Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1725
Opinion(ISSN: 2641-1725) 
Dynamic Magnetic Resonance Imaging Provides Insight into Upper Airway Mechanics in Obstructive Sleep Apnoea Volume 4 - Issue 5
Michael J Brennick*
- Pulmonary and Critical Care Medicine, Department of Medicine, Johns Hopkins University, USA
Received: January 31, 2020; Published: February 13, 2020
*Corresponding author: J Brennick, Pulmonary and Critical Care Medicine, Department of Medicine, Johns Hopkins University, Baltimore, MD, USA,
DOI: 10.32474/LOJMS.2020.04.000197
Keywords: Obstructive Sleep Apnoea (OSA); Magnetic Resonance Imaging, Upper Ariway, Regional Respiratory Movement, Sleep Disordered Breathing
Opinion
Key point of interest
This perspective should help in the interpretation of the paper:
‘Regional respiratory movement of the tongue is coordinated during
wakefulness and is larger in severe obstructive sleep apnoea’ by
Juge et al. [1]. A powerful imaging technique for dynamic motion
tracking has been used to better understand airway mechanics.
Linked articles: This Perspectives article highlights an article
by Juge et al. [1]. To read this paper, visit https://doi.org/10.1113/
JP278769.
A new study from Juge et al. [1] from L. Bilston’s laboratory
gives us basic information on tongue motion from mechanical
analysis of motion-tagged magnetic resonance imaging (MRI) in a
cross-section of normal and severe sleep apnoea patients (N 63).
They concluded, contrary to their own hypothesis, that in the awake
state severe sleep apnoea patients have greater tongue motion,
including coordination of regional tongue muscle compartmental
motion compared to normal patients without sleep apnoea, based
on the apnoea hypopnoea index (AHI) Although only a relatively
limited cross-section was studied (without strong statistical
power to control for weight and other variable factors), the basic
correlation between increased severity of sleep apnoea (as AHI)
and awake tongue motion was shown. Not surprisingly, this result
is consistent with seminal work by Mezzanotte et al. [2] who
also found that genioglossus EMG was increased in awake sleep
apnoea patients. However, the current paper by Juge et al. [1] is not
simply a validation of EMG-based studies because analysis of MRItracked
tissue motion in the upper airway gives direct insight into
respiratory-related mechanical changes. The final output measure,
i.e. displacement of the tongue and soft tissues, does not rely on
assumptions of geometry that would include the length–tension
relationship of EMG to muscle force and displacement [3].
MRI tagging for muscle motion analysis has been predominantly
used for cardiac analysis and differential diagnosis in myo- cardial
infarcts [4]. However, in a series of papers, including Brown et al.
[5], this powerful technique has been used in the Bilston lab to
help understand the pathogenesis of obstructive sleep apnoea.
Thus, although sophisticated tools are available in MRI, knowledge
of how to apply MRI to help understand mechanics in the upper
airway requires experience and carefully designed experiments
because data overload can often sidetrack an important objective.
One of the key aspects to this study is the way in which the upper
airway geometry is visualized and regionally divided to maximize
tongue motion data collected in two-dimensional (2-D) (midsagittal
sections but with high time resolution so that movement is revealed.
One drawback to this study was that airway dimensions and some
airway soft tissues such as the soft palate were obtained from spinecho
and Dixon fat discriminating image protocols that do not
resolve the acquisitions in the same respiratory time frame as the
fast gradient echo motion-tagged imaging used for the midsagittal
tongue images. However, the lack of 3-D time-resolved analysis
does not prevent us from learning valuable information from the
overall data and results in this paper.
The current study has an a priori system to examine tongue
regions and within those defined regions so that soft tissue motion
evaluation can be averaged and compared between patients. Thus,
the concept of ‘coordinated motion’ among regions in the tongue
was assessed and those results were compared across the patient
data set.
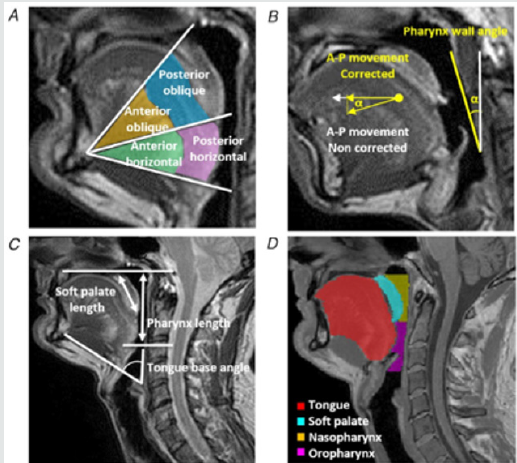
The regions as shown in Figure 1 of Juge´ et al. [1] were
chosen from previous work from this lab where patterns of tongue
movement were characterized. When the genioglossus fibres
contract during neural activation, tongue motion in the anterior
direction results and this tongue protrusion can lead to either
nasopharyngeal and oropharyngeal airway dilatation or both.
Thus, a physiologically relevant method of examining motion of the
tongue has been used and this approach helps to compare tongue
motion that is not always the same in the overall population,
whether normal or apnoeic patients are studied.
Finally, the conclusions from this work support the view that patients with severe sleep apnoea have to compensate for an existing predisposition to occlusion during awake breathing by tongue activity and tongue motion that is both co-ordinated in several regions of the tongue and is quantifiably greater in displacement that tends to dilate the upper airway. These findings should be of great inter- est towards the continued development of hypoglossal electrical stimulation treatment methodologies, especially where selective hypoglossus nerve stimulation may be used to augment specific regions of tongue motion at specific respiratory phases for the most efficient improvement of airway patency in predisposed individuals.
Additional Information
Competing interests
The author declares no conflicts of interest.
Author contributions
The author is the sole contributor to the essay.
References
- Juge´ L, Knapman F, Burke PGR, Brown E, Bosquillon de Frescheville AF, et al. (2019) Regional respiratory movement of the tongue is coordinated during wakefulness and is larger in severe obstructive sleep apnoea. J Physiol598(3): 581-597.
- Mezzanotte WS, Tangel DJ, White DP (1992)Waking genioglossal electromyogram in sleep apnea patients versus normal controls (a neuromuscular compensatory mechanism). J Clin Invest 89(5): 1571-1579.
- Gordon AM, Huxley AF, Julian FJ (1966)The variation in isometric tension with sarcomere length in vertebrate muscle fibres. J Physiol 184(1): 170-192.
- Axel L, Dougherty L (1989)MR imaging of motion with spatial modulation of magnetization. Radiology 171(3): 841-845.
- Brown EC, Cheng S, McKenzie DK, Butler JE, Gandevia SC, et al. (2013)Respiratory movement of upper airway tissue in obstructive sleep apnea. Sleep 36(7): 1069-1076.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...