Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2638-6003
Research Article(ISSN: 2638-6003) 
Diagnostic Accuracy of Clinical Tests in Detecting Rotator Cuff Pathology Volume 2 - Issue 4
C Τsompos1*, C Panoulis2, K Τοutouzas3, A Triantafyllou4, CG Ζografos3, K Tsarea5, M Karamperi5 and A Papalois5
- 1 Department of Gynecology, General Hospital of Thessaloniki “St. Dimitrios” Thessaloniki, Hellas, Greece
- 2 Department of Obstetrics & Gynecology, Aretaieion Hospital, Athens University, Athens, Attiki, Hellas, Greece
- 3 Department of Surgery, Ippokrateion General Hospital, Athens University, Athens, Attiki, Hellas, Greece
- 4 Department of Biologic Chemistry, Athens University, Athens, Attiki, Hellas, Greece
- 5 Experimental Research Centre ELPEN Pharmaceuticals, S.A. Inc., Co., Pikermi, Attiki, Hellas, Greece
Received:January 28, 2019; Published: February 05, 2019
Corresponding author: Tsompos Constantinos, Department of Gynecology, General Hospital of Thessaloniki “St. Dimitrios” Thessaloniki, Hellas, Greece
DOI: 10.32474/OSMOAJ.2019.02.000142
Abstract
Aim: This study calculated the effects on acid phosphatase (ACP) levels, after treatment with either of 2 drugs: the erythropoietin (Epo) and the antioxidant lazaroid (L) drug U-74389G. The calculation was based on the results of 2 preliminary studies, each one of which estimated the certain influence, after the respective drug usage in an induced ischemia reperfusion (IR) animal experiment.
Materials and Methods: The 2 main experimental endpoints at which the serum ACP levels (ACPl) were evaluated was the 60th reperfusion min (for the groups A, C and E) and the 120th reperfusion min (for the groups B, D and F). Specially, the groups A and B were processed without drugs, groups C and D after Epo administration; whereas groups E and F after the L administration.
Results: The first preliminary study of Epo presented a non-significant hypophosphatasemic effect by 9.29%+6.36% (p-value=0.1396). The second preliminary study of U-74389G presented a significant hypophosphatasemic effect by 74.46%+9.63% (p-value=0.0000). These 2 studies were co-evaluated since they came from the same experimental setting. The outcome of the coevaluation was that L is 8.011334-fold [7.996741 - 8.025953] more hypophosphatasemic than Epo (p-value=0.0000).
Conclusion: The anti-oxidant capacities of U-74389G ascribe 8.011334-fold more hypophosphatasemic effects than Epo (p-value=0.0000).
Keywords:Ischemia; Erythropoietin; U-74389G; Acid Phosphatase Levels; Reperfusion
Introduction
The lazaroid U-74389G (L) may be not famous for its hypophosphatasemic1 capacity (p-value=0.0000). U-74389G as a novel antioxidant factor, implicates exactly only 260 published studies. The ischemia reperfusion (IR) type of experiments was noted in 18.84% of these studies. A tissue protective feature of U-74389G was obvious in these IR studies. The U-74389G chemically known as 21-[4-(2,6-di-1-pyrrolidinyl-4-pyrimidinyl)- 1-piperazinyl]-pregna-1,4,9(11)-triene-3,20-dione maleate salt is an antioxidant complex, which prevents the lipid peroxidation either iron-dependent, or arachidonic acid-induced one. Animal kidney, liver, brain microvascular endothelial cells monolayers and heart models were protected by U-74389G after IR injury. U-74389G also attenuates the leukocytes; down-regulates the proinflammatory gene; treats the endotoxin shock; produces cytokine; enhances the mononuclear immunity; protects the endothelium and presents antishock property.
Erythropoietin (Epo) even if is not famous for its hypophosphatasemic action (p-value=0.1396), it can be used as a reference drug for comparison with U-74389G. Although Epo is met in over 30,616 published biomedical studies, only a 3.57% of them negotiate the known type of IR experiments. Nevertheless, Epo as a cytokine, it is worth of being studied about its effects on acid phosphatase levels (ACP) levels too. This experimental work tried to compare the effects of the above drugs on a rat induced IR protocol. They were tested by calculating the serum ACP levels (ACPl) reductions.
Material and Methods
Animal Preparation: The Vet licenses under 3693/12-11- 2010 & 14/10-1-2012 numbers, the granting company and the experiment location are mentioned in preliminary references [1,2]. The human animal care of Albino female Wistar rats, the 7 days pre-experimental ad libitum diet, the non-stop intra-experimental anesthesiologic techniques, the acidometry, the electrocardiogram, the oxygen supply and post-experimental euthanasia are also described in preliminary references. Rats were 16 - 18 weeks old. They were randomly assigned to six (6) groups consisted in N=10. The stage of 45 min hypoxia was common for all 6 groups. Afterwards, reperfusion of 60 min was followed in group A; reperfusion of 120 min in group B; immediate Epo intravenous (IV) administration and reperfusion of 60 min in group C; immediate Epo IV administration and reperfusion of 120min in group D; immediate U-74389G IV administration and reperfusion of 60min in group E; and immediate U-74389G IV administration and reperfusion of 120min in group F. The dose height assessment for both drugs are described at preliminary studies as 10 mg/Kg body mass.
Ischemia was caused by laparotomic clamping the inferior aorta over renal arteries with forceps for 45min. The clamp removal was restoring the inferior aorta patency and reperfusion. After exclusion of the blood flow, the protocol of IR was applied, as described above for each experimental group. The drugs were administered at the time of reperfusion; through inferior vena cava catheter. The ACPl were determined at 60th min of reperfusion (for A, C and E groups) and at 120th min of reperfusion (for B, D and F groups). Along, a weak relation was rised between ACPl values with animals’ mass (p-value=0.0903).
Statistical Analysis
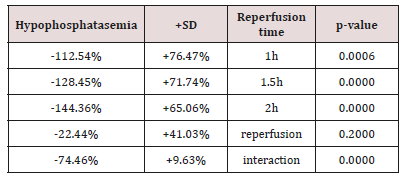
Table 1: The (%) hypophosphatasemic influence of erythropoietin in connection with reperfusion time.
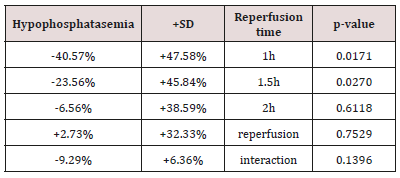
Table 1 presents the (%) hypophosphatasemic influence of Epo regarding reoxygenation time. Also, Table 2 presents the (%) hypophosphatasemic influence of U-74389G regarding reperfusion time. Chi-square tests were applied using the ratios which produced the (%) results per endpoint. The outcomes of chi-square tests are depicted at Table 3. The statistical analysis was performed by Stata 6.0 software [Stata 6.0, StataCorp LP, Texas, USA].
Table 3: The U-74389G / erythropoietin efficacies ratios on serum acid phosphatase levels after chi-square tests application.
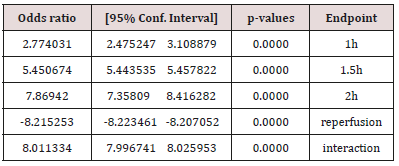
Results
The successive application of chi-square tests revealed that U-74389G reduced the ACPl by 2.774031-fold [2.475247 - 3.108879] more than Epo at 1h (p-value=0.0000), by 5.450674-fold [5.443535 - 5.457822] more than Epo at 1.5h (p-value=0.0000), by 7.86942-fold [7.35809 - 8.416282] more than Epo at 2h (p-value=0.0000), opposite by 8.215253-fold [-8.223461 - -8.207052] (p-value=0.0000) without drugs and by 8.011334-fold [7.996741 - 8.025953] more than Epo whether all variables have been considered (p-value=0.0000).
Discussion
The unique available study investigating the hypophosphatasemic effect of U-74389G on ACPl was the preliminary one1. Although the most famous activities of neuroprotection and membrane-stabilization properties, it accumulates in the cell membrane, protecting vascular endothelium from peroxidative damage but hardly penetrates the blood-brain barrier. It elicits a beneficial effect in ototoxicity and Duchenne muscular dystrophy. It increases γgt, superoxide dismutase (SOD) and glutathione (GSH) levels in oxygen-exposed cells. It treats septic states and acts as immunosuppressant in flap survival. It prevents the learning impairments, it delays the early synaptic transmission decay during hypoxia improving energetic state of neurons. It shows antiproliferative properties on brain cancer cells and is considered as a new promising anti-inflammatory drug for the treatment of reperfusion syndrome in IR injuries.
The same authors confirmed [2] the short-term hypophosphatasemic effect of Epo preparations in noniron deficient individuals. Otero JE et al reported [3] a 7-year-old boy with generalized arterial calcification (AC) of infancy (GACI) referred for profound, acquired, skeletal disease. AC was gone after 5 months of the first-generation bisphosphonate etidronate (EHDP) ethane-1-hydroxy-1,1-diphosphonic acid, also known as 1-hydroxyethylidene-bisphosphonate) therapy during infancy, but GACI-related joint calcifications progressed. He was receiving EHDP, 200mg/day orally, and had odynodysphagia, diffuse opioid-controlled pain, plagiocephaly, facial dysmorphism, joint calcifications, contractures, and was wheelchair bound. Serum osteocalcin was low and the brain isoform of creatine kinase and tartrate-resistant acid phosphatase 5b (TRAP-5b) were elevated as in osteopetrosis. Şehirli AÖ, et al. [4] investigated whether melatonin protects against ischemic heart failure (HF). Αcid phosphatase, and cathepsin-D) were studied in plasma samples. Melatonin treatment reversed all the functional and biochemical changes. The present results demonstrate that Mel ameliorates ischemic heart failure in Wistar albino rats. Spaans F et al found [5] that ATP infusion decreased interstitial trophoblast invasion on day 17 and spiral artery remodeling on day 17 and 20, increased activated tartrate resistant acid phosphatase (TRAP)-positive macrophages on day 15, decreased NK cells on day 17 and 20, and decreased inducible nitric oxide synthase (iNOS)-positive and CD206-positive macrophages and TNF-α and IL-33 expression at the end of pregnancy (day 20). These ATP-induced modifications were preceded by an increase in activated TRAP-positive macrophages and coincided with NK cell numbers. Trophoblast invasion and spiral artery remodeling may be inhibited by ATP-induced activated macrophages and decreased NK cells in the rat mesometrial triangle in preeclampsia. Marina N et al invented [6] that facilitated breakdown of extracellular ATP in the rostral ventrolateral medulla oblongata by virally-driven overexpression of a potent ectonucleotidase transmembrane prostatic acid phosphatase results in a significant reduction in the arterial blood pressure in the spontaneously hypertensive rats (but not in normotensive animals).
These results suggest that in the spontaneously hypertensive rat, lower PO2 of brainstem parenchyma may be associated with higher levels of ambient ATP and l-lactate within the presympathetic circuits, leading to increased central sympathetic drive and concomitant sustained increases in systemic arterial blood pressure. Ogundele OM et al hypothesize [7] that differential oxygen concentration in both forms of ischemia affect ROS production rate in the mitochondria; thus distinguishing the cytotoxicity pattern for cyanide (CN) and vascular occlusion (VO). In addition, CN recorded a significant increase in GSH when compared with the control and VO (p < 0.001). The level of ROS was also proportional to the degree of lipid peroxidation (GPx) and autophagic cell response (ACP/CAD+). Song C et al started [8] the treatment with 30ng/ml RANKL at 12hr after seeded RAW264.7 with the density of 6.25 × 103 cells/cm2 manifested an significantly increased number of multinucleated osteoclastic cells, through tartrate-resistant acid phosphatase (TRAP) staining. Overall, these results independently induce RAW 264.7 cell osteoclastic differentiation, which could efficiently generate osteoclasts in vitro in RANKL-treated RAW264.7 cells.
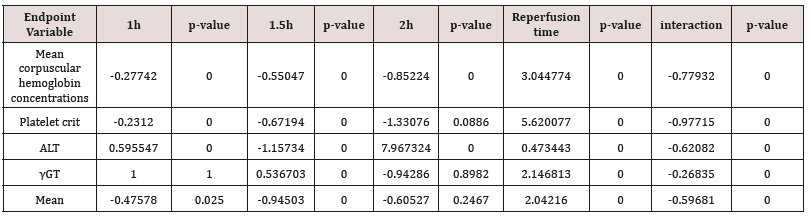
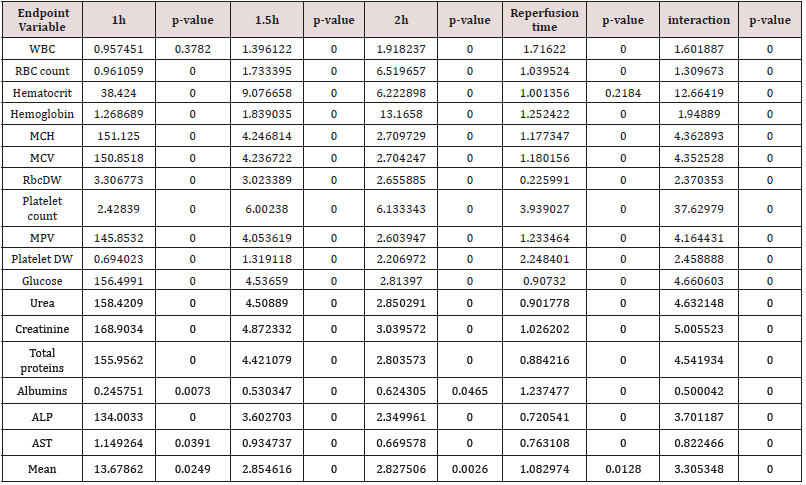
According to above, Table 3 shows that U-74389G has 8.011334-fold [7.996741 - 8.025953] more hypophosphatasemic effect than Epo (p-value=0.0000) whether all variables have been considered (p-value=0.0000); a trend accentuated along time, in Epo non-deficient rats. A meta-analysis of these ratios from the same experiment, for 20 other seric variables, provides comparable results (Tables 4 & 5) [9].
Table 4: A U-74389G / erythropoietin efficacies ratios meta-analysis on 21 hematologic variables (17 variables with balancing efficacies and 4 variables with opposite efficacies) [9].
Conclusion
The anti-oxidant agent U-74389G was proved having 8.011334- fold [7.996741 - 8.025953] more hypophosphatasemic effect than Epo whether all variables have been considered (p-value=0.0000); a trend accentuated along the short-term time frame of the experiment in rats. A biochemical investigation remains about how U-74389G mediates in these actions.
References
- Tsompos C, Panoulis C, Toutouzas K, Triantafyllou A, Zografos G, et al. (2016) The effect of the antioxidant drug “U-74389G” on acid phosphatase levels during ischemia reperfusion injury in rats. Appl Med Res.
- C Τsompos, C Panoulis, K Τοutouzas, G Ζografos, A Papalois (2014) The effect of erythropoietin on acid phosphatase levels during ischemia reperfusion injury in rats. Actual Osteol 10(3): 247-253.
- Otero JE, Gottesman GS, McAlister WH, Mumm S, Madson KL, et al. (2013) Severe skeletal toxicity from protracted etidronate therapy for generalized arterial calcification of infancy. J Bone Miner Res 28(2): 419- 430.
- Şehirli AÖ, Koyun D, Tetik Ş, Özsavcı D, Yiğiner Ö, et al. (2013) Melatonin protects against ischemic heart failure in rats. J Pineal Res 55(2): 138- 148.
- Spaans F, Melgert BN, Chiang C, Borghuis T, Klok PA, et al. (2014) Extracellular ATP decreases trophoblast invasion, spiral artery remodeling and immune cells in the mesometrial triangle in pregnant rats. Placenta 35(8): 587-595.
- Marina N, Ang R, Machhada A, Kasymov V, Karagiannis A ,et al. (2015) Brainstem hypoxia contributes to the development of hypertension in the spontaneously hypertensive rat. Hypertension 65(4): 775-783.
- Ogundele OM, Wasiu Gbolahan B, Emmanuel Cobham A, Azeez Olakunle I, Abdulbasit A (2017) Differential oxidative stress thresholds distinguishes cellular response to vascular occlusion and chemotoxicity in vivo. Drug Chem Toxicol 40(1): 101-109.
- Song C, Yang X, Lei Y, Zhang Z, Smith W, et al. (2018) Evaluation of efficacy on RANKL induced osteoclast from RAW264.7 cells. J Cell Physiol.
- Τsompos C, Panoulis C, Τοutouzas K, Triantafyllou A, Zografos GC, et al. (2018) Comparison of the Hypertransferasemic Effects of Erythropoietin and U-74389G on Aspartate Aminotransferase Levels. Clin Res Hematol 1(2): 1-7.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...