Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-6660
Research Article(ISSN: 2637-6660) 
Synthesis of Some Piperidine, Pyrmidine and Diazipine Compounds Containing Furyl Derivatives Volume 1 - Issue 5
MS Al-Ajely1* and I M Shaban2
- 1Environmental Department, College of Environment and Technology ,University of Mosul, Iraq
- 2Department of Science, College of Agriculture, Northin Technical University
Received: April 02, 2019; Published:April 17, 2019
*Corresponding author:MS Al-Ajely, Environmental Department, College of Environment and Technology, University of Mosul, Iraq
DOI: 10.32474/ANOAJ.2019.01.000125
Abstract
It was known that nearly 65% of anti-cancer drugs granted market approved by FDA between 210-1015 are heterocyclic compounds and about 95% of heterocyclic compounds are drugs. Heterocyclic compounds containing nitrogen atoms in their structures (nitrogen based)were also proved by FDA during the above period that they form two third of cancer drugs. There are many corresponding reviews associated with the impact and usage of different heterocyclic compounds as anti- platelet, antihypertensions and a lot of biological and medicinal applications. So according to the importance of heterocyclic compounds in therapeutic and a variety of pharmaceutical, medicinal applications which attracts attentions of many researchers on synthesizing new compounds of this class. We are here synthesizing some new nitrogen containing heterocyclic compounds, Piperidine compounds(E4-8), 1,4-Dihydropyridine derivatives(E9-20) And finally diazepine compounds(E31-37)The synthesized compounds were studied using IR and1H NMR Spectral methods and were discussed.
Keywords: Piperidine; Pyrmidine; Azipine; Furfural
Introduction
Furfural is known as important intermediate for a variety of chemical compounds for example semi carbazides and semi carbazones [1], 2-(2-Furyl) [1,3] dioxolane [2]. It was also found that furfural is also used for medicinal and industrial applications [3] S Lewkowski in (2001) has synthesized 2,5-furan dicarboxylic acid by multistep synthesis starting from furfural [4].It was known that mucochloric acid had remarkable activity. against malaria disease . This compound was used as a precursor for the synthesis of 4-amino-5-hydrony-2-(5H) furan [4] Dichalcon compound was prepared from furfural on treatment with ketones [5]. Mucobromic acid is a derivative of furfural [6]. 6-(2-furyl)1,4,5,6-tetralydro tetrazine 3(2H)thione was also prepared by treatment of furfural with thiocarbohydrizide [5]. Mandalika in 2012 has synthesized furil from furfural which was the first compound used as insect sized [3] while in 2013 furfural diethyl acetel was prepared by treatment of furfural with ethanol in presence of some selective catalysis [7-9]. This derivative was used for many synthetic routes. N-(E)-furan-2-methyleden-2-(1,3-beuzothiuzol-2-yl-sulfonyl) aceto hydrazide was prepared from the reaction of furfural with 2-methyl benzothiezole by conventional and microwave methods [10] in (2011) M Baummann et al. have publish a review for the most selling 5-membered ring heterocyclic, pharmaceuticals which indicate the importance of furfural as a moiety for drug synthesis [11]. Furthermore, flavon compound contains furfural ring, furfur aldehyde exhibited IC so values of 75.9, 51.0 and 59.3 mM for HT29, MCF7 and A498 respectively as anti-cancer cell lines [10]. it was also used for the synthesis of 2,3-diamino quinoline [12]. Mucobromic acid was also used for the synthesis of 2-hydroxy-3-(2-indolyl)- 4-bromo furan-5-one by Suzuki reaction13.Many heterocyclic compounds containing furfural moiety have proved to have many pharmaceutical applications [13,14]. In our present study we started synthesizing nitrogen based heterocycles from furfural as precursor for these compounds which are peridines, pyrimidines and azipene these nitrogen containing compounds have proved their activities against nervous system diseases including depression, psychological disorder and schizophrenia [15-17] so they are used as a pharmaceutical compounds for many applications [18-20]. Benzodiazepines are effective for treating a range of psychological and neurological disorders, due to its effects on the neurons that trigger stress and anxiety reactions Carbamazepine, or CBZ, is an anticonvulsant medication and a mood-stabilizing agent. It acts by decreasing the amount of excitement in the brain and is used for epilepsy and the treatment of bipolar disorders. Carbamazepine is effective in controlling seizures by blocking specific brain impulses [21]. It is also used to treat ADD (Attention Deficit Disorder), ADHD (Attention Deficit Hyperactive Disorder), and schizophrenia (a psychiatric condition). According to the above importance of both furfural as a furyl heterocyclic moiety and of these new nitrogen compounds and in continuing of our previous work [22] we have investigating these new series of compounds which are promising nitrogen base heterocyclic compounds in which they need further study to prove their biological actions.
Experimental
All melting points were un corrected using Electro thermal melting point apparatus. The chemical compounds were supplied by Aldrich, Fluka and BDH chemical companies.IR spectra were performed using Infrared spectrophotometer Tenson Bruker Co.Germany.1H NMR were recorded using Bruker 400MHz/ Gazisosmanpasa, University (Turkey)in DMSO D6 as solvent. 4,5-Dibromo furfural(1) was prepared using the same published procedure [Dsim] reagent(2) was prepared according to the published procedure. Cellulose sulfuric acid(3) was also prepared following the published procedure.
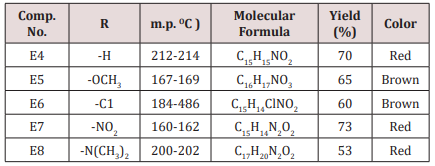
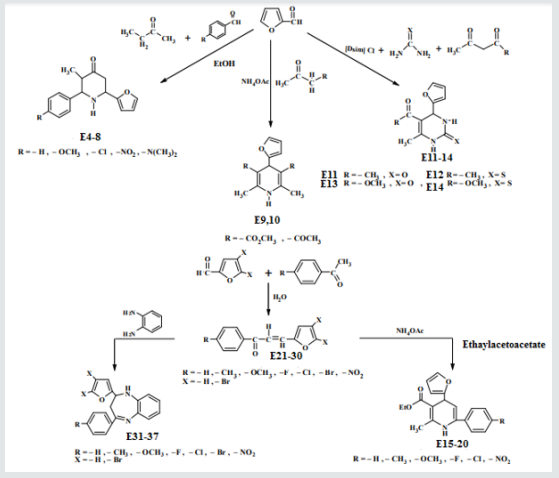
Synthesis of 2-(furan-2-yl)-6-aryl Piperidine-4-one(E4-8)
A mixture of 0.01mol. of ammonium acetate in 30ml. ethanol was mixed with 0.05mol. of benzaldehyde, 0.05mol. of furfural and 0.025mol. of 2-butanon.The mixture was refluxed for 2h, cooled and left for 24 h. at room temperature after that 15ml. of conc. HCl was then added, 20ml. of acetone. The suspended solution was treated ammonia solution then with water. The sold product filtered off and crystallized from ethanol physical and IR spectral data are presented in (Tables 1).
Synthesis of some 1,4-dihydro Pyridine Derivatives (E9,10)
A mixture of (0.96g, 0.01mol.) furfural, 0.02mol. of (methyl aceto acetate or acetyl), 0.013mol. of ammonium acetate and 25 ml. of water. The reaction mixture was stirred at 70o C for 90 min. After that ethanol was added. The precipitated compound was filtered off, dried and crystallized from ethanol, physics and IR spectral data were as follows: compound 6, mp. 188-190o C , yield 74% as white crystals, compound 10 has mp. 177-179o C, yield 62% as yellow crystals . The IR spectra of those two compounds showed the following main absorption bands: 3346-3448cm-1,1705,1697,1653 and 1192, 1099,1219,1122.
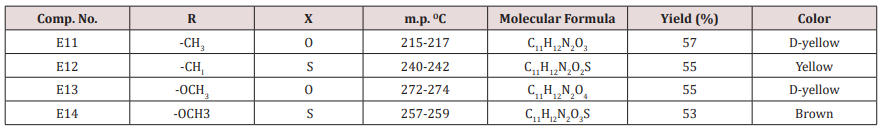
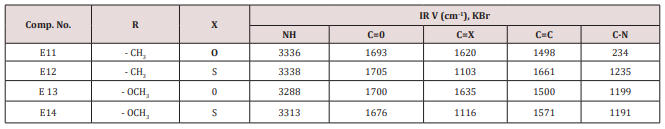
Synthesis of some 1,4-dihydro Pyrimidine Derivatives (E11-14)
A mixture of furfural (2.4g, 0.025mol.), Urea or thiourea (0.033mol.) and acetyl acetone or methyl acetoacetate ( 0.12 g., 0.004mol.) of compound (2) was refluxed on 90o C for 20 min. using oil bath, cooled, water was added to precipitate the product which was filtered off and crystallized from ethanol , physical and IR spectral data were shown in (Table 2).
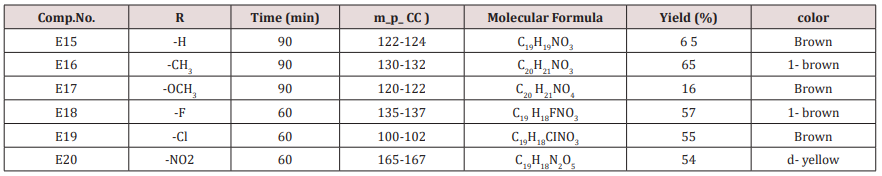
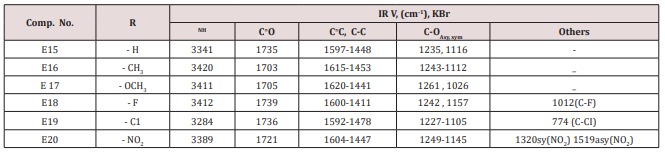
Synthesis of 2-Methyl-4-(furan -2yl)-6-substituted phenyl-1,4- dihydro-3-pyridine carboxylate (E15-20)
Ethyl acetoacetate (0.001mol. and compounds E25-31) each 0.01mol. and 0.01mol. Ammonium acetate were mixed with compounds (3), (0.05g.) in 5ml. of H2 O. The final mixture was refluxed with stirring for the indicated time (Table 5), cooled, 25mol. of dichloromethane was then added , filtered. The filtrate was evaporated under reduced pressure. The solid product was recrystallized from ethanol, physical and IR spectral data were shown in (Tables 3).
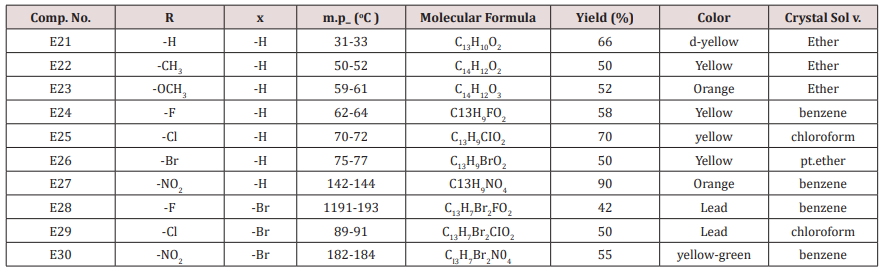
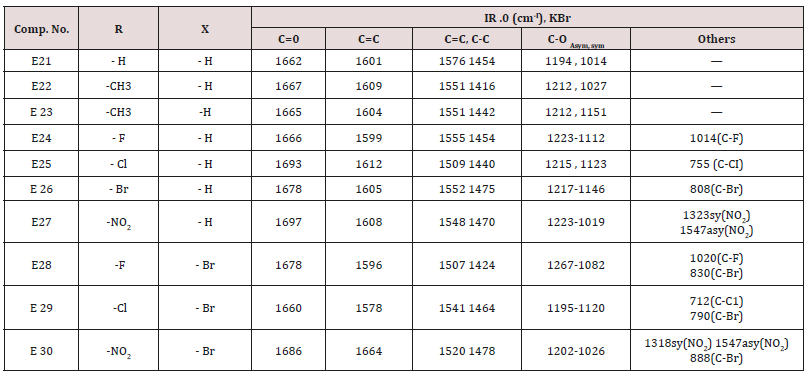
Synthesis of 1-(4-substituted phenyl)-3-(4,5-dibrono furan-2-yl or furan-2yl)-2-propene-1-one(E21-30)
Furfural or 4,5-dibromo furfural (0.034mol.) and (0.034mol.) of acetophenone or its substitutes were mixed together and stirred at 5-15o C for 10 min., sodium hydroxide 10% was then added drop wise. After complete addition, the mixture was stirred for further 3-4 hours. The reaction mixture was left for 24 hours. The solid ppt. was washed with cold water, physical and IR spectral data are presented in (Tables 4).
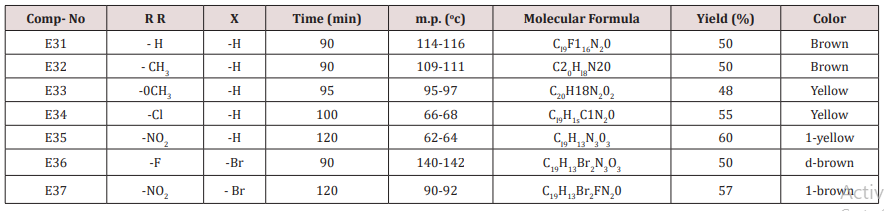
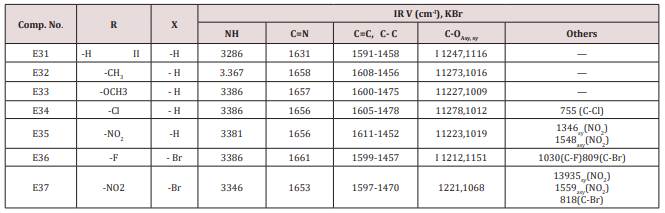
Synthesis of 2-(Furan-2-yl)-4-phenyl -2,3-dihydro -1H-benzo[b] ,[1,4]-diazepines(E31-37)
Compounds (E13-23), 0.01mol., ortho phenylene diamine (0.01mol.) were mixed together, cellulose- sulfonic acid (0.01 mol.) was then added . The reaction mixture was refluxed at 80o C for a given time Tables (10) the reaction was monitored by TLC after completion it was cooled and extracted with 3x10 of ether. The combined ether was dried, and the solvent was evaporated under reduced pressure. The solid product was crystallized from etherethyl acetate, Physical and spectral data are presented in (Table 5).
Results and Discussion
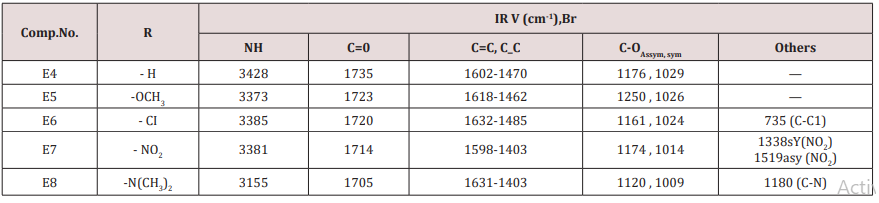
Compounds (E4-8) were prepared from the reaction of furfural with benzaldehyde or its derivatives in presence of 2-butanone and ammonium acetate in ethanol as a solvent (scheme 1) .These compounds were characterized by IR cm-1: 3155-3428 belong to NH,1705-1735 for C=O while the aromatic appeared at 1632-1662, C-O sym and assym . absorbed at 1009 and 1176 respectively . Proton magnetic resonance of compound E5 showed the following resonating signals ʃppm : 8.64(s) for NH, (m) signal at 7.04-7.06 for benzene and furan ring (7H) , singlet signal at 3.82 belongs to –OCH3 of the phenyl substituted (3H), doubled signal at 3.39 and 3.44 for the CH proton Piperidine ring (2H) while the CH2 of Piperidine ring appeared at 2.50ppm. The 1HNMR spectrum of compound E10 was characterized by the following resonating signals ʃ ppm.: 9.01(s) for NH proton, 7.40 (d) for 5H of furan ring, 6.24 for 4H of the furan ring, 5.83 (d) for proton 3 of furan ring , 5.04 (s) for the Piperidine ring proton, 3.60 for OCH3 protons and 2.25 (s) for CH3 (6H) (Table 6).
Compound (E11-14)
This series of compounds were prepared by condensation of compounds (E4-8) with Urea, thiourea , acetyl acetone or methyl aceto acetate using [Dsim] as catalyst(scheme1). The synthesized compound was characterized by IR (Table 7).
The first two compounds of this series (E11-12) were characterized by 1HNMR as follows: For E11 compound (s) signal at 9.30ppm. Belongs to NH proton, (d) at 7.50ppm. For proton at position 5 of furan ring, (t) signal at 6.35ppm. fpr proton of position 4 of furan ring, (d) signal at 6.10ppm. For proton of position 3 of furan ring, (d) at 5.3ppm. For the Piperidine proton, (5) of 3.37ppm. belongs to C=OCH3 protons and (d) at 2.23ppm. for CH3 protons. While compound E12 characterized by the following resonating signals ʃppm.: 10.31, 9.7 (s) belong to NH proton, (s) at 7.58 for position (5) proton of furan ring, (t) at 6.38 for position (4) proton, (d) at 6.17 for position (3) proton of furan ring, (d) at 5.36 for the Piperidine proton, (s) at 3.30 assigned to COCH3 and (s) at 2.20 to CH3 equivalent to 3H.
Compounds (E15-20)
This series of compounds were prepared from the condensation of the corresponding chalcones with ethyl aceto acetate, ammonium acetate using cellulose sulfonic acid as catalyst. These compounds were characterized by the following IR absorption bands; see (Table 8). This Table revealed the presence of NH absorbed at 3284-3420cm-1 for NH, 1703-1739cm-1 for C=C while C-O sym and assym appeared at 1026-1261cm-1, see Table 8. 1HNMR spectrum of compound E16 gave the following resonating signals ʃppm.: multiplet(m) signal at 7.42,7.59,7.65,7.77,7.82 and 8.07 related to 5 position protons of furan and the aromatic ring protons, NH proton respectively. Triplet signal at 6.40 belongs to proton of furan ring, (d) at 6.33 for position 3 proton of furan ring, (d) at 5.00 for proton 5 of furan ring, (d) at 4.63 for Piperidine proton of position 4, (q) at 4.18 for C2 of ester moiety, (s) at 3.88 for CH3 of the substituted at 2 position of Piperidine, (s) at 2.50 for CH3 of ester, 1.94 as double belongs to CH3 of ester see.
Compounds (E<21-30)
This series of chalcone compounds were prepared from the condensation of 4,5-di bromo furfural with some acetophenones (Scheme 1). The synthesized compounds were characterized by IR, Table. This Table revealed the presence of C=O stretching vibration at 1660-1697cm-1, C=C Aliphatic appeared at 1578-1664cm-1, while the aromatic C=C absorbed at 1416-1576cm-1 together with C-O sym and assym as indicated in (Table 9). These chalcone compounds were used as starting materials for the preparation of diazipine compounds by the condensation with ortho phenylene diamine using sulfamic acid to increase the positive character of the carbonyl chalcones. The final(diazipin) compounds were characterized by the following IR bands cm-1: 3188-3346 for NH, 1631-1661 for C=N, 1452-1611 for Aromatic C=C stretching, C-O sym and assym. of 1009-1253, see (Table 10 ). As a representative sample of this series compound E36 was studied by 1HNMR . The spectrum showed the following resonating signals: (m) signal at 6.96,7.36 and 7.81 for furan ring protons in which aryl and phenyl ring protons appeared within the same range, (s) signal at 6.26 for proton of position 2 of azipine ring, (s) signal at 3.33 for NH, singlet (s) signal at 2.50 for CH3 which is consider with DMSO signal, (d) at 2.24 belongs CH2 of a zipine ring protons.
Conclusion
As it was stated in the introduction part of this work on the importance of heterocyclic compounds as drugs or co- drug and especially for those when furyl moiety and nitrogen residue were part of the whole compound structure .In this investigation we are focusing on those two important categories, So according to the resulted data given in the experimental part together with discussed results we have succeeded in getting these type of heterocyclic compounds with nitrogen base and at the same time having furyl moiety from furfural precursors which means more activities for both type of compounds that becomes two in one final compound structures, pyridines, pyrimidines and azipines.
Acknowledgments
Authors would like to thank Iraqi Ministry of Higher Education and Research for Ibtisam Mohhammad Shaban a Scholarship to do this work, Thanks also extended to the chemistry Dept and The Education college of Mosul University for providing the facilities to do this work which is part of Ibtisam PhD Thesis.
References
- D Tarasova, Ya L Gold farb (1965) Synthesis from 4,5- Dibromo-2- furaldehyde. Russian Chemical Bulletin 29(11): 1978-1979.
- TA Stroganova, AV Butin, LN Sorotskaya, VG Kulʼnevich (2000) (Aryl) (2-furyl)alkanes and their derivatives, 20.1Synthesis of symmetric bisand tris(2-furyl)methanes. Arkat USA, Inc. 2000(4): 641-659.
- S Ramachandran, PK Sreekumar (2011) Synthesis, characterization and antibacterial evalution of 2(5H)- Furanone derivatives from highly functionalized Mucobromic acid. Int Pharm Pharm Sci 3(1): 225-228.
- TJ Ch Hoskins (2008) Carbon - Carbon bond forming reactions of biomass derived aldehydes. MSc Thesis, Georgia Institute of Technology.
- AS Mandalika (2012) Enabling the development of furan-based bio refineries. MSc Thesis, University of Wisconsin, Madison, USA.
- P Arayarat, H Singh, E Lattman (2001) Solid phase synthesis of substituted 4-amino -5- hydroxyl-2(5H)-furanones. Science Asia 27: 121-125.
- Baner HKS Kumara, M Banerjee (2012) Medicinal Significance of furan derivatives. International J Rev Life Sci 2(1): 7-16.
- CK Desai, KR Desai (2000) Etherial base azo-2-naphtholfurfuraldehyde oligomer dyes. Iranian Polymer Journal 9(3): 177-181.
- K Yan, J Liao, X Wu, X Xie (2013) Facile synthesis of ecofriendly Cuydrotalcite catalysts for highly selective synthesis of furfural diethyl acetal and benzoin ethyl ether. Adv Mat Lett 4(9): 702-707.
- P Martins, J Jesus, S Santos, LR Raposo, C Roma-Rodrigues, et al. (2015) Heterocyclic Anticancer Compounds: Recent Advances and the Paradigm Shift towards the Use of Nanomedicine’s Tool Box. 20(9): 16852-16891.
- Anthonia E, Eseyin P, H Steele (2015) An overview of the applications of furfural and its derivatives. International journal of advances chemistry 3(2): 42-47.
- K Chibal, MD Coleman, C Masimirembwa (2012) Drug discovery in africa. Springer Heidelberg New York Dordrecht London, pp. 103-105.
- H Takeuchi, SY kota, S Shimada, Y Ottani, S Miura, et al. (1993) Curr Ther Res Chin Exp 53: 427-434.
- B Hue, B Palomba, M Giacordy-pety-Bottai, R Alric, P Prtit (1998) Ther Drug Manit 20: 335-339.
- S Kapur, R Cho, C Jones, G Mckay , RB Zipursky (1999) Biological stress system. Biol Psychol 45: 1220-1217.
- TP Seivan, CRJames, PV Dniandev, SV Valzita (2012) A Mini review of pyrimidine and fused pyrimidine marketed drugs. Research in Pharmacy 2(4): 1-9.
- A Altaf, A Shahzad, Z Gul, N Rasool, A Badshah (2015) A review on the Medicinal importance of pyridine derivatives. J Of Drug Design and Medicineal Chemistry 1(1): 1-11.
- http://www.medicalook.com/
- MS Al-Ajely, IM Shaban (2018) Synthesis of Some Heterocyclic Compounds Derived from Furfural.
- Ya L Golʼdfarb, LD Tarasova (1965) Bromination products of furfural. Bulletin of the Academy of Sciences of the USSR Division of Chemical Science 14(6): 1041-1042.
- A Zare, T Yousofi, ARM-Zare (2012) Ionic Liquid 1,3- disulfonic acid imidazolium hydrogen sulfate: a novel and highly efficient catalyst for the preparation of 1- carbamatoalkyl -2- naphthols and 1- amidoalkyl2- naphthols. RSC Advances, pp. 7988-7991
- SR Sarda, WN Jadhav, NB Kolhe, MG Landge, RP Pawar (2009) Solvent free one pot synthesis of benzo- [b] - 1,4- diazepines using reusable sulfamic acid catalyst. J Iran Chem Soc 6(3): 477-482.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...