Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1725
Review Article(ISSN: 2641-1725) 
Flu Viruses Quinolines-Covid-19 Volume 5 - Issue 2
Krishnasarma pathy*
- IPL Research centre IndiaHead QC, QA
Received: June 02, 2020; Published: June 18, 2020
*Corresponding author: Krishnasarma pathy, IPL Research centre India Head QC, QA, India
DOI: 10.32474/LOJMS.2020.05.000208
Abstract
Firstly, COVID-19 and influenza viruses have a similar disease presentation. That is, they both cause respiratory disease, which presents as a wide range of illness from asymptomatic or mild through to severe disease and death.Secondly, both viruses are transmitted by contact, droplets and fomites. As a result, the same public health measures, such as hand hygiene and good respiratory etiquette (coughing into your elbow or into a tissue and immediately disposing of the tissue), are important actions all can take to prevent infection.Human H1N1 pandemic developed from the originally localized Mexican source early in the spring 2009. For the emergency created by the epidemic of « influence of the pigs » in Mexico it was correct not to create alarmismes being victims of a bad information. Cytokine storm should be mentioned asone of the key pathogenic events contributing to the overall mortality in substantial portion of patients. If active immunization is assumed to be preventive measure of proven efficacy, clinicians are still in doubt how to treat a complicated course of infection. The possibility that the virus arrives in other parts of the world is real as for all the types of influence virus. In order that a strain has a wide distribution, its antigenic characteristics must ensure that it escapes the neutralization of antibodies of the host and of the surrounding population.
So the outbreaks will happen with those strains that have dominant antigens that fit the deficiency, or better, the abscences of antibody in the population.It seems, in conclusion that the flu virus showes an ability and an aptitude for survival built on the possibility of emergence of new models that allow the virus being confused easily through populations still partly immune to previous antigenic forms. According to this view, the changes in the influenza A can be designed in single meaning, in the context of a principle and of an evelutionary progress, from Burnet said immunological drift or steering immunology. The antiviral drugs (inhibitors of the neuraminidasis, receptor of the virus surface) should be assumed within 48 hours by the appearance of the influence symptoms and for the subjects that have had a close contact with people infected by the flu virus. The vaccination against the influence is the most effective method to prevent the illness.
a. Neuraminidases inhibitors intake, which prevent adhering of viral capsid to the eukaryotic (e.g. Mammal or human) cell
membrane and„decoratingof RNA into cytosol. Different pharmaceuticalmarkets, with different rate of success, had experiences
with oseltamivir, zanamivir and peramivir, applied through several dosing regimes and routes of administration and consequently
choice of drug forms used,
b. Bacterial super infections antibiotics treatment, most commonly situated in lower respiratory tract, according to available
evidence-based guidelines on hospital acquired pneumonia management,
c. Low dose cortisol analogues are proper only with developed respiratory distress syndrome and together with
neuraminidasesinhibitor. High doses are not recommended even as adjuvant therapy and have neither proven efficacy nor
safety in this indication.
From the moment that we find the isolation of a new flu virus, we must wait for the preparation of a new specific vaccine that will be ready for the next influence season in Autumn.
Keywords: COVID 19 Virus;influenza; flu; Avian; Swine flu
Discussion
An acute respiratory disease, caused by a novel coronavirus
(SARS-CoV-2, previously known as 2019-nCoV), the coronavirus
disease 2019 (COVID-19)19 has spread throughout China and
received worldwide attention. On 30 January 2020, World Health
Organization (WHO) officially declared the COVID-19 epidemic as a
public health emergency of international concern. The emergence
of SARS-CoV-2, since the severe acute respiratory syndrome
coronavirus (SARS-CoV) in 2002 and Middle East respiratory
syndrome coronavirus (MERS-CoV) in 2012, marked the third
introduction of a highly pathogenic and large-scale epidemic
coronavirus in to the human population in the twenty-first century.
As of 30 March 2020, a total of 4,87,137 confirmed cases globally,
deaths (7%) had been reported by WHO. Meanwhile, several
independent research groups have identified that SARS-CoV-2
belongs to β-coronavirus, with highly identical genome to bat
coronavirus, pointing to bat as the natural host.
The novel coronavirus uses the same receptor, angiotensinconverting
enzyme 2 (ACE2) as that for SARS-CoV, and mainly
spreads through the respiratory tract. Importantly, increasingly
evidence showed sustained human-to-human transmission,
along with many exported cases across the globe. The clinical
symptoms of COVID-19 patients include fever, cough, fatigue and
a small population of patients appeared gastro-intestinal infection
symptoms. The elderly and people with underlying diseases are
susceptible to infection and prone to serious outcomes, which may
be associated with acute respiratory distress syndrome (ARDS) and
cytokine storm. Currently, there are few specific antiviral strategies,
but several potent candidates of antivirals and repurposed drugs
are under urgent investigation. In this review, we summarized the
latest research progress of the epidemiology, pathogenesis, and
clinical characteristics of COVID-19, and discussed the current
treatment and scientific advancements to combat the epidemic
novel coronavirus1The history of flu viruses teaches that the
influence has origin from animals birds, generically aquatic, then
transfered to man through the leap into pigs. The promiscuity of
the herds, as it is in use in Asia, determines this transition and then
the spread.
The Spanish influence (1918, H1N1), the one from Asia (1957,
H2N2), that of Hong Kong (1968, H3N2) and so on have had this
originThe strains common in some years may have also relations
with those of other years.The persons mostly old people have
antibodies directed towards the antigens more important of the
strains with which they were in contact. With the progress of the
age it is a broader spectrum immunity that is reflected in antibodies
polyvalent made through the contact with many antigens primary
and secondary present in strains that they meet during the
following years. But each contact following with a flu virus of type
A involves not only specific antibodies, but also an increase in those
directed towards the strain responsible for the first flu infection of
the subject (phenomenon of Davenport or doctrine of original sin).
In this way, the immunization to a particular strain, spread
in a certain period, involves progressively increasing difficulty in
its further distribution and creates the selective advantage, for
some variant of the virus, to multiply and spread. Chloroquine and
Hydroxychloroquine and Emerging Viruses Chloroquine (CQ) is an
aminoquinoline known since 1934. It was synthesized to be used
as an antimalarial drug, but its properties and mechanism of action
encouraged its use for the treatment of Microorganisms 2020, 8,
85 8 of 26 different diseases. Currently, CQ and its hydroxy-analog
hydroxychloroquine (hydroxyCQ) cannot be used as antimalarial
drugs in wide areas where the resistance of malaria parasites
emerged.
They are commonly used for connective tissue disorders,
such as rheumatoid arthritis. Due to low toxicity and cost, high
tolerability and immunomodulatory properties, CQ and hydroxyCQ
have also been proposed for use against viral infections. Even if
their specific mechanisms in individual diseases are not clear, it
is well assessed that the antiviral activities of the aminoquinoline
take advantage of their strong anti-inflammatory activity. The
major proposed mechanisms of actions of CQ analogs which are
suggested to influence the anti-viral activity are, among the others:
the inhibition of cytokine production and release by T cells: IL-1, 2,
6, or 18, tumor necrosis factor TNF-α and IFN-γ, reduced levels of
chemokines CCL2 and CXCL10, inhibition of micro-RNA expression,
decreased TH17-related cytokines, decreased DNA, RNA and
protein synthesis in thymocytes (reviewed in).
The in vitro antiviral effect of CQ was first reported approximately
40 years ago, and since that time, its use as an antiviral drug has
been extensively discussed. In particular, CQ/hydroxyCQ have been
used for the treatment of emerging chikungunya virus (CHIKV)
infection, recently causing numerous outbreaks in the world.
Khan et al. showed that the treatment of infected Vero cells with
different micromolar concentrations of CQ reduced virus yield and
viral RNA copy number. De Lamballerie and colleagues confirmed
the inhibition of CHIKV replication in Vero-E6 cells using CQ. The
efficacy of CQ was inversely related to the concentration of the viral
inoculum used, an unfavorable observation, considering the high
viremia measured at the acute stage of CHIKV infection (up to 1010
virus copies/mL serum). Sourisseau and colleagues treated HeLa
cells with CQ, obtaining a potent inhibition of CHIKV replication and
its relative cytopathic effects. A double-blind placebo-controlled
trial was designed to evaluate the efficacy and safety of CQ for the
treatment of CHIKV infection in 2006 in French Reunion Island
(Indian Ocean).
No significant difference was observed between the CQ and
placebo groups, either in the mean duration of febrile arthralgia or
in the rate of viremia decrease. However, the number of patients
included in the study was too small to draw definitive conclusions
regarding the efficacy of CQ treatment Aminoquinolines were
proposed for the treatment of other viral infections, such as ZIKV.
In 2017, it was demonstrated that CQ and AQ exerted anti-ZIKV
activity in Vero cells, with low micromolar IC50s. These results
were in agreement with the decreasing number of ZIKV-infected
cells after CQ treatment. Additionally, CQ protected the cells from
further ZIKV infection, as measured by cell viability at noncytotoxic
concentrations. The activity of CQ has also been indirectly
demonstrated against DENV infection. The results obtained by
Kleber and colleagues showed that CQ suppressed TNF-α and IFN-γ
production, and it was hypothesized that CQ might be used to treat
patients suspected of having dengue disease, avoiding the more
severe form of dengue hemorrhagic fever and/or shock. A clinical
trial was also established to verify the effect of CQ versus placebo in
DENV-infected patients in Brazil.
CQ promoted a reduction in the intensity of pain and an
improvement in the well-being of patients with DENV infection but
did not alter the duration of the disease or the intensity and days of
fever. To study the effects of CQ against EBOV, a group led by Dowall
conducted an in vitro investigation using the human cell line MRC-5
and in vivo studies with the well-characterized guinea pig model.
They were able to demonstrate that CQ reduced EBOV replication
in MRC-5 cells. In contrast, the administration of CQ to 12 Guinea
pigs did not protect the infected animals against the Ebola disease.
Madrid and colleagues suggested that CQ could interfere with the
late stages of EBOV replication and assembly.
Despite these positive in vitro results, the clinical trials were
sometimes conflicting. For this reason, later, the literature was
reviewed to clarify the efficacy of CQ in the treatment of filovirus
infection. It was concluded that the efficacy of CQ against the
viruses belonging to this family was dependent on the CQ plasma
concentrations, which must be sustained in patients until the
clearance of the viremia. Microorganisms 2020, 8, 85 9 of 26 CQ was
shown to inhibit the replication and spread of coronavirus (CoV)
in vitro and to prevent infection with CoV in newborn mice. Since
the suppressive effect of CQ was also present when the cells were
treated before the infection, a prophylactic advantage of CQ use was
suggested. 4.2. Chloroquine and Hydroxychloroquine and HCV CQ
and its analogs have effects against HCV. In particular, the treatment
of JFH-1 or Huh-7 cells with CQ reduced HCV entry, replication,
and infection in a dose-dependent manner. Furthermore, CQ, in
combination with IFN-α, prevented the replication of HCV and
enhanced the antiviral effect of IFN-α. In this regard, two phase I/II
clinical trials were initiated to verify the efficacy of the combination
treatment of hydroxyCQ and ribavirin, but no results were posted
due to limited recruitment.
Chloroquine and Hydroxychloroquine and HIV The anti-HIV-1
and anti-HIV-2 activities of CQ and its analogs were tested in vitro
and in vivo. The first report about the in vitro use of CQ as an anti-
HIV-1 agent was published in 1990 by Tsai et al., which showed the
suppressive effects of CQ on the replication of HIV-1 in a T cell line.
A few years later, Sperber and colleagues confirmed these results,
showing the ability of CQ and hydroxy to inhibit HIV-1 replication
not only in T cells but also in monocytes. Subsequently, the same
group demonstrated the CQ and hydroxy anti-HIV-1 and anti-HIV-2
in vitro effects at concentrations that are clinically achievable.
CQ had an additive effect against HIV-1 when used in combination
with other antiretroviral agents. Naarding et al. demonstrated
that CQ reduced HIV-1 transmission to and replication in CD4 +
T-lymphocytes. Similarly, Martinson et al. observed that CQ had a
preventive role in HIV infection, reducing CD8 + T cell activation
upon HIV replication. The antiviral activity of hydroxy was
demonstrated in vivo by several clinical trials. The so ministration of
hydroxy was able to reduce the amounts of plasma HIV-1 RNA and
IL-6 in patients treated for eight weeks compared to those of the
placebo group. During a second clinical trial, hydroxyCQ was shown
to reduce the HIV-1 RNA plasma level, although at a lower level than
the antiviral zidovudine. In contrast, the results published in 2012
by Paton and colleagues showed negative results, with an increase
in viral load and a decrease in CD4 number.
Another double-blind, randomized placebo-controlled trial
testing the effects of CQ chronically HIV-infected persons was
conducted in Minnesota. The results showed that the administration
of CQ during chronic HIV infection resulted in decreased immune
activation, but no data regarding HIV status were reported. Very
recently, the AIDS Clinical Trials Group A5258 was completed. It
was a randomized, double-blind, placebo-controlled study in HIV-1-
infected participants off antiretroviral therapy and participants on
antiretroviral therapy. CQ modestly reduced immune activation in
antiretroviral therapy-treated HIV-infected participants. Finally, the
recruitment of 1499 patients was concluded a few months ago in a
randomized, controlled, open-label, phase III trial of the standard
of care with CQ prophylaxis compared to no prophylaxis in HIVpositive
patients in Malawi. Chloroquine and Hydroxychloroquine
and Other RNA Viruses CQ was shown to inhibit the in vitro
replication of H1N1 and H3N2 IAV strains. A phase II clinical trial
aiming to verify the effect of CQ compared to that of placebo on IAV
was started in Singapore in 2005, and 1516 patients were recruited.
However, CQ was not shown to prevent infection with IAV. CQ
had inhibitory effects on the entry and replication of enterovirus
(EV)-A71 in cell-based systems. Yong and colleagues studied
the efficacy of CQ against several EV serotypes and evaluated its
therapeutic capacity in vitro in RD cells and in vivo in a murine
model. They Microorganisms 2020, 8, 85 10 of 26 demonstrated
the potential of CQ as an antiviral in the treatment of hand, foot, and
mouth disease caused by EV infection. The positive results obtained
in the murine model of infection were indicative of the fact that CQ
may mitigate the disease severity in mammals. 4.4. Amodiaquine
and Emerging Viruses Amodiaquine (AQ) was originally developed
and has been widely used for the treatment of malaria. However,
subsequent studies revealed that it was active against a wide range
of human pathogens, including several viruses. In a study published
in 2014, it was investigated whether quinolone derivatives could
inhibit the replication of DENV. The time-course analysis suggested that AQ was stable and that it reproducibly inhibited DENV
infectivity. The data also showed that viral entry and internalization
were partially inhibited by the drug, but the major effect occurred
at a later stage of the viral life cycle. It is known that AQ inhibits
EBOV replication in vivo.
A recent study demonstrated that AQ was active against
severe fever with thrombocytopenia syndrome (SFTS) caused by
SFTS virus (SFTSV). 4.5. Primaquine Primaquine was tested as
an antiviral on primary chicken embryo cells (CECs) infected by
Newcastle disease virus . It was demonstrated that primaquine
had an effect on the accumulation of viral hemagglutinin on the
cell surface. In addition, primaquine inhibited protein synthesis in
virus-infected cells.
Dealing with suspected or confirmed H1N1 infection cases at different levels of care
We can easily notice that there are developed and from WHO
and CDC recommended procedures in handling the diseased.
In that sense from historical military sanitary doctrine it is well
known that epidemiological surveillance and proper organization
on the field are much more important for raising survival rates
than experienced physicians or quality of equipment available.
Therefore, in accordance with this we present an algorithm on
handling infected persons in primary care setting. Recommended
procedure differs significantly at secondary and terciary levels of
care and assumes selection and follow up of patients in line with
natural course of disease and response to treatment. In order to
more easily find the assistance on clinical decision making we gave
the next algorithm No 2. The new strains will be in conditions of
an increase in visitors, regardless of whether they have or not an
immunologic experience with the previous strains. As a result of
that, shortly after the appearance of a new type, the old forms will
disappear and the new family will become dominant for a period
which in general covers 10-20 years, in which there is, for the
emergence of minor antigenic variation, the subdivision in various
subtypes.
The outcrops of a new epidemic strain may, therefore, be
regarded as a process of development interesting the characteristics
of the strain and the susceptibility of the population. In order that
a strain has a wide distribution, its antigenic characteristics must
ensure that it escapes the neutralization of antibodies of the host and
of the surrounding population. So the outbreaks will happen with
those strains that have dominant antigens that fit the deficiency,
or better, the absences of antibody in the population. It seems, in
conclusion, that the flu virus showes an ability and an aptitude
for survival built on the possibility of emergence of new models
that allow the virus being confused easily through populations still
partly immune to previous antigenic formsAccording to this view,
the changes in the influenza A can be designed in single meaning,
in the context of a principle and of an evolutionary progress, from
Burnet said immunological drift or steering immunology (Figures
1,2). Very important to remember that it was demonstrated the
presence of antibodies to the more recent strains of 1957 Asian flu
(A2) in older segment of that population: in Asian influence there
were obviously strains with dominant characters, other than those
that had characterized the previous years, more or less, but similar
to those of the strains widespread much before(1889-90pandemic).
For the emergency created by epidemic of avian flu in Asia it was
right not to create panic as victims of a bad information [1].
Figure 1: Dissolve hydroxy chloroquine base in IPA and water. Charcoalize the RM at 700C-800C. Charge aqueousSulphuric acid to raw material solution at 450C–550C. Distill out 10-15% IPA.Filter the solid, wash with IPA. Dry the wet Hydroxychloroquine Sulphate at850C–950C.

The possibility that the avian virus entries in other parts of the
world it was like the rest for all types of flu viruses. It is clear that the
dead animal is harmless, and therefore there are other veterinary
and agricultural interests There is a potential risk of genetic
recombination with human flu viruses that might hesitate to a
viral variant capable of a transmission from human to human.Some
important medicinal natural herbs or plants used for the treatment
of swine flu Basil: Ocimum sanctum. and Ocimum basilicum also
known as Tulsi (Hindi) and Holy Basil (English), is an aromatic
plant of the family Lamiaceae. The plant, as a whole, is a treasure
house of potent compounds with its leaves, seeds, and roots, as well
as flower being medicinally important and is considered divine by
the Hindus. Ocimum sanctum. and Ocimum basilicum are great
Ayurvedic treatment option for swine flu.
Ayurvedic practitioners claim that basil not only keeps the
nasty swine flu virus at bay, but it also assists in the fast recovery
of an affected person. They claim that basil improves the body’s
overall defense mechanism, thereby increasing its ability to fight
viral diseases. It is also believed to strengthen the immune system
of the afflicted person. For the control and prevention of many
disease, Ocimum extracts are used in ayurvedic remedies for
common colds, headaches, stomach disorders, inflammation, heart
disease, various forms of poisoning, and malaria. For the control
and prevention of swine flu, basil must be consumed in the fresh
form. The paste or juice of a minimum of 25 leaves (medium size)
should be consumed twice a day. Moreover, it should be had on an
empty stomach. O. sanctum is considered to be an adaptogen par
excellence [2,3]. It harmonizes different processes in the body and is
helpful in acclimatizing to stress. The main chemical constituents of
O.sanctum are oleanolic acid, ursolic acid, rosmarinic acid, eugenol,
carvacrol, linalool, and β-caryophyllene [4].O.sanctum is reported
to be an effective treatment for diabetes and high cholesterol[5]
O.sanctum also shows promise for protection against radiation
damage [6,7].O.sanctum leaves contain highest percentage of
essential oils, infusion of which is given in malaria. Juice of the
leaves is taken internally and is very effective in skin diseases such
as itches fungal infections. Fresh leaves also cure chronic fever and
when mixed with honey and ginger juice, it is useful in cough and
bronchitis [8]. The antimicrobial properties of O. sanctum make it
useful for the prevention of novel H1N1 flu. Basil is safe, with no
side effects and is great to prevent swine flu from spreading like
wildfire
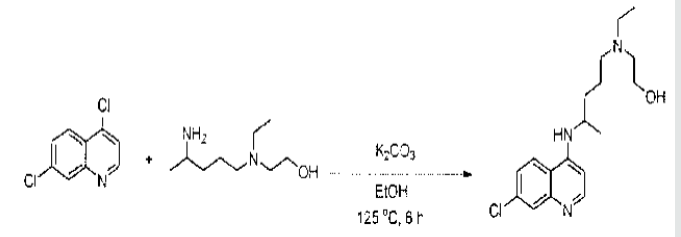
Hydroxychloroquine sulfate
A. Manufacturing process
a. STAGEHydroxychloroquine Base: Charge 4,7-Dichloroquine,Hydroxynovaldiamine, Sodium carbonate and Potassium iodide. Heat the RMto 1250C–1300C and maintain for 60-70 hours. Cool the RM to RT. Extractthe RM in Ethyl acetate. Wash the Ethyl acetate layer with water. Acidify theRM with glacial Acetic acid to remove impurities. Then extract the RM inEthyl acetate at pH 12–13. Distill out solvent, charge Ethyl acetate to RM.Charcoalize the RM. Crystallize the Hydroxy chloroquine at 00C–100C.Hydroxy chloroquine base is again crystallizing from Ethyl acetate at 00C–100C. Dry the wet hydroxy chloroquie base at 500C–550C.
b. Stage Hydroxychloroquine Sulphate: Dissolve hydroxy chloroquine base in IPA and water. Charcoalize the RM at 700C-800C. Charge aqueousSulphuric acid to raw material solution at 450C–550C. Distill out 10-15% IPA.Filter the solid, wash with IPA. Dry the wet Hydroxychloroquine Sulphate at850C–950C.In the course of epidemic of avian influenza that struck in 2005 10 Asian countries (China, Pakistan, Thailand, Cambodia, Indonesia, North Korea, South Korea, Taiwan, Laos, Vietnam) with 80 million chickens died or sacrificed and 42 fatal human cases it was identified H5N1 as an etiologic agent, the same as the one that in 1997 had caused an epidemic outbreak in Hong Kong with 18 human subjects infected and 6 dead and with the sacrifice of 1.5 million chickens [9].
The Avian Influenza recent outbreaks with involvement of viral strains as H9N2 in 1999, infected two children and other individuals, and in 2003, infected a boy in Hong Kong, while H5N1 hit three subjects of a family killing two in 2003. At the same time in the Netherlands an epidemic from avian influenza viruses H7N7 hit 83 people and led to death a veterinarian.In 2005 in the USA outbreaks of avian influenza have been identified in Texas and in Delaware (virus H7N2), and in the last State together with territories of Maryland and of Virginia there are working 14,000 people and 1,900 families that produce the 8% of the meat of American poultry, with a budget of one and a half billion dollars. In 2003 the American export in Europe has reached the share of eight million and eight hundred thousand eggs and 452 thousand chicks, respectively for 20 million and 3 million of Euro.
For the emergency created by the epidemic of “influence of the
pigs” in Mexico it is correct not to create alarmisms being victims of
a bad information [10]. The possibility that the virus arrives in other
parts of the world is real as for all the types of influence virus [11].
For the SARS a direct contact was necessary, in practical terms the
so-called droplets of Pflugge, for this swine influence it is different,
in fact, it also spreads through the air to distance. And a potential
risk exists of a panic syndrome that it often happens through a bad
information or a scarce knowledge of the phenomenon. Then no
alarmism because the number of the victims is decidedly inferior
to other pandemics [12].Few years ago there was the announce in
Naples of an outbreak of disease of Newcastle in a group of parrots
coming from Pakistan. The disease of Newcastle represents an
useful paradigm of the influence infection in man.
Fortunately there is not a reported human pathology to
this virus, for which the discovery of the outbreak of disease of
Newcastle in Naples did not give worries of any sort for the health
of the Neapolitans. Finally, the risks of the disease of Newcastle are
more tied to the breeding’s of home volatile that, not immune to this
virus, can be exposed to the epidemic [13].The vaccination against
the influence is the most effective method to prevent the illness.
From the moment that we find the isolation of a new flu virus, we
must wait for the preparation of a new specific vaccine that will be
ready for the next influence season in Autumn.The antiviral drugs
(inhibitors of the neuraminidasis, receptor of the virus surface)
should be assumed within 48 hours by the appearance of the
influence symptoms and the subjects that have had a close contact
with people infected by the flu virus.In conclusion The history of
flu viruses teaches that influence originates from birds, usually
aquatic, then it is transferred to man through the leap into pigs.
The promiscuity of the herds facilitates this transition and then
the spread. Three pandemics caused by influenza A viruses, which
occurred in the 20th century, have all had this origin: the ‘Spanish
flu’ (1918, H1N1), the ‘Asian flu’ (1957, H2N2) and the ‘Hong Kong
flu’ (1968, H3N2). The 2009 H1N1 influenza virus acted during
the winter in Australia and New Zealand yielding a pattern effect
for the treatment of patients during the winter in the Northern
Hemisphere. The performance of rapid diagnostic test for the
detection of novel influenza A (H1N1) virus was evaluated by the
Centers for Disease Control and Prevention. In the absence of any
vaccines to prevent COVID-19, there are many clinical trials (CT)
taking place to find a treatment. These CTs are mainly focusing on
either repurposing or repositioning the existing molecules. WHO
has published a landscape of therapeutics which could be used for
treating COVID-19, and some of them are undergoing CTs as well?
Generally speaking, patents are not a concern when it comes to old
molecules under CTs because these molecules are already out of
patent protection.
However, a few of these molecules are still under patent
protection in many countries. Two in particular – Remdesivir and
Favipiravir – are under patent protection in India. The generic
availability of these medicines can facilitate compassionate use and
CTs in India without depending on supply from the patent holders.
Therefore, the Government of India should use the compulsory
licence or government use license to facilitate the generic production
of these medicines.Some of these medicines are very old molecules,
such as chloroquine and hydroxychloroquine, and some are of very
recent origin, such as the Lopinavir-Ritonavir combination, which
is used to treat HIV/AIDS. Some of these new molecules are still
under patent protection in many countries, which are currently
using a part of CTs or compassionate use to treat COVID-19 [14-17].
Responding to the patent barriers, a few countries like Israel and
Chile have issued compulsory licenses to allow a generic company
to produce or use the patented medicine/invention without the
permission of the patent holder. Germany even amended its patent
law to facilitate the quick issuance of compulsory licenses, while
Canada will do so soon. The national assembly of Ecuador passed a
resolution empowering the health minister to issue these licenses.
Now, there is one patent granted to Remdesivir, and our search
found another application is pending. The recently granted patent
will expire only in 2035. Favipiravir is already part of five patents in
India, although one of these patents has already expired. However,
only a detail analysis can reveal whether existing patents block
generic manufacturing.
Remdesivir is believed to be the most promising drug which
can be used for treatment against COVID 19. It is a broad-spectrum
antiviral drug, which is currently undergoing clinical trials in China.
The results are expected towards the end of April. The drug works
by inhibiting the virus’s ability to replicate itself inside cells. This
indicates that the drug would effective even when a person is in
the initial stage of infection and the virus is still reproducing in the
upper respiratory tract.The findings of severe respiratory disease
concurrent with the circulation of H1N1 influenza was proved by
the aforementioned test. Even the potential impact of pandemic
influenza during the Hajj pilgrimage was taken in account to reduce
the substantial effect on the crowd to spread the infection.
References
- Enserink M, Cohen J (2009) Virus of the year. The novel H1N1 influenza. Science326(5960): 1607.
- SwineCustom Vaccines. Novartis.
- Swine FluThe predictable pandemic.
- Influenza FactsheetCenter for Food Security and Public Health, Iowa State University.
- Ramirez A, Capuano AW, Wellman DA, Lesher KA, Setterquist SF (2006)Preventing Zoonotic Influenza Virus Infection. Emerging Infect Dis12 (6): 996–1000.
- (2009) Key Facts about Swine Influenza (Swine Flu) – Diagnosis. Centers for Disease Control and Prevention.
- (2009) Chlorine Bleach: Helping to Manage the Flu Risk. Water Quality & Health Council.
- Selfprotection measures. LHC.
- Tarro G, Esposito C (2011) Emerging viral agents at risk in global health approaches to early diagnosis and prompt therapy. International Conference on Bioinformatics and Computational Biology (BIOCOM’11).
- Chowell G (2009) Severe respiratory disease concurrent with the circulation of H1N1 influenza”.New England Journal of Medicine361(7): 674-679.
- Dawood FS (2009) Emergence of a novel swine-origin influenza A (H1N1) virus in humans. New England Journal of Medicine360(25): 2605-2615.
- Lister P (2009) Swineorigin influenza virus H1N1, seasonal influenza virus, and critical illness in children. Lancet 374(9690): 605-607.
- Morens DM (2009) The persistent legacy of the 1918 influenza virus. New England Journal of Medicine361(3): 225-229.
- Esposito C (2012) Tracking the 2009 H1N1 influenza virus in the Italian region Campania. Journal of Cellular Physiology227(7): 2813-2817.
- (2009) WHOClinical management of human infection withpandemic (H1N1). revised guidance.
- (2009) WHOClinical management of human infection with pandemic (H1N1) 2009 virus infection, nterimGuidance from Expert Consultation.
- COVID-19

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...