Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1652
Research Article(ISSN: 2641-1652) 
Ursodiol Infusion in the management of Severe Alcoholic Hepatitis: An Investigational Clinical Trial Volume 4 - Issue 2
Malladi Umadevi1*, Veerendra Kumar Panuganti3, Dinesh Kumar reddy2, Pusapati Madan Ranjit3, Bhaavan Paladugu2, Tony Pious2, Purushothama Narasiyappa4, Krishna Murthy T4 and Pradeep Shivakumar4
- 1Professor, Department of Gastroenterology, Osmania General Hospital, India
- 2Residents in Department of Gastroenterology, Osmania General Hospital, India
- 3Clinical Affairs Department, Shilpa Medicare Limited, India
- 4Shilpa Medicare Limited, R & D, India
Received: November 10, 2022; Published: November 21, 2022
*Corresponding author: Malladi Umadevi, Professor, Department of gastroenterology, Osmania General Hospital, India
DOI: 10.32474/CTGH.2022.04.000181
Abstract
Background and Aims: Alcoholic hepatitis (AH) are an acute hepatic inflammation associated with significant morbidity and mortality in patients, who consume excessive amounts of alcohol. The currently recommended treatment options (corticosteroid, pentoxifylline) mainly target only on reduction of inflammation response in AH. None of these recommended drugs were unable to reduce the elevated bile enzymes. A pathophysiologically oriented approach for management of alcoholic hepatitis with Ursodiol injection with the objective of knowing improvement in mortality rate, liver parameters, Model for end-stage liver disease (MELD) & Maddrey’s Discriminant Function (MDF) scores, and quality of life.
Methods: An investigational clinical trial was conducted in the Department of Gastroenterology, Osmania General Hospital. A total of two Arms in the study, 04 patients were on treatment arm A [standard of care (SOC) + supportive therapy] and 06 patients were on treatment Arm B [Ursodiol injection 3500mg/day in two divided doses + standard of care + supportive therapy.
Results: Our study observed a lower mortality (14.28%) rate in Ursodiol injection + SOC arm B when compared to the SOC arm A (20%). Our results were correlated with published literature on lower bilirubin levels on 7 and 14 associated with a favorable prognosis and increased survival. The quick reduction of toxic bile levels with parenteral administration of Ursodiol might be beneficial for severe alcoholic hepatitis patients in critical conditions. There was a gradual reduction of MDF & MELD scores of patients in both treatment arms from day 0 to day 60 and not statistically significance was observed when compared to the two arms but % change from baseline was improved numerically in arm B from day 14 to day 60 when compared to arm A. We have observed, patients with arm B (Ursodiol injection+ SOC+ ST) presented significant improvement in quality of life (SF-36) when compared to SOC arm A. Rapid improvement was observed in energy, physical health, physical functioning, sleep, appetite emotional well-being, and abdominal pain at day 7 of Ursodiol+ SOC arm.
Conclusion: Parenteral infusion of Ursodiol in severe alcoholic hepatitis at intensive care unit (ICU), resulted in marked improvement in elevated liver function tests and improvement in quality of life, statistically significant reduction in serum bilirubin and improvement in serum albumin and protein levels which are continued at 60 days also with no side effects. Earlier research studies also concluded that decreased total bilirubin on day 7 and 14 was associated with lower mortality and thereby clinical improvement. Severe alcoholic hepatitis is associated with a high mortality rate due to comorbid conditions such as severe kidney injury, uncontrolled sepsis, and active gastrointestinal bleeding, these conditions might hamper study outcome if not properly standardized during inclusion and exclusion in the study. So, in order to establish adequate efficacy and safety of Ursodiol injection, a well-organized, randomized, controlled large clinical trials need to be conducted in, with repeated treatment cycles in severe alcoholic hepatitis patients.
Alcoholic hepatitis; MDF; MELD; SF-36; LFTs
Introduction and overview
Alcoholic hepatitis (AH) is an acute hepatic inflammation associated with significant morbidity and mortality in patients, who consume excessive amounts of alcohol [1]. In its most severe form, short-term mortality of AH remains high (20–30%) probably due to poor patient characterization and the need for modern targeted therapies [2]. The Model for end-stage liver disease (MELD) and Maddrey’s Discriminant Function (MDF) scores are used to assess the severity of alcoholic hepatitis [3,4]. Chronic alcohol consumption leads to liver inflammation and liver injury. Inflammatory cytokines, such as TNF-α and IFN-γ were induced from liver injury in alcoholic liver disease (ALD) [5]. Severe alcoholic hepatitis is manifested by increased total bilirubin (TB) levels over 10–15 mg/dL, levels of aspartate aminotransferase (AST) between 100 and 200 U/L, and alanine aminotransferase (ALT) in the range of 50–150 U/L [6] and other typical clinical findings include fever, leukocytosis, ascites, and hepatic toxicity & encephalopathy [7]. It is generally assumed that exposure of hepatocytes to high concentrations of toxic bile acids is mainly responsible for liver injury and inflammation [8].
Currently, corticosteroid therapy is the most effective treatment option even though it is helpful in only about 60% of AH patients and only significant in reducing short-term mortality i.e one-month [9,10]. However, patients with severe alcoholic hepatitis (SAH) who developed an infection after corticosteroid treatment have a higher rate of mortality [11]. Whereas several other treatment options (Pentoxifylline, Infliximab, Etanercept) demonstrated initial benefit, but most of the studies failed in efficacy and even have harmful effects. The currently recommended treatment options (corticosteroid, pentoxifylline) mainly target only on reduction of inflammation response in AH. None of these recommended drugs were unable to reduce the elevated bile enzymes [Aspartate aminotransferase (AST), alanine aminotransferase (ALT)] and total bilirubin (TB) and other liver parameters] in AH. Current therapies such as corticosteroids and pentoxifylline fail in many alcoholic hepatitis patients and there is a clear need to develop new pathophysiologically oriented approaches.
Ursodiol is beneficial in patients with alcohol-induced hepatitis?
Earlier research confirmed that Ursodiol protected the alcoholinduced damage in human hepatocytes cell line [12] and alcoholinduced liver mitochondria injury in rat models [13]. Ursodiol (UDCA) is normally present in only 4% amounts in the bile and is approved for the treatment of various cholestatic disorders [14,15]. Several reports explained that Ursodiol is a hydrophilic bile acid that protects the liver from damage.
Multiple mechanisms of action of Ursodiol have been described as follows
a) Protection of injured cholangiocytes against toxic effects of bile acids.
b) Stimulation of impaired biliary secretion.
c) Stimulation of detoxification of hydrophobic bile acids.
d) Inhibition of apoptosis of hepatocytes [16]. Ursodiol is a well-known hepatoprotective agent, that inhibits the pro-inflammatory cytokines like TNF-α, IL-1β, IL-2, and IL-4 at the mRNA and protein levels and also increased an antiinflammatory cytokine interleukin 10 (IL-10). Ursodiol has strong antioxidant properties and remarkable scavenging efficiency.
In a placebo-control study of alcoholic hepatitis, Ursodiol was given orally (15 mg/kg per day) for 4 weeks and showed an improvement in liver parameters [17]. In alcohol-induced hepatitis patients with very higher levels of serum hydrophobic bile acids could be cytotoxic, which shall further deteriorate liver hepatocytes. Due to the high first-pass metabolism, only 30% of Ursodiol is bioavailable after oral administration. To achieve 100% bioavailability resulting in higher liver uptake a parenteral formulation Ursodiol injection has been developed. The safety and tolerability of parenteral Ursodiol injection was established up to 10,500mg/day in healthy humans and also efficacy has been studied in various liver disorders such as NASH, gallbladder transparent stones, and as an adjuvant during cancer chemotherapy, where significant reduction of abnormal liver parameters at the dose of 3,500 mg/day has been established [18-21].
Based on the Ursodiol mechanisms and available literature data from public domain, we hypothesized that the Ursodiol injection form could be beneficial in alcoholic hepatitis patients. Experimental studies of o fluorescent biliary salts showed that oral bile salts administration would not have homogenous effects on the whole liver parenchyma. Administration of bile salts into the systemic circulation, the bile salts were reaching to the liver via the hepatic artery to achieve regular distribution and homogenous uptake by the sinusoid’s portal spaces and widespread function to reach a higher concentration with the faster and better pharmacological outcome. The objective of the study is to establish the safety and to know the beneficial effects of Ursodiol injection in severe alcoholic hepatitis patients in terms of mortality rate, improvement in liver parameters, MELD & MDF scores and quality of life.
Patients and Methods
Ethical requirement
The study was conducted at Osmania General Hospital, 15-5- 104, Begum Bazar, Afzal Gunj, Hyderabad, Telangana 500012, India. The study document such as study protocol, Informed Consent Documents (ICD) (English and local language), and Case report form (CRF) were approved by the Institutional Ethics Committee (IEC). The study was conducted from March 2022 to October 2022 and the study was registered in the clinical trial registry, India, CTRI/2022/03/041331 [Registered on: 24/03/2022].
Patient recruitment
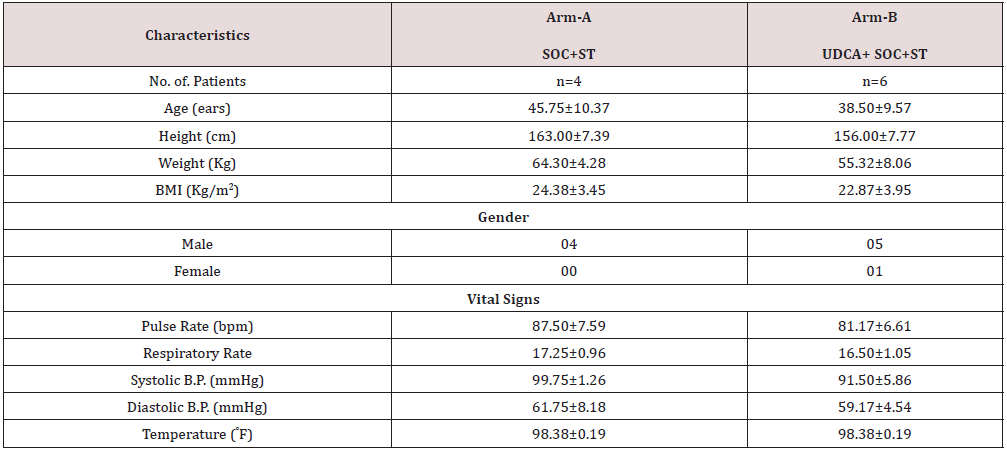
All Alcoholic hepatitis patients with AST and ALT levels ≥ 1.5 times upper normal limit (UNL), Model for End-Stage Liver Disease (MELD) ≥ 20 and Maddrey MDF ≥ 32 were enrolled for the study. A total of 20 patients were registered in the study. Among the enrolled patients, 02 patients were screen failed and 18 patients were selected for the study dosing. Among the 18 patients, 06 patients were withdrawn from the study period due to alcohol consumption after enrollment in the study and 10 patients completed the study period. 02 patients died in each arm due to co-morbid conditions. Among 10 patients, 04 patients were on treatment arm A standard of care [Pentoxyphylline tablets 400mg + N-acetyl cysteine tablets 300mg + S-Adenosyl L-Methionine tablets 200mg] + supportive therapy [Multivitamin capsules and calcium carbonate 500 mg & vitamin D3 250 IU tablets] and 06 patients were on treatment Arm B [Ursodiol injection 3500mg/day in two divided doses + standard of care [Pentoxyphylline tablets 400mg + N-acetyl cysteine tablets 300mg + S-Adenosyl L-Methionine tablets 200mg + supportive therapy [Multivitamin capsules and calcium carbonate 500 mg & vitamin D3 250 IU tablets].
Study Design
Patients were randomly assigned to receive either Arm-A or Arm-B. In Arm B, a total dose of 3500mg/day of Ursodiol injection was given daily for 7 days in two divided doses and standard of care + ST was continued for the period of 60 days. In Arm A standard of care + ST was continued for the period of 60 days. The study parameters were, mortality rate (Day 3 and day 60), improvement in liver parameters (Day 0,3,7,14, 30, 45 and 60), MELD & MDF scores (Day 0,3, 7, 14, 30, 45 and 60) and quality of life (Day 0, Day 7 and Day 60) were assessed on respective days.
Inclusion Criteria
The study includes patients with age between 21-70 years of patients, who were on recent alcohol consumption > 50g/day for > 6 months, continuing within two months before enrollment. Patients with AST and ALT levels ≥ 1.5 times upper normal limit (UNL), MELD ≥ 20, and MDF ≥ 32 were enrolled for the study.
Exclusion Criteria
Patients shock, signs of uncontrolled systemic infection (Fever > 38°C) and positive blood or ascites cultures and on appropriate antibiotic therapy for ≥ 3 days within 3 days of inclusion, Patients with acute gastrointestinal bleeding requiring >2 units blood transfusion within the previous 4 days, Patients Positive with HBsAg and HCV antibody, history of treatment for tuberculosis; history of any malignancy except skin cancer but including hepatocellular carcinoma within the last five years, Patients with positive HIV infection, recent previous treatment with corticosteroids or other immunosuppressive medications including specific anti-TNF therapy, calcineurin inhibitors within the previous 3 months, Treatment with corticosteroids for > 3 days within 3 days of inclusion, serious cardiac, respiratory or neurologic disease or evidence of other liver diseases such as autoimmune hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, Wilson disease, hemochromatosis, secondary iron overload due to chronic hemolysis, alpha-1-antitrypsin deficiency and Acute or chronic kidney injury with serum creatinine > 3.0 mg/dl and Patients with COVID-19 positive were excluded from the study.
Study variables
Demographic (age, gender, height/length, weight), vital signs [PR (bpm), RR (breaths per min), systolic and diastolic BP (mm Hg), and body temperature (°F)] were recorded on day 0, day 1 to 7, and day 14, day, 30 and day 45 and end day of the study period. The clinical parameters (Hemograms, LFTs, Lipid profile, Serum electrolytes, Serum creatinine, PT, PTINR) were collected at the time of screening and followed by day-3 and 7. Further clinical chemistry was performed on Day 14, Day 30, Day 45, and the end day (Day 60) of the study period. Both MELD and MDF are calculated on day 0 (Before dosing) and day-3, day-7,14,30, 45, and 60. The safety assessment was performed up to the end of the study (i.e 60 days). Quality of life (SF-36) assessment was performed on day 0, day 7, and day 60.
Investigational product administration
A dose of 3500 mg/day Ursodiol injection (25mg/ml) was administered via infusion route in two divided doses. 25 ml of Ursodiol Injection 25 mg/ml was transferred to 100mL of 0.9% NaCl to make the final concentration of 5.0 mg/mL of Ursodiol in 0.9% NaCl. The investigational product was administered as an intravenous infusion via an infusion pump over a period of 60 minutes (±5 minutes) for Arm- B. For Arm B test product was administered daily for 7 days and standard of care (SOC) was continued for the treatment for a period up to day 60. For arm A, only standard of care (SOC) was administrated orally daily up to day 60.
Adverse events
All observed adverse events (AEs) were documented between the start of the study treatment till the end of the study visit or early termination visit (ET).
Statistical Analysis
Statistical analysis was done in SAS 9.4. ANCOVA was used for statistical analysis considering percent change from baseline as a dependent variable, Treatment group as the independent variable, and Baseline as a covariate. A level of significance of 5% was considered for testing the significance of the p-value. ANCOVA was done using Proc GLM in SAS version 9.4. Quantitative data are expressed as means ±SD.
Results
Baseline Characteristics of the Patients
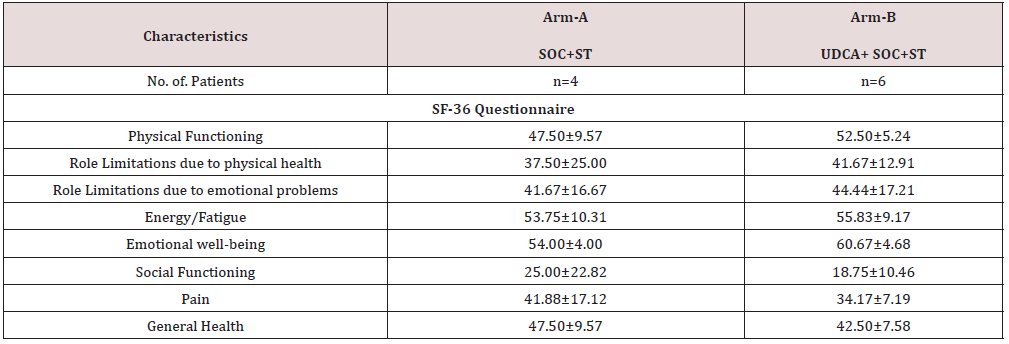
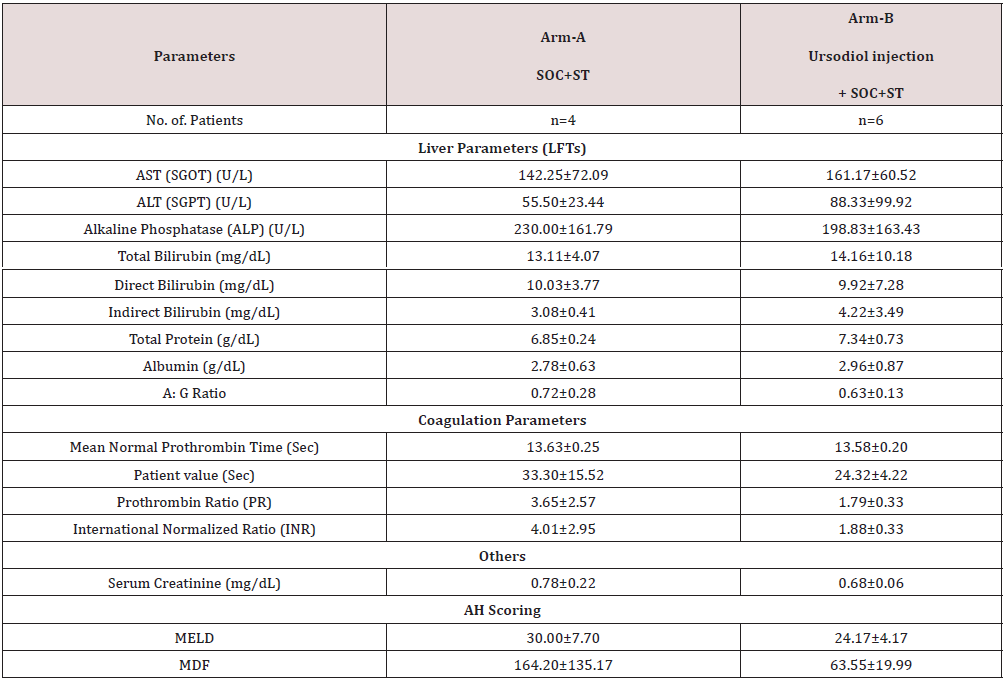
All the patients who fulfilled eligibility criteria for Alcoholic hepatitis (AH) were of the mean age of Arm-A is 45.75 ± 10.37 and Arm-B 38.50 ± 9.57. A total number of 10 patients completed the end of the study. The baseline characteristics (number of patients and demographic data, Vital signs, quality of life (SF-36), and parameters [Liver parameters (LFTs), coagulation parameters, AH scorings] were recorded in Tables 1A-1C for both arms.
Mortality
There were two mortalities observed in each arm during day 3 due to comorbid conditions. These two subjects were not considered for statistical analysis.
Laboratory Values and AH score
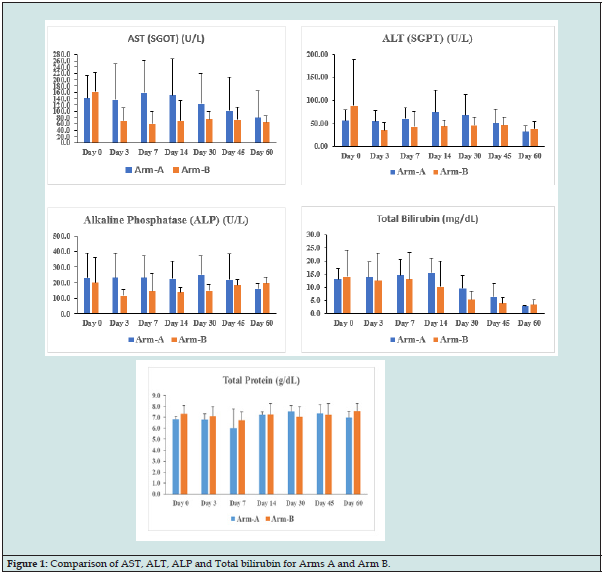
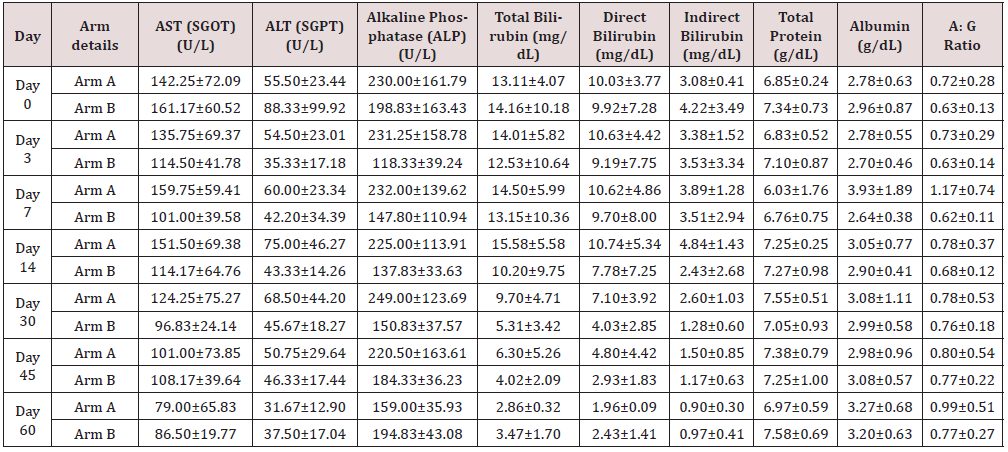
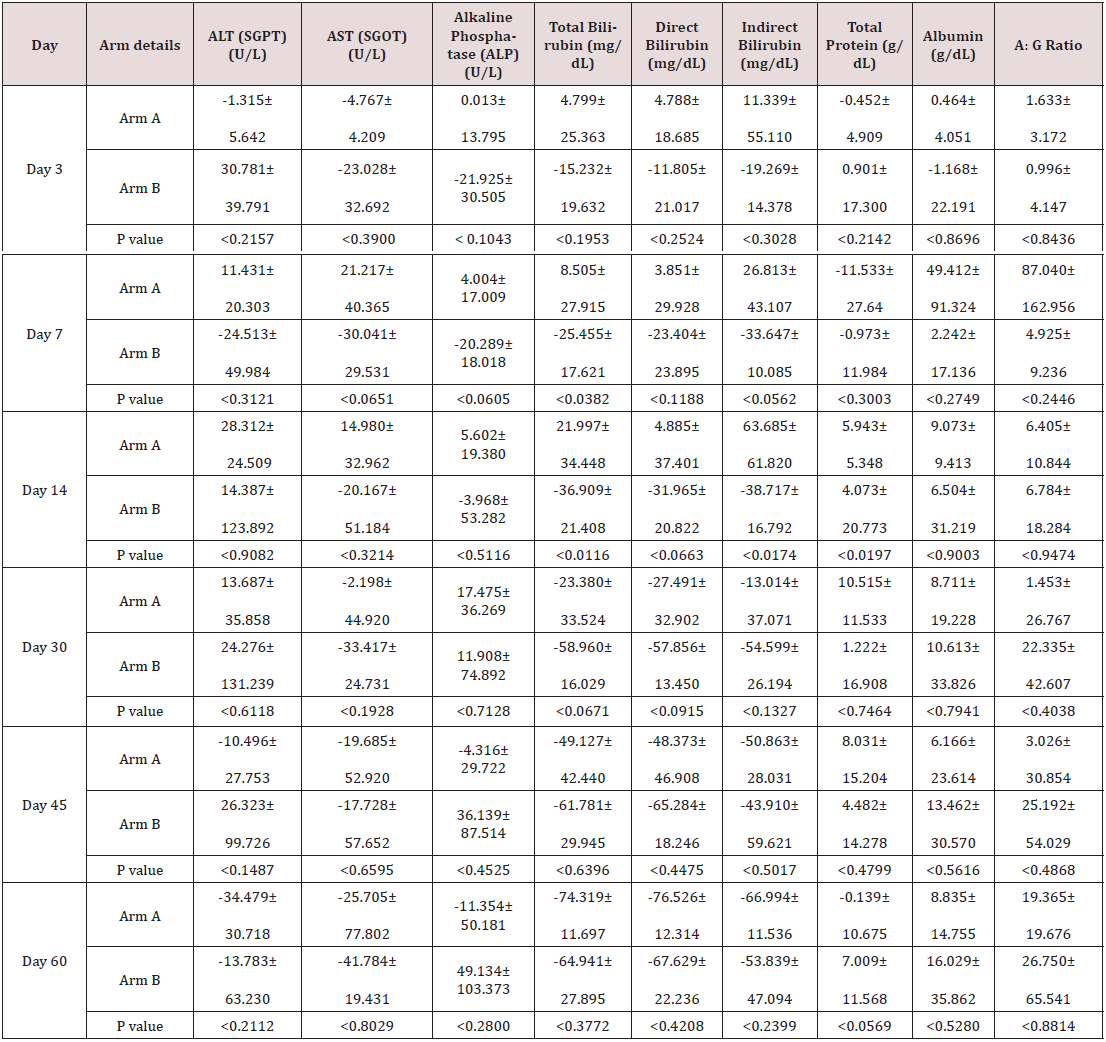
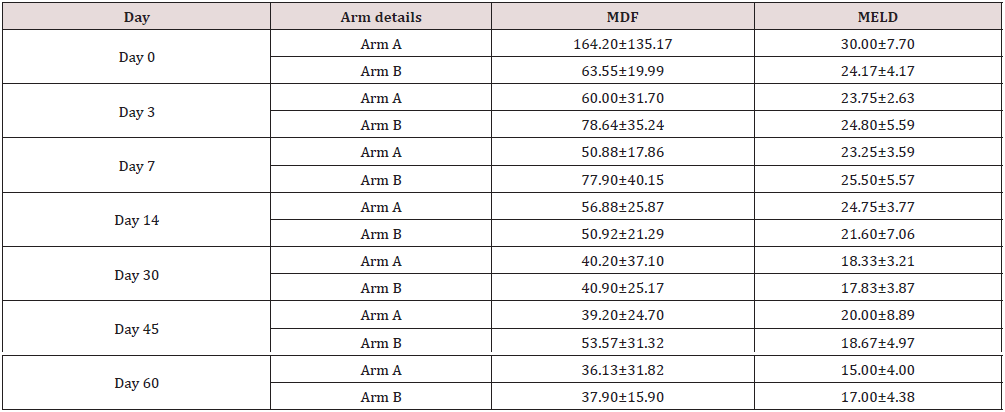
The mean key LFTs (AST, ALT, and Total Bilirubin) values were decreased (Table 2), (Figure 1) in both treatment arms from baseline (Day 0) to day 3, 7, 14, 30, 45, and 60 of post-treatment. The percent (%) change in reduction (Table 3), of key LFTs from baseline (Day 0) up to day 60 was calculated. Among LFTs, total bilirubin (mg/ dL) showed statistically significant (p<0.0382; P<0.016) with percent (%) change in reduction elevated value from baseline (Day 0) to day 7 and day 14 respectively. There were improvements in total protein levels significant (p<0.0569) higher in the Ursodiol injection Arm on day 60. The rest of the liver parameters were numerically improved in Arm B. There was a gradual reduction of elevated MDF & MELD scores of patients in both treatment arms from day 0 to day 60 (Tables 4 & 5) and not statistically significant was observed when compared to the two arms but a numerical % change improved in Arm B was observed from day 14 to day 60 when compared to Arm A.
Arm A: SOC+ST; Arm B: Ursodiol injection + SOC+ST.
Arm A: SOC+ST; Arm B: Ursodiol injection + SOC+ST.
Arm A: SOC+ST; Arm B: Ursodiol injection + SOC+ST.
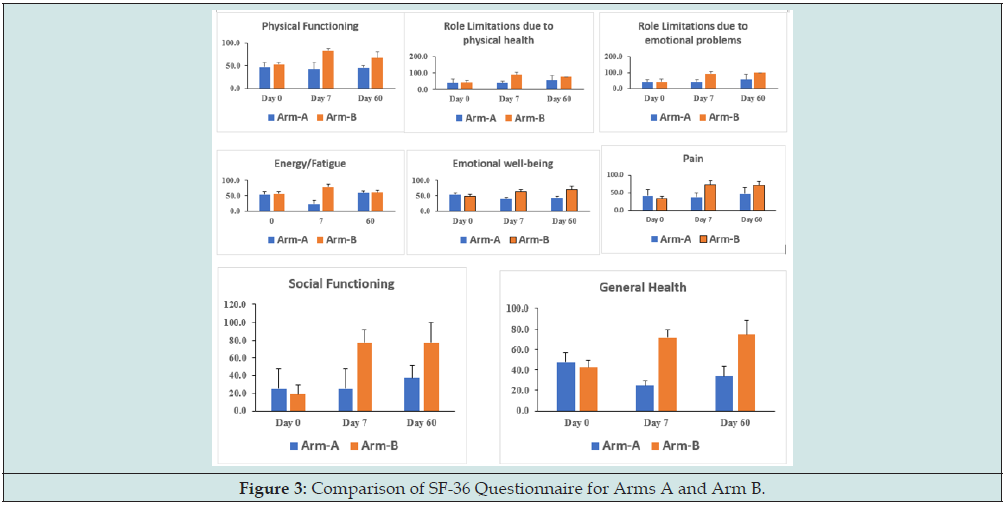
Quality of life
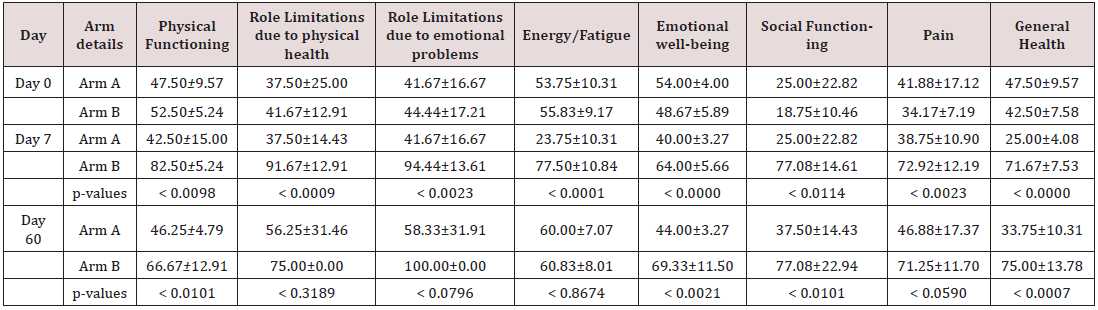
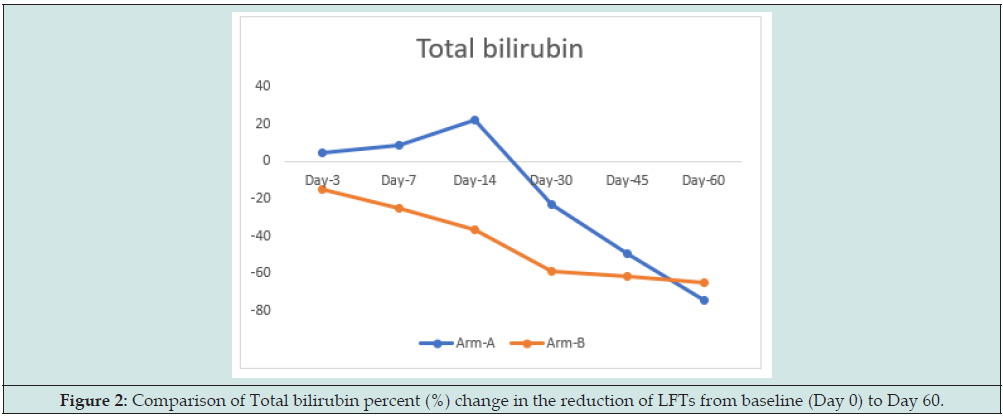
The summary of the SF-36 questionnaire was presented in (Table 6), (Figure 2). Quality of life (SF-36) assessment was performed on day 0 (Baseline), day 7, and day 60 of post-treatment. The results clearly indicated that, a statistically significant improvement was observed in the quality-of-life parameters in arms B patients (with Ursodiol injection + SOC+ST) from baseline (Day 0) to day 7.
Arm A: SOC+ST; Arm B: Ursodiol injection + SOC+ST.
Figure 2: Comparison of Total bilirubin percent (%) change in the reduction of LFTs from baseline (Day 0) to Day 60.
Adverse events
There were no infusion-related adverse events (like irritation, edema, cellulitis, and redness) observed during the study period. Mild adverse events like fever (01- in Arm A and 01 in Arm-B) and headache (01- in Arm A and 01 in Arm-B) were observed.
Discussion
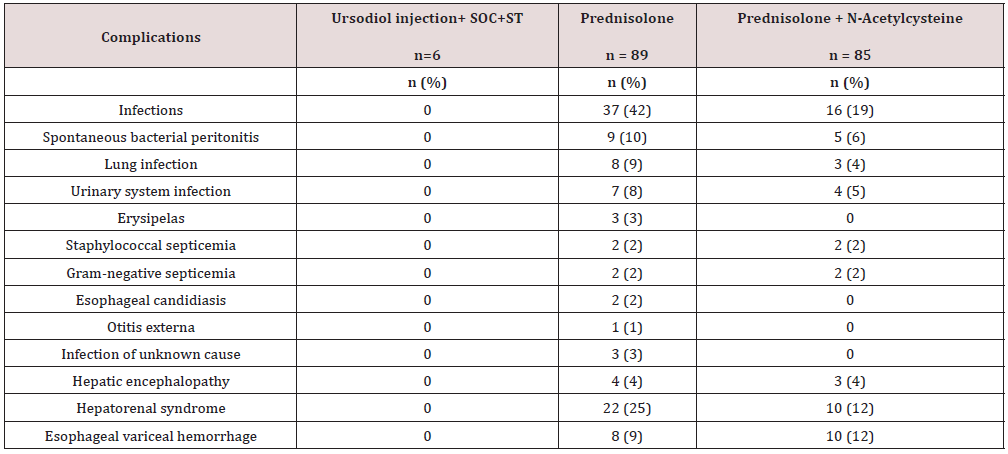
Ursodiol has unique and multiple liver-protective properties like alteration of bile acid pool, anti-inflammatory, and antiapoptotic properties. Based on the pathophysiology of Acholic hepatitis; the need to regulate inflammation, cell damage, and fast reduction of elevated liver enzymes and total bilirubin (TB). Higher levels of total bilirubin are cytotoxic to hepatocytes which lead to hepatotoxicity and also encephalopathy. Based on Ursodiol properties, we have investigated the safety and efficacy of Ursodiol injection with a combination of a standard of care (SOC). The proposed new formulation of Ursodiol injection is an alternative to the oral dosage form with an aim to enhance bioavailability, liver distribution, and quick reduction of elevated liver enzymes in severe acholic hepatitis. In alcoholic hepatitis clinical trials Nguyen-Khac et al., reported mortality rate significantly higher in the prednisolone group (38%) than in the prednisolone–Nacetylcysteine group (27%) at 6 months. Mortality was significantly lower in the prednisolone–N-acetylcysteine group (8%) at 1 month when compared to the prednisolone group (24%) [22]. Another study reported mortality was significantly higher among patients receiving prednisolone (35.29%) as compared to 14.71% among those receiving pentoxifylline [23]. De BK et al., also reported the cause of a higher % of deaths in the prednisolone group is due to hepatorenal syndrome (50%), gastrointestinal bleeding (16.67%), infection (Sepsis) (16.67%), and hepatic encephalopathy (8.33%) and other reasons (8.33%).
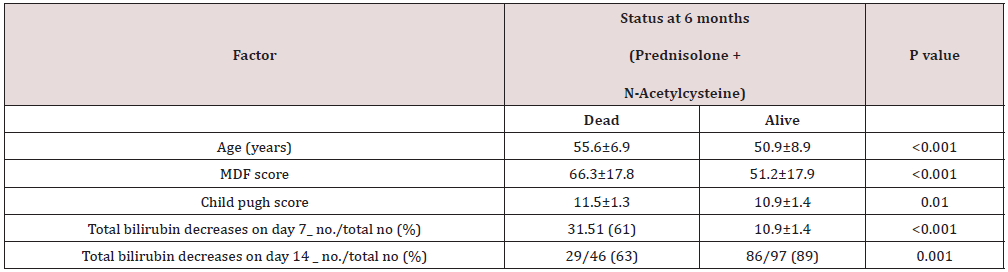
A multicentered, randomized clinical study reported treatment-related complications with prednisolone (P) alone and prednisolone + N-Acetylcysteine (NA). Complications like infection (P=42%; NA=19%), hepatic encephalopathy (P=4%; NA=3%), hepatorenal syndrome (P=25%; NA= 12%) and esophageal variceal hemorrhage (P=9%; NA= 12%) were observed. In our study, there were no treatment-related complications observed during the study period except mild & general adverse events like fever (01- in Arm A and 01 in Arm-B) ad headache (01- in Arm A and 01 in Arm-B) reported during the study period in both arms. A STOPAH (Steroids or Pentoxifylline for Alcoholic Hepatitis) clinical trial, a multicenter study in which more than 1000 patients with severe alcoholic hepatitis (SAH) concluded that, pentoxifylline did not improve survival in patients with alcoholic hepatitis. Prednisolone was associated with a reduction in 28-day mortality that did not reach significance and with no improvement in outcomes at 90 days or 1 year [24] and the number of randomized clinical trials also concluded that current therapies (i.e., corticosteroids and pentoxifylline etc.) fail in many patients [25-27]. Nguyen-Khac et al., reported factors associated with mortality at 6 months (Tables 7 & 8) in dead and alive patients.
In patients who are alive showed statistically significant lower values of total bilirubin decreased on day 7 and day 14. A prospective validation of the finding that a decrease in the bilirubin level after 7 days of treatment is associated with a favorable prognosis and also decrease of bilirubin on day 14 is associated with increased survival. In our study, we observed a marked reduction of elevated LFTs with the Ursodiol injection + SOC arm when compared to only SOC arm. A significant (p<0.0382; P<0.016) reduction of major toxic bile acid total bilirubin on day 7 and 14 was observed and also significant (p<0.0569) improvement in total protein levels were observed in Ursodiol injection + SOC Arm on day 60. Our study observed that, lower mortality (14.28%) rate in Ursodiol injection + SOC arm B when compared to the SOC arm A (20%). Our results were correlated with published literature on lower bilirubin levels on 7 and 14 associated with a favorable prognosis and increased survival. The quick reduction of toxic bile levels with parenteral administration of Ursodiol might be beneficial for severe alcoholic hepatitis patients in critical conditions. However, upon treatment with Ursodiol injection some liver parameters (AST, ALT ALP etc.) were numerically decreased, but not statistically significant, this might be due to a very small sample size.
There was a gradual reduction of MDF & MELD scores of patients in both treatment arms from day 0 to day 60 and no statistical significance was observed when compared to the two arms but % change from baseline was improved numerically in Arm B from day 14 to day 60 when compared to Arm A. There was no difference in MDF values between the pentoxifylline group and placebo group of a double-blind, placebo-controlled trial in AH patients with 4 weeks of treatment duration [28] with low sample size. A published and American Association for the Study of Liver Diseases (AALSD) guidance suggesting both MELD and MDF is useful for predicting short-term mortality in patients with Alcoholic hepatitis [3-4].
We have observed, patients with arm B (Ursodiol injection+ SOC+ ST) presented significant improvement in quality of life (SF-36) when compared to SOC arm A (Figure 3). Rapid improvement was observed in energy, physical health, physical functioning, sleep, appetite emotional wellbeing and abdominal pain at day 7 of UDCA + SOC arm.
Conclusion
In alcoholic hepatitis, the exposure of hepatocytes to high concentrations of toxic bile acids is responsible for liver injury and inflammation. Based on observed results we have concluded that the beneficial effects of intravenous administration of Ursodiol to patients with alcoholic liver diseases are to be considered in terms of quick reduction of highly elevated liver enzymes, which leads to improvement in quality of life in patients who were in critical condition. Research studies also concluded that decreased total bilirubin on day 7 and 14 is associated with lower mortality and thereby clinical improvement. Parenteral infusion of Ursodiol in severe alcoholic hepatitis at intensive care unit (ICU), resulted in marked improvement in elevated liver function tests and improvement in quality of life, statistically significant reduction in serum bilirubin and improvement in serum albumin and protein levels which are continued at 60 days also with no side effects. High dose Ursodiol injection might be a promising, safe, and effective therapy for management of severe alcoholic hepatitis. Severe alcoholic hepatitis is associated with a high mortality rate due to comorbid conditions such as severe kidney injury, uncontrolled sepsis, and active gastrointestinal bleeding, these conditions might hamper study outcome if not properly standardized during inclusion and exclusion in the study. So, in order to establish adequate efficacy and safety of Ursodiol injection, a well-organized, randomized, controlled large clinical trials need to be conducted in, with repeated treatment cycles in severe alcoholic hepatitis patients.
Acknowledgments
The author is thankful to Shilpa Medicare Limited, Hyderabad, India for providing the test samples and funding support for this research work.
References
- Liangpunsakul S (2011) Clinical characteristics and mortality of hospitalized alcoholic hepatitis patients in the United States. J Clin Gastroenterol 45(8): 714-719.
- Dominguez M, Rincón D, Abraldes JG, Miquel R, Colmenero J, et al. (2008) A new scoring system for prognostic stratification of patients with alcoholic hepatitis. Am J Gastroenterol 103(11): 2747-2756.
- Dunn W, Jamil LH, Brown LS, Wiesner RH, Kim WR, et al. (2005) MELD accurately predicts mortality in patients with alcoholic hepatitis. Hepatology 41(2): 353-358.
- Crabb DW, Im GY, Szabo G, Mellinger JL, Lucey MR (2020) Diagnosis and treatment of alcohol‐associated liver diseases: 2019 practice guidance from the American Association for the Study of Liver Diseases. Hepatology 71(1): 306-333.
- Ko WK, Lee SH, Kim SJ, Jo MJ, Kumar H, et al. (2017) Anti-inflammatory effects of ursodeoxycholic acid by lipopolysaccharide-stimulated inflammatory responses in RAW 264.7 macrophages. PLoS One 12(6): e0180673.
- Morgan TR (2007) Management of Alcoholic Hepatitis. Gastroenterol Hepatol 3(2): 97-99.
- Tang L, Zhang M, Li X, Zhang L (2019) Glucuronidated bilirubin: Significantly increased in hepatic encephalopathy. Prog Mol Biol Transl Sci 162: 363-376.
- Allen K, Jaeschke H, Copple BL (2011) Bile acids induce inflammatory genes in hepatocytes: a novel mechanism of inflammation during obstructive cholestasis. Am J Pathol 178(1): 175-186.
- Szabo G, Kamath PS, Shah VH, Thursz M, Mathurin P, et al. (2019) Alcohol-Related Liver Disease: Areas of Consensus, Unmet Needs and Opportunities for Further Study. Hepatology 69(5): 2271-2283.
- Tornai D, Szabo G (2020) Emerging medical therapies for severe alcoholic hepatitis. Clin Mol Hepatol 26(4): 686-696.
- Louvet A, Wartel F, Castel H, Dharancy S, Hollebecque A, et al. (2009) Infection in patients with severe alcoholic hepatitis treated with steroids: early response to therapy is the key factor. Gastroenterology 137(2): 541-548.
- Neuman MG, Cameron RG, Shear NH, Bellentani S, Tiribelli C (1995) Effect of tauroursodeoxycholic and ursodeoxycholic acid on ethanol-induced cell injuries in the human Hep G2 cell line. Gastroenterology 109(2): 555-563.
- Tabouy L, Zamora AJ, Oliva L, Montet AM, Beaugé F, et al. (1998) Ursodeoxycholate protects against ethanol-induced liver mitochondrial injury. Life Sci 63(25): 2259-2270.
- Tint GS, Salen G, Colalillo A, Graber D, Verga D, et al. (1982) Ursodeoxycholic acid: a safe and effective agent for dissolving cholesterol gallstones. Ann Internal Med 97(3): 351-356.
- Lazaridis KN, Gores GJ, Lindor KD (2001) Ursodeoxycholic acid 'mechanisms of action and clinical use in hepatobiliary disorders'. J Hepatol 35(1): 134-146.
- Paumgartner G, Beuers U (2002) Ursodeoxycholic acid in cholestatic liver disease: mechanisms of action and therapeutic use revisited. Hepatology 36(3): 525-531.
- Plevris JN, Hayes PC, Bouchier IAD, Plevris J (1991) Ursodeoxycholic acid in the treatment of alcoholic liver-disease. Eur J Gastroenterol Hepatol 3(9): 653-656
- Palmieri B, Vadalà M (2020) Safety Pilot Study on Doctors Intravenous UDCA Self-Infusion among “The Doctor-Care-Yourself” Network. Int J Innov Sci Technol 5(3): 1020-1023.
- Palmieri B, Vadalà M (2020) Intravenous UDCA Administration during Cancer Chemotherapy, Liver Failure and Oral Route not Available. Int J Innov Sci Technol 5(3): 941-946.
- Palmieri B, Vadalà M (2020) Nash Clinical Study. Int J Innov Sci Technol 5(3): 1200-1208.
- Palmieri B, Palmieri L, Vadalà M (2020) Intravenous bile salts indication in gallbladder transparent stones and colicky pains. Int J Innov Sci Technol 5(3): 1162-1166.
- Nguyen-Khac E, Thevenot T, Piquet MA, Benferhat S, Goria O, et al. (2011) Glucocorticoids plus N-acetylcysteine in severe alcoholic hepatitis. New England Journal of Medicine 365(19): 1781-1789.
- De BK, Gangopadhyay S, Dutta D, Baks, SD, Pani A, et al. (2009) Pentoxifylline versus prednisolone for severe alcoholic hepatitis: a randomized controlled trial. World J Gastroenterol 15(13): 1613-1619.
- Thursz MR, Richardson P, Allison M, Austin A, Bowers M, et al. (2015) Prednisolone or pentoxifylline for alcoholic hepatitis. New England Journal of Medicine 372(17): 1619-1628.
- Louvet A, Diaz E, Dharancy S, Coevoet H, Texier F, et al. (2008) Early switch to pentoxifylline in patients with severe alcoholic hepatitis is inefficient in non-responders to corticosteroids. J Hepatol 48(3): 465-470.
- Sidhu SS, Goyal O, Singla P, Gupta D, Sood A, et al. (2012) Corticosteroid plus pentoxifylline is not better than corticosteroid alone for improving survival in severe alcoholic hepatitis (COPE trial). Dig Dis Sci 57(6): 1664-1671.
- European Association for the Study of Liver (2012) EASL clinical practical guidelines: management of alcoholic liver disease. J Hepatol 57(2): 399-420.
- Akriviadis E, Botla R, Briggs W, Han S, Reynolds T, et al. (2000) Pentoxifylline improves short-term survival in severe acute alcoholic hepatitis: a double-blind, placebo-controlled trial. Gastroenterology 119(6): 1637-1648.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...