAN in Japan. The 2017 IgAN guidelines state that tonsillectomy + pulse therapy can improve urinary findings in patients with IgAN and inhibits the progression of renal dysfunction. Therefore, it should be considered as a treatment option [22]. If more data showing the efficacy of EAT are collected, it can be recommended to patients with IgAN. All 21 patients who visited my clinic for EAT did not exhibit subjective otolaryngological symptoms. Patients with chronic epipharyngitis commonly have non-specific complaints or localized throat discomfort or postnasal drip. Sugita reported that several otolaryngologists overlook the presence of chronic epipharyngitis even if patients complain of localized symptoms in the epipharynx [23]. Furthermore, chronic epipharyngitis may be missed after an endoscopic examination as the epipharynx is commonly examined while in a chronic inflammatory state [23]. If patients with IgAN visit an otolaryngologist, they do not complain of otolaryngological symptoms; therefore, the epipharynx should be visualized and an abrasive examination of the epipharynx should be performed if chronic epipharyngitis is suspected. EAT is the diagnostic treatment for epipharyngitis, and examinations for bleeding can help diagnose chronic epipharyngitis.
In this study, 21 patients with IgAN who visited my clinic presented with chronic epipharyngitis. The epipharynx is at the forefront of the nasopharynx-associated lymphoid tissues where inhaled antigens from the nostrils are initially encountered. Therefore, this area is physiologically mildly inflamed even among healthy individuals. The exacerbation of glomerular vasculitis in patients with IgAN is associated with inflammation of the acute upper respiratory tract where epipharyngeal inflammation is inevitable and highly inflammatory [18]. After tonsillectomy, patients with IgAN can present with gross hematuria associated with acute pharyngitis. Hotta showed that chronic epipharyngitis caused by chronic irritation by antigens in the epipharynx may play a focal infectious role similar to that in chronic inflammation of the palatine tonsils. Moreover, it may be involved in the immunological effects of the epipharynx–kidney axis on the kidneys and the development of secondary diseases [24]. In this study, 9 of 19 patients who completed EAT had negative urinary occult blood test result after treatment. A previous study [25] assessed 24 patients who underwent EAT. Results showed that hematuria disappeared in 83% of patients. Among them, 19 continually experienced hematuria after combined tonsillectomy and pulse therapy. In addition, this study showed that patients with severe hematuria were treated with additional steroid pulse therapy to reduce inflammation. Moreover, patients with mild hematuria had a high rate of resolution after EAT alone [25]. Persistent urine occult blood in patients with IgAN indicates residual focal inflammation causing glomerular vasculitis. In this study, there was a difference in the time wherein urinary occult blood disappeared between groups A and B because the remaining inflammatory lesions were in the epipharynx and EAT was effective in reducing the volume of urinary occult blood in these patients.
The absence of hematuria reflects the disappearance of glomerular vasculitis, which contributes to a better long-term prognosis among patients with IgAN [26]. Thus, in this study, urinary occult blood level was used to assess the therapeutic efficacy of EAT against chronic epipharyngitis among patients with IgAN. Further, hematuria is an important indicator for treatment efficacy among patients with IgAN. In this study, the urinary occult blood and protein levels significantly reduced among 19 patients who underwent all EAT sessions. In group B, the Cr and protein levels and eGFR did not improve with EAT. However, the urinary occult blood levels were significantly better. Hence, EAT is effective for urinary occult blood. In the current research, several patients with IgAN had reduced urinary findings. Group A included patients who just had tonsillectomy or who were on steroid pulse therapy. In such a case, whether the improvement in urinary findings was attributed to EAT alone could not be confirmed. By contrast, group B included patients who had tonsillectomy + steroid pulse therapy, which has been completed and has elapsed time, or those who relapsed after a mild recovery based on urinary findings. Hence, the assessment of group B may explain the efficacy of EAT alone. Tonsillectomy + pulse therapy with EAT may be useful regardless of the patient’s current treatment if it has some benefits among patient with IgAN. If there is a need to limit patients, EAT is better indicated for group B.
The current study included patients with IgAN who came to my clinic for EAT. Patients with IgAN who do not prefer EAT are basically monitored by nephrologists. It is ethically impossible to followup patients who come to my clinic for EAT without performing EAT. Thus, future prospective studies including a control group and collaborating with otolaryngologists and nephrologists must be performed. In this study, not all patients showed a reduction in the urinary occult blood level with the addition of EAT. The reason for no improvement in the urinary occult blood level in patients after tonsillectomy, steroid pulse therapy, and EAT is still not known. Lastly, it is yet to be confirmed if additional steroid pulses are the only option for treatment. After tonsillectomy and/ or intravenous steroid pulse therapy, the epipharyngeal findings of patients temporarily improved. Sakamoto et al. reported the local epipharyngeal findings in seven patients before and after pulse therapy [27]. Nevertheless, future studies should be conducted to validate how epipharyngeal findings change over the course of IgAN treatment. In conclusion, the urinary findings of 19 patients treated until the end of the study improved with tonsillectomy + pulse therapy with EAT. Group B had a significantly faster improvement in urine freshness than group A. This result might be attributed to the fact that the site of glomerulo vasculitis focal inflammation in group B was only the nasopharynx. Therefore, I believe that the chronic epipharyngitis, which is thought to be the cause of the focal inflammation, was treated with a therapeutic approach and that the chronic epipharyngitis was considerably alleviated by EAT, consequently relieving urine occult blood in patients with IgA nephropathy. Hence, EAT can be an additional treatment for patients with IgAN. If there is a need to limit patients, EAT is better indicated for group B.
Acknowledgment and Funding Source
I express my sincere gratitude to Dr. Hirobumi Ito for his suggestions on the clinical practice of chronic epipharyngitis. I wish to thank Yosuke Sasaki of the SATISTA Corporation for his statistical analysis. I would like to thank Editage (www.editage.com) for English language editing. No funding was received for this study.
Meeting Information
This study was presented at the 120th Annual Meeting of the Oto-Rhino-Laryngological Society of Japan, Osaka City, Japan, May 9th, 2019.
Conflicts of Interest
No conflicts of interest are declared by the authors.
References
- Berger J (1969) IgA glomerular deposits in renal disease. Transplant Proc 1: 939-944.
- Nitta K, Goto S, Masakane I (2020) on behalf of the Japanese Society for Dialysis Therapy Renal Data Registry Committee. Annual dialysis data report for 2018, JSDT Renal Data Registry: Survey methods, facility data, incidence, prevalence, and mortality. Ren Replace Ther 6: 1-18.
- Koyama A, Igarashi M, Kobayashi M (1997) Natural history and risk factors for immunoglobulin A nephropathy in Japan. Research Group on Progressive Renal Diseases. Am J Kidney Dis 29: 526-532.
- Hotta O, Miyazaki M, Furuta T (2001) Tonsillectomy and steroid pulse therapy significantly impact on clinical remission in patients with IgA nephropathy. Am J Kidney Dis 38: 736-743.
- Hotta O, Tanaka A, Tani S (2016) Epipharyngeal treatment in systemic disease: anticipated breakthrough. Stomato-Pharyngology 29: 99-106.
- Hotta O, Tanaka A, Oda T (2019) Chronic epipharyngitis: A missing background of IgA nephropathy. Autoimmun Rev 18: 835-836.
- Kaneko T, Mii A, Fukui M (2015) IgA nephropathy and psoriatic arthritis that improved with steroid pulse therapy and mizoribine in combination with treatment for chronic tonsillitis and epipharyngitis. Intern Med 54: 1085-1090.
- Fujimoto M, Katayama K, Nishikawa K (2020) A kidney transplant recipient with recurrent Henoch-Schönlein purpura nephritis successfully treated with steroid pulse therapy and epipharyngeal abrasive therapy. Nephron144 Supplement 1:54-58.
- Mogitate M, Sasaki Y, Komiyama A (2021) Outcome of an outpatient specialty clinic for chronic epipharyngitis. Auris Nasus Larynx 48: 451-456.
- Horiguchi S (1975) The discovery of the nasopharyngitis and its influence on general diseases. Acta Otolaryngol Suppl. 329:1-120.
- Hotta O, Nagano C, Tanaka A (2017) Possible mechanisms underlying epipharyngeal abrasive therapy (EAT) with ZnCl2 solution for the treatment of autoimmune diseases and functional somatic syndrome. J Antivir Antiretrovir 9: 4.
- Gorna R, MacDermott N, Rayner C (2021) Long COVID guidelines need to reflect lived experience. Lancet 397: 455-457.
- Nalbandian A, Sehgal K, Gupta A (2021) Post-acute COVID-19 syndrome. Nat Med 27: 601-615.
- Imai K, Yamano T, Nishi S (2022) Epipharyngeal abrasive therapy(EAT) has potential as a novel method for Long COVID treatment. Viruses 14:907.
- Hagino H (2021) Efficacy of epipharyngeal abrasive therapy on postural orthostatic tachycardia syndrome (POTS) - Treatment of epipharyngeal lesions, a recent focus of attention in otolaryngology, its methods and effectiveness. Adolescentology 39:283-289.
- Tanaka A, Hotta O (2022) The efficacy of intranasal sphenopalatine ganglion stimulation (INSPGS) in long COVID, and its possible mechanisms. Sch J Oto 8(2): 860-864.
- Nishi k, Yoshimoto S, Nishi S (2022) Epipharyngeal abrasive therapy down-regulates the expression of SARS-CoV-2 entry factors ACE2 and TMPRSS2. In vivo 36: 371-374.
- Hotta O (2010) Chronic epipharyngitis and its possible focal―infection role. Stomato-Pharyngology 23: 37-42.
- Tanaka A (2015) A case of IgA nephropathy successfully treated with epipharyngeal zinc chloride abrasive therapy (B spot therapy) for chronic epipharyngitis. Stomato-Pharyngology 28:326.
- Higa T, Kayou Y, Maeda H (2019) Effectiveness of epipharyngeal abrasive therapy in patients with IgA nephropathy suspected to have chronic epipharyngitis. J Jpn Soc Immunol Allergol Otoralyngol JJIAO 37: 210.
- Japanese Society for Dialysis Therapy (2018) Statistical Survey Committee of the Japanese Society for Dialysis Therapy. Current status of chronic dialysis therapy in Japan (as of December 31, 2017). Japanese Society for Dialysis Therapy 51: 699-766.
- Maruyama S (2017) Evidence-based guidelines for immunoglobulin A nephropathy. Igakusha, Tokyo, Japan 20: 511-535.
- Sugita R (2010) A diagnostic method of the nasopharyngitis and a follow-up by cytology. Stomato-Pharyngology 23: 23-35.
- Hotta O (2020) Involvement of epipharynx-kidney axis in chronic kidney disease. Stomato-Pharyngology 33: 17-23.
- Sevillano AM, Gutiérrez E, Yuste C (2017) Remission of hematuria improves renal survival in IgA nephropathy. J Am Soc Nephrol 28: 3089-3099.
- Sakamoto K, Takemiya Y, Suzuki A. (2018) Endoscopic observation of local epipharyngeal findings before and after pulse therapy and tonsillectomy of IgA nephropathy. Stomato-Pharyngology 31: 392.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...