Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2638-6062
Review Article(ISSN: 2638-6062) 
Kabuki Syndrome: Current Understanding of Symptoms and Treatment Strategies Volume 5 - Issue 1
Yalda Zhoulideh*
- Graduated with master’s degree in Genetics at Islamic Azad University of Tabriz, Genetic Researcher, Tehran, Iran
Received: May 29, 2023; Published:June 07, 2023
*Corresponding author: Yalda Zhoulideh, Graduated with master’s degree in Genetics at Islamic Azad University of Tabriz, Genetic Researcher, Tehran, Iran
DOI: 10.32474/PRJFGS.2023.05.000202
Abstract
Kabuki syndrome can be mentioned as one of the most severe and, at the same time, rare genetic abnormalities. The inheritance pattern of this disorder can be an autosomal dominant or X-linked pattern. In this disease, KMT2D AND KDM6A genes are disrupted, which encode histone methyltransferase and histone demethylase, respectively. The severity of the disease and associated signs and symptoms can vary widely but may include distinct facial features, developmental delay, intellectual disability, and limb deformities. Kabuki syndrome treatment may vary based on the specific symptoms that appear in each individual. This review will examine the genes involved in this disease, phenotype, clinical manifestations, ways of diagnosis, and treatment of this disease.
Keywords: Genetic disorder; kabuki; KDM6A; KMT2D; niikawa-kuroki
Introduction
Kabuki syndrome (KS: OMIM 147920) is a rare genetic disorder that has two inheritance patterns: X-linked (associated with the KDM6A gene) and autosomal dominant (associated with the KMT2D gene). Other names for this disease include Kabuki-makeup syndrome (KMS) and Niikawa-Kuroki syndrome. The reason for the name of this disease is the appearance of the face of affected people with the type of make-up of the actors of the traditional Kabuki show in Japan [1-6]. For the first time, this disease was defined and identified in 1981 by two groups of researchers, led by Dr. Norio Nikawa, a Japanese geneticist, and Yoshikazu Kuroki, a Japanese physician. Kabuki syndrome affects women and men in the same proportion and does not increase among certain ethnic groups. Also, its frequency can be considered as one in 32,000 live births. However, since this disease includes a wide range of mild to severe symptoms, some cases of the mild disease may never be detected, and as a result, the number of cases is higher than the estimated amount [7-10].
This heterogeneous disease causes very different symptoms in affected people and varies from person to person. The severity of the disease and associated signs and symptoms can vary widely; still, they may include distinctive facial features, Permanent pads between the fingers, hypermobility of the joints, sensorineural deafness, diaphragmatic hernia, arched eyebrows, and elongated eyelids [11-14]. This disease overlaps with some diseases, such as CHARGE syndrome, Turner syndrome, Van der Woude syndrome, 22q11 deletion syndrome, and other disorders. Therefore, the accurate diagnosis of this disease is both a challenging and vital task because the subsequent treatments and measures to improve the physical condition and the spirit of the patient and his family depend on the correct diagnosis. Since this disease can endanger the affected person’s life in severe cases, it is imperative to start treatment early. Also, genetic counseling for family members is one of the most fundamental approaches to prevent more cases. Considering the importance of genetic disorders and their spread in some societies, we reviewed Kabuki syndrome, its disease mechanism, diagnosis, and other related issues.
Role of KMT2D
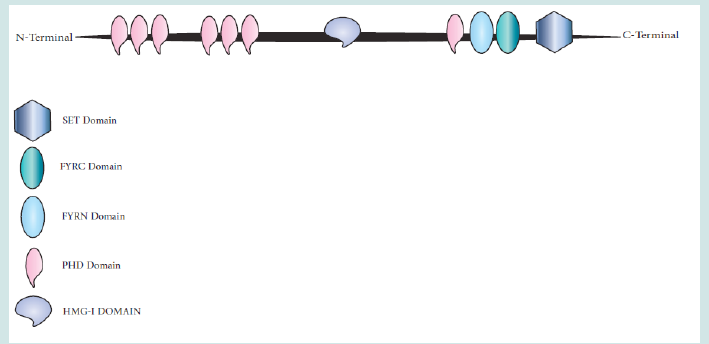
Set1-like H3K4 HMTs consists of six subsets including KMT2A, KMT2B, KMT2C, KMT2F, KMT2G, and KMT2D. KMT2D (Histonelysine N-methyltransferase 2D) is a critical histone H3 lysine 4 mono-methyltransferase in mammals [15,16]. The other name of this gene is MLL2, which plays an essential role in the embryo’s development and suppressing tumors. A loss of function mutation in the KMT2D gene can cause Kabuki syndrome. Other diseases that can occur due to disruption in this gene include congenital heart disease and some types of cancer, such as pheochromocytoma, non- Hodgkin lymphomas, pancreatic cancer, and prostate cancer [17- 26]. An enzyme called methyltransferase, produced by the KMT2D gene changes histones by adding a methyl group. KMT2D, which has been altered, is inoperable. As a result, histone lysine-specific methylation is disturbed [27-33]. This gene is located in the long arm of chromosome 12. KMT2D gene produces a protein with the same name, which has different domains. In the C-Terminal, there is a SET domain that regulates methyltransferase activity, then FYRC and FYRN domains, and then PHD and HMG-I domains. On the N-Terminal side, there are six PHD domains [34-48]. The structure of the KMTD2 protein is shown in Figure 1.
Role of KDM6A
KDM6A (Lysine-specific demethylase 6A) belongs to the 2-oxoglutarate (2OG)-dependent dioxygenase family, which is a protein-coding gene whose protein product plays a role in normal development and tumor suppression. Another name for this gene is UTX, located in Xp11.3 and causes random inactivation of one of the X chromosomes [49,50]. Its mutation can cause various diseases such as Kabuki syndrome and cancers such as multiple myeloma, breast, and colon. A methyl group from lysine is removed by the KDM6A gene product, an enzyme that demethylates histones. KDM6A with a mutation cannot function, and developmental abnormalities result from the disruption of histone lysine-specific demethylation. The protein product of the KDM6A gene has six domains on the N-Terminal side called tetratricopeptide repeat (TPR) and one on the C-Terminal side called Jumonji C (JmjC) that play a role in regulating demethylation [51-54]. The structure of the KDM6A protein is shown in Figure 2.
Clinical Features
The symptoms of Kabuki syndrome overlap with various diseases such as CHARGE syndrome, Turner syndrome, Van der Woude syndrome, 22q11 deletion syndrome, and other disorders [55-65]. However, the clinical symptoms of this disease can be generally categorized as follows:
Facial Features
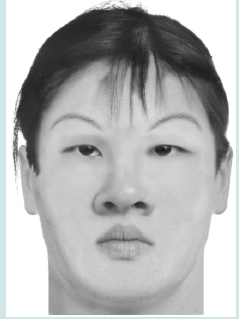
About one-third of patients have also been reported to have mid-face hypoplasia, hypodontia, cleft palate, and a trapezoidal philtrum. Over 40% of people with KS have dental anomalies. Other signs and symptoms that can be seen in almost all affected individuals include persistently drooping eyelids, arched eyebrows, a sunken tip to the nose, a long eyelid furrow, long eyelashes, a small chin, and a wide nose [66-71]. Figure 3 shows a picture of a person with Kabuki syndrome.
Figure 3: Image related to Kabuki syndrome facial features. Note the wide nose with a flat tip, downward cleft eyelids, and arched eyebrows. (Note: out of respect for patients’ rights, this image is not related to a natural person and is designed by the software).
Features of hearing
15 to 25 percent of patients have hearing and ear issues. Some of the most typical otolaryngologic findings in KS include dysmorphic pinnae, large, cup-shaped ears, otitis media, and hearing loss. It seems that otitis media is expected [72,73].
Characteristics of the Reproductive and Urinary System
Over 30% of KS patients have some form of renal abnormalities, such as hydronephrosis, renal hypoplasia or dysplasia, or kidney fusion defects, such as the horseshoe kidney, like CHARGE syndrome. Patients with kidneys that were noticeably dysplastic required transplantation. About 31% of cases have been reported to have hypospadias, small penis, and cryptorchidism [74-78].
Developmental Delay
Patients with KS appear to experience specific delays in language and speech maturation, and these delays or absences may be present in KS patients. During the first year of life, most patients experience failure to thrive and postnatal growth retardation. Although they eat a healthy diet, these individuals may not grow to their full height and have excessive obesity [79-85]
Eye Features
About 35 to 60 percent of patients with KS have ophthalmologic abnormalities. Strabismus and blue sclerae are among the most typical findings. Refractive errors, cataracts, delayed visual maturation, optic nerve hypoplasia, ptosis, nystagmus, palsy of the third cranial nerve, and Marcus Gunn’s phenomenon are less frequent findings [86-88].
Heart Problems
The prevalence of congenital heart defects in this population has been estimated to be between 40 and 50 percent, and many different congenital heart defects have been described in patients with KS. Juxta ductal coarctation of the aorta, a relatively uncommon heart defect, appears to be the most frequent finding, followed by VSD and ASD. Males seem to experience coarctation of the aorta in KS much more frequently than females [89-91].
Digestive Problems
While malrotation of the intestines has been documented in a few patients with KS, gastrointestinal abnormalities are uncommon, occurring in only about 7% of cases. Instead, most patients with KS have abnormalities of the anus or rectum, such as anal atresia, anovestibular fistulas, or anteriorly placed anuses. Additionally, biliary atresia and neonatal sclerosing cholangitis that required a liver transplant have been documented [92-95].
Abnormalities related to the Brain
Autism or behaviors resembling autism have been identified in several patients. 90% of affected patients exhibit mild to severe mental retardation, and in most cases, their IQ level is below 85. Only 10% of affected patients have average intelligence. Seizures occur in 15 to 40% of KS patients. A large arachnoid cyst, polymicrogyria, non-specific cerebral atrophy or enlarged ventricles, and significant brain structural abnormalities have all been seen in MRI studies of KS patients [96-100].
Skeletal Abnormalities
In KS, skeletal abnormalities are frequent. Other abnormalities include the sagittal cleft, short fifth digits with short fifth metacarpals, cone-shaped proximal second through fifth phalange epiphyses, coronal synostosis, dislocation of the hip joints, and various vertebral anomalies [101-107].
Ways to diagnose Kabuki
For prenatal diagnosis of patients with KS, the ultrasound method can detect some abnormalities, such as the lack of proper development of fingers or toes, twisting of the intestine, facial abnormalities, and other features. Individuals with specific morphological features associated with KS may be candidates for genetic testing for KMT2D and KDM6A genes at birth [108]. In addition, sometimes, the symptoms of the disease are minimal, and NGS can be done to be sure. Currently, there are no consensus diagnostic criteria for KS; however, five principal components can be used for the early diagnosis of KS, including skeletal abnormalities, specific facial features, mental retardation, developmental delay, and interdigital pads.
Therapeutic Approaches
Kabuki disease can present various phenotypes and complications depending on the mutated gene and the general conditions. The proposed treatment depends on the phenotype and the affected organ because not all individuals with this disease exhibit the same symptoms. In addition, there is no universally effective cure for this illness [109,110]. All available treatments have aimed to improve patients’ quality of life because there is currently no proven standard treatment for this illness. Genetic counseling is one of the most crucial recommendations to Kabuki patients and their family because it can aid in more effective and better disease management. Since this patient exhibits symptoms almost from birth, medical monitoring is required to prevent severe complications of the disease because, occasionally, some of the complexities of this condition can endanger a person’s life. For kids with Kabuki, pediatricians are frequently the first point of contact for medical attention. The psychology of illness should be one of the key topics covered during treatment. People close to the affected person or in charge of their care may experience emotional stress. For these people to live peaceful daily lives with their ill loved ones, psychological counseling is helpful. Cognitive difficulties can be one of the signs of Kabuki. Although not all affected patients have this disease, it is better to educate those who do so that they can safeguard their health from the disease’s emergency.
Conclusion
KMT2D and KDM6A genes contribute to the complex genetic condition known as Kabuki. It appears that more tests should be considered to find additional causes of this disease and that genome sequencing can be helpful. However, some genes effective in causing the disease to remain unknown. Furthermore, there is no known cure for this illness, and all current therapies mainly serve as supportive care. CRISPR/Cas9 and gene therapy research may someday result in the ability to treat this genetic condition.
Conflict of Interest
The author declares that the research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Paderova J, Drabova J, Holubova A, Vlckova M, Havlovicova M, et al. (2018) Under the mask of Kabuki syndrome: Elucidation of genetic-and phenotypic heterogeneity in patients with Kabuki-like phenotype. Eur J Med Genet 61(6): 315-321.
- Bokinni Y (2012) Kabuki syndrome revisited. J Hum Genet 57(4): 223-227.
- Subbarayan A, Hussain K (2014) Hypoglycemia in Kabuki syndrome. Am J Med Genet A 164A(2): 467-471.
- Bernier FE, Schreiber A, Coulombe J, Hatami A, Marcoux D (2017) Pilomatricoma Associated with Kabuki Syndrome. Pediatr Dermatol 34(1): e26-e27.
- Kurosawa K, Kawame H, Ochiai Y, Nakashima M, Tohma T, et al. (2002) Patellar dislocation in Kabuki syndrome. Am J Med Genet 108(2): 160-163.
- Hamahata A, Kamei W, Ishikawa M, Konoeda H, Yamaki T, et al. (2013) Multiple Pilomatricomas in Kabuki Syndrome. Pediatr Dermatol 30(2): 253-255.
- Vaux KK, Hudgins L, Bird LM, Roeder E, Curry CJR, et al. (2005) Neonatal phenotype in Kabuki syndrome. Am J Med Genet A 132A(3): 244-247.
- Ming JE, Russell KL, McDonald-McGinn DM, Zackai EH (2005) Autoimmune disorders in Kabuki syndrome. Am J Med Genet A 132A(3): 260-262.
- Defloor T, van Borsel J, Schrander Stumpel CTRM, Curfs LMG (2005) Expressive language in children with Kabuki syndrome. Am J Med Genet A 132A(3): 256-259.
- Ciprero KL, Clayton-Smith J, Donnai D, Zimmerman RA, Zackai EH, et al. (2005) Symptomatic Chiari I malformation in Kabuki syndrome. Am J Med Genet A 132A(3): 273-275.
- Scherer S, Theile U, Beyer V, Ferrari R, Kreck C, et al. (2003) Patient with Kabuki syndrome and acute leukemia. Am J Med Genet A 122A(1): 76-79.
- Tumino M, Licciardello M, Sorge G, Cutrupi MC, di Benedetto F, et al. (2010) Kabuki syndrome and cancer in two patients. Am J Med Genet A 152A(6): 1536-1539.
- Kawame H, Hannibal MC, Hudgins L, Pagon RA (1999) Phenotypic spectrum and management issues in Kabuki syndrome. J Pediatr 134(4): 480-485.
- Li Y, Bögershausen N, Alanay Y, Simsek Kiper PÖ, Plume N, et al. (2011) A mutation screen in patients with Kabuki syndrome. Hum Genet 130(6): 715-724.
- Schwenty-Lara J, Nürnberger A, Borchers A (2019) Loss of function of Kmt2d, a gene mutated in Kabuki syndrome, affects heart development in Xenopus laevis. Dev Dyn 248(6): 465-476.
- Schwenty-Lara J, Nehl D, Borchers A (2020) The histone methyltransferase KMT2D, mutated in Kabuki syndrome patients, is required for neural crest cell formation and migration. Hum Mol Genet 29(2): 305-319.
- Paulussen ADC, Stegmann APA, Blok MJ, Tserpelis D, Posma-Velter C, et al. (2011) MLL2 mutation spectrum in 45 patients with Kabuki syndrome. Hum Mutat 32(2): E2018-2025.
- Banka S, Howard E, Bunstone S, Chandler KE, Kerr B, et al. (2013) MLL2 mosaic mutations and intragenic deletion–duplications in patients with Kabuki syndrome. Clin Genet 83(5): 467-471.
- Micale L, Augello B, Fusco C, Selicorni A, Loviglio MN, et al. (2011) Mutation spectrum of MLL2 in a cohort of kabuki syndrome patients. Orphanet J Rare Dis 6: 38.
- Hannibal MC, Buckingham KJ, Ng SB, Ming JE, Beck AE, et al. (2011) Spectrum of MLL2 (ALR) mutations in 110 cases of Kabuki syndrome. Am J Med Genet A 155A(7): 1511-1516.
- Ng SB, Bigham AW, Buckingham KJ, Hannibal MC, McMillin MJ, et al. (2010) Exome sequencing identifies MLL2 mutations as a cause of Kabuki syndrome. Nat Genet 42(9): 790-793.
- Kokitsu-Nakata NM, Petrin AL, Heard JP, Vendramini-Pittoli S, Henkle LE, et al. (2012) Analysis of MLL2 gene in the first Brazilian family with Kabuki syndrome. Am J Med Genet A 158A(8): 2003-2008.
- Makrythanasis P, van Bon BW, Steehouwer M, Rodríguez-Santiago B, Simpson M, et al. (2013) MLL2 mutation detection in 86 patients with Kabuki syndrome: a genotype–phenotype study. Clin Genet 84(6): 539-545.
- Zarate YA, Zhan H, Jones JR (2012) Infrequent Manifestations of Kabuki Syndrome in a Patient with Novel MLL2 Mutation. Mol Syndromol 3(4): 180-184.
- Cappuccio G, Rossi A, Fontana P, Acampora E, Avolio V, et al. (2014) Bronchial isomerism in a Kabuki syndrome patient with a novel mutation in MLL2 gene. BMC Med Genet 15(1): 1-5.
- Banka S, Veeramachaneni R, Reardon W, Howard E, Bunstone S, et al. (2012) How genetically heterogeneous is Kabuki syndrome?: MLL2 testing in 116 patients, review and analyses of mutation and phenotypic spectrum. Eur J Hum Genet 20(4): 381-388.
- Stadelmaier RT, Kenna MA, Barrett D, Mullen TE, Bodamer O, et al. (2021) Neuroimaging in Kabuki syndrome and another KMT2D-related disorder. Am J Med Genet A 185(12): 3770-3783.
- Baldridge D, Spillmann RC, Wegner DJ, Wambach JA, White FV, et al. (2020) Phenotypic expansion of KMT2D-related disorder: Beyond Kabuki syndrome. Am J Med Genet A 182(5): 1053-1065.
- Cocciadiferro D, Augello B, de Nittis P, Zhang J, Mandriani B, et al. (2018) Dissecting KMT2D missense mutations in Kabuki syndrome patients. Hum Mol Genet 27(21): 3651-3668.
- Schott DA, Blok MJ, Gerver WJM, Devriendt K, Zimmermann LJI, et al. (2016) Growth pattern in Kabuki syndrome with a KMT2D mutation. Am J Med Genet A 170(12): 3172-3179.
- Lin JL, Lee WI, Huang JL, Chen PKT, Chan KC, et al. (2015) Immunologic assessment and KMT2D mutation detection in Kabuki syndrome. Clin Genet 88(3): 255-260.
- Lu J, Mo G, Ling Y, Ji L (2016) A novel KMT2D mutation resulting in Kabuki syndrome: A case report. Mol Med 14(4): 3641-3645.
- de Billy E, Strocchio L, Cacchione A, Agolini E, Gnazzo M, et al. (2019) Burkitt lymphoma in a patient with Kabuki syndrome carrying a novel KMT2D mutation. Am J Med Genet A 179(1): 113-117.
- Luperchio TR, Applegate CD, Bodamer O, Bjornsson HT (2020) Haploinsufficiency of KMT2D is sufficient to cause Kabuki syndrome and is compatible with life. Mol Genet Genomic Med 8(2): e1072.
- Lehman N, Mazery AC, Visier A, Baumann C, Lachesnais D, et al. (2017) Molecular, clinical and neuropsychological study in 31 patients with Kabuki syndrome and KMT2D mutations. Clin Genet 92(3): 298-305.
- Cheon CK, Sohn YB, Ko JM, Lee YJ, Song JS, et al. (2014) Identification of KMT2D and KDM6A mutations by exome sequencing in Korean patients with Kabuki syndrome. J Hum Genet 59(6): 321-325.
- Yamamoto PK, de Souza TA, Antiorio ATFB, Zanatto DA, Garcia-Gomes M de SA, et al. (2019) Genetic and behavioral characterization of a Kmt2d mouse mutant, a new model for Kabuki Syndrome. Genes Brain Behav 18(8): e12568.
- Faundes V, Malone G, Newman WG, Banka S (2018) A comparative analysis of KMT2D missense variants in Kabuki syndrome, cancers and the general population. J Hum Genet 64(2): 161-170.
- Cuvertino S, Hartill V, Colyer A, Garner T, Nair N, et al. (2020) A restricted spectrum of missense KMT2D variants cause a multiple malformations disorder distinct from Kabuki syndrome. Genet Med 22(5): 867-877.
- Piro E, Schierz IAM, Antona V, Pappalardo MP, Giuffrè M, et al. (2020) Neonatal hyperinsulinemic hypoglycemia: Case report of kabuki syndrome due to a novel KMT2D splicing-site mutation. Ital J Pediatr 46(1): 136.
- van Laarhoven PM, Neitzel LR, Quintana AM, Geiger EA, Zackai EH, et al. (2015) Kabuki syndrome genes KMT2D and KDM6A: functional analyses demonstrate critical roles in craniofacial, heart and brain development. Hum Mol Genet 24(15): 4443-4453.
- Moon JE, Lee SJ, Ko CW (2018) A de novo KMT2D mutation in a girl with Kabuki syndrome associated with endocrine symptoms: A case report. BMC Med Genet 19(1): 102.
- Yap CS, Jamuar SS, Lai AHM, Tan ES, Ng I, et al. (2020) Identification of KMT2D and KDM6A variants by targeted sequencing from patients with Kabuki syndrome and other congenital disorders. Gene 731: 144360.
- Xin C, Wang C, Wang Y, Zhao J, Wang L, et al. (2018) Identification of novel KMT2D mutations in two Chinese children with Kabuki syndrome: A case report and systematic literature review. BMC Med Genet 19(1): 31.
- Bögershausen N, Gatinois V, Riehmer V, Kayserili H, Becker J, et al. (2016) Mutation Update for Kabuki Syndrome Genes KMT2D and KDM6A and Further Delineation of X-Linked Kabuki Syndrome Subtype 2. Hum Mutat 37(9): 847-864.
- Murakami H, Tsurusaki Y, Enomoto K, Kuroda Y, Yokoi T, et al. (2020) Update of the genotype and phenotype of KMT2D and KDM6A by genetic screening of 100 patients with clinically suspected Kabuki syndrome. Am J Med Genet A 182(10): 2333-2344.
- Porntaveetus T, Abid MF, Theerapanon T, Srichomthong C, Ohazama A, et al. (2018) Expanding the Oro-Dental and Mutational Spectra of Kabuki Syndrome and Expression of KMT2D and KDM6A in Human Tooth Germs. Int J Biol Sci 14(4): 381-389.
- Topa A, Samuelsson L, Lovmar L, Stenman G, Kölby L (2017) On the significance of craniosynostosis in a case of Kabuki syndrome with a concomitant KMT2D mutation and 3.2 Mbp de novo 10q22.3q23.1 deletion. Am J Med Genet A 173(8): 2219-2225.
- Guo Z, Liu F, Li HJ (2018) Novel KDM6A splice-site mutation in kabuki syndrome with congenital hydrocephalus: A case report. BMC Med Genet 19(1): 206.
- Banka S, Lederer D, Benoit V, Jenkins E, Howard E, et al. (2015) Novel KDM6A (UTX) mutations and a clinical and molecular review of the X-linked Kabuki syndrome (KS2). Clin Genet 87(3): 252-258.
- Miyake N, Koshimizu E, Okamoto N, Mizuno S, Ogata T, et al. (2013) MLL2 and KDM6A mutations in patients with Kabuki syndrome. Am J Med Genet A 161(9): 2234-2243.
- Lederer D, Shears D, Benoit V, Verellen-Dumoulin C, Maystadt I (2014) A three generation X-linked family with Kabuki syndrome phenotype and a frameshift mutation in KDM6A. Am J Med Genet A 164A(5): 1289-1292.
- Priolo M, Micale L, Augello B, Fusco C, Zucchetti F, et al. (2012) Absence of deletion and duplication of MLL2 and KDM6A genes in a large cohort of patients with Kabuki syndrome. Mol Genet Metab 107(3): 627-629.
- Faundes V, Goh S, Akilapa R, Bezuidenhout H, Bjornsson HT, et al. (2021) Clinical delineation, sex differences, and genotype–phenotype correlation in pathogenic KDM6A variants causing X-linked Kabuki syndrome type 2. Genet Med 23(7): 1202-1210.
- Cheon CK, Ko JM (2015) Kabuki syndrome: clinical and molecular characteristics. Korean J Pediatr 58(9): 317-324.
- Schrander-Stumpel CTRM, Spruyt L, Curfs LMG, Defloor T, Schrander JJP (2005) Kabuki syndrome: Clinical data in 20 patients, literature review, and further guidelines for preventive management. Am J Med Genet A 132A(3): 234-243.
- di Candia F, Fontana P, Paglia P, Falco M, Rosano C, et al. (2022) Clinical heterogeneity of Kabuki syndrome in a cohort of Italian patients and review of the literature. Eur J Pediatr 181(1): 171-187.
- Aref-Eshghi E, Bourque DK, Kerkhof J, Carere DA, Ainsworth P, et al. (2019) Genome-wide DNA methylation and RNA analyses enable reclassification of two variants of uncertain significance in a patient with clinical Kabuki syndrome. Hum Mutat 40(10): 1684-1689.
- Adam MP, Hudgins L (2005) Kabuki syndrome: a review. Clin Genet 67(3): 209-219.
- Bögershausen N, Wollnik B (2013) Unmasking Kabuki syndrome. Clin Genet 83(3): 201-211.
- Milunsky JM, Huang XL (2003) Unmasking Kabuki syndrome: chromosome 8p22–8p23.1 duplication revealed by comparative genomic hybridization and BAC-FISH. Clin Genet 64(6): 509-516.
- Schulz Y, Freese L, Mänz J, Zoll B, Völter C, et al. (2014) CHARGE and Kabuki syndromes: a phenotypic and molecular link. Hum Mol Genet 23(16): 4396-4405.
- Verhagen JMA, Oostdijk W, Terwisscha van Scheltinga CEJ, Schalij-Delfos NE, van Bever Y (2014) An unusual presentation of Kabuki syndrome: Clinical overlap with CHARGE syndrome. Eur J Med Genet 57(9): 510-512.
- Ming JE, Russell KL, Bason L, McDonald-McGinn DM, Zackai EH (2003) Coloboma and Other Ophthalmologic Anomalies in Kabuki Syndrome: Distinction from Charge Association. Am J Med Genet 123A(3): 249-252.
- Butcher DT, Cytrynbaum C, Turinsky AL, Siu MT, Inbar-Feigenberg M, et al. (2017) CHARGE and Kabuki Syndromes: Gene-Specific DNA Methylation Signatures Identify Epigenetic Mechanisms Linking These Clinically Overlapping Conditions. Am J Hum Genet 100(5): 773-788.
- Petzold D, Kratzsch E, Opitz C, Tinschert S (2003) The Kabuki syndrome: four patients with oral abnormalities. Eur J Orthod 25(1): 13-19.
- Wang YR, Xu NX, Wang J, Wang XM (2019) Kabuki syndrome: review of the clinical features, diagnosis and epigenetic mechanisms. World J Pediatr 15(6): 528-535.
- Yap KL, Johnson AEK, Fischer D, Kandikatla P, Deml J, et al. (2019) Congenital hyperinsulinism as the presenting feature of Kabuki syndrome: clinical and molecular characterization of 10 affected individuals. Genet Med 21(1): 262-265.
- Atar M, Lee W, O'Donnell D (2006) Kabuki syndrome: oral and general features seen in a 2-year-old Chinese boy. Int J Paediatr Dent 16(3): 222-226.
- Tunovic S, Barkovich J, Sherr EH, Slavotinek AM (2014) De novo ANKRD11 and KDM1A gene mutations in a male with features of KBG syndrome and Kabuki syndrome. Am J Med Genet A 164A(7): 1744-1749.
- So PL, Luk HM, Yu KPT, Cheng SSW, Hau EWL, et al. (2021) Clinical and molecular characterization study of Chinese Kabuki syndrome in Hong Kong. Am J Med Genet A 185(3): 675-686.
- Peterson-Falzone SJ, Golabi M, Lalwani AK (1997) Otolaryngologic manifestations of kabuki syndrome. Int J Pediatr Otorhinolaryngol 38(3): 227-236.
- Pepe G, Negri M, Falcioni M, di Lella F, Vincenti V (2020) Bonebridge implantation for mixed hearing loss in a patient with Kabuki syndrome. Acta Biomed 91(3): e2020079.
- Courcet JB, Faivre L, Michot C, Burguet A, Perez-Martin S, et al. (2013) Clinical and Molecular Spectrum of Renal Malformations in Kabuki Syndrome. J Pediatr 163(3): 742-746.
- Barry KK, Tsaparlis M, Hoffman D, Hartman D, Adam MP, et al. (2022) From Genotype to Phenotype-A Review of Kabuki Syndrome. Genes 13(10): 1761.
- Matsumoto N, Niikawa N (2003) Kabuki make-up syndrome: A review. Am J Med Genet C Semin Med Genet 117C(1): 57-65.
- Hamdi Kamel M, Gilmartin B, Mohan P, Hickey DP (2006) Successful long-term outcome of kidney transplantation in a child with Kabuki syndrome. Pediatr Transplant 10(1): 105-107.
- Theodore-Oklota C, Egan S, Paulich M, Evans CJ, Hartman DS, et al. (2020) Caregiver-reported clinical characteristics and the burden associated with Kabuki syndrome. Am J Med Genet A 182(7): 1592-1600.
- Vaux KK, Jones KL, Jones MC, Schelley S, Hudgins L (2005) Developmental outcome in Kabuki syndrome. Am J Med Genet A 132A(3): 263-264.
- Tsai IC, McKnight K, McKinstry SU, Maynard AT, Tan PL, et al. (2018) Small molecule inhibition of RAS/MAPK signaling ameliorates developmental pathologies of Kabuki Syndrome. Sci Rep 8(1): 10779.
- de Los Angeles Serrano M, Demarest BL, Tone-Pah-Hote T, Tristani-Firouzi M, Joseph Yost H (2019) Inhibition of Notch signaling rescues cardiovascular development in Kabuki Syndrome. PLoS Biol 17(9): e3000087.
- Zhang L, Pilarowski G, Pich EM, Nakatani A, Dunlop J, et al. (2021) Inhibition of KDM1A activity restores adult neurogenesis and improves hippocampal memory in a mouse model of Kabuki syndrome. Mol Ther Methods Clin Dev 20: 779-791.
- Morgan AT, Mei C, Costa A da, Fifer J, Lederer D, et al. (2015) Speech and language in a genotyped cohort of individuals with Kabuki syndrome. Am J Med Genet A 167(7): 1483-1492.
- Upton S, Stadter CS, Landis P, Wulfsberg EA (2003) Speech characteristics in the Kabuki syndrome. Am J Med Genet A 116A(4): 338-341.
- Ruault V, Corsini C, Duflos C, Akouete S, Georgescu V, et al. (2020) Growth charts in Kabuki syndrome 1. Am J Med Genet A 182(3): 446-453.
- Evans SL, Kumar N, Rashid MH, Hughes DS (2004) New ocular findings in a case of Kabuki syndrome. Eye (Lond) 18(3): 322-324.
- Chen YH, Sun MH, Hsia SH, Lai CC, Wu WC (2014) Rare ocular features in a case of Kabuki syndrome (Niikawa-Kuroki syndrome). BMC Ophthalmol 14(1): 143.
- Turner CLS, Lachlan K, Amerasinghe N, Hodgkins P, Maloney V, et al. (2005) Kabuki syndrome: new ocular findings but no evidence of 8p22-p23.1 duplications in a clinically defined cohort. Eur J Hum Genet 13(6): 716-720.
- Digilio MC, Gnazzo M, Lepri F, Dentici ML, Pisaneschi E, et al. (2017) Congenital heart defects in molecularly proven Kabuki syndrome patients. Am J Med Genet A 173(11): 2912-2922.
- Yoon JK, Ahn KJ, Kwon BS, Kim GB, Bae EJ, et al. (2015) The strong association of left-side heart anomalies with Kabuki syndrome. Korean J Pediatr 58(7): 256-262.
- Moral S, Zuccarino F, Loma-Osorio P (2009) Double aortic arch: An unreported anomaly with Kabuki syndrome. Pediatr Cardiol 30(1): 82-84.
- Kung GC, Chang PM, Sklansky MS, Randolph LM (2010) Hypoplastic left heart syndrome in patients with Kabuki syndrome. Pediatr Cardiol 31(1): 138-141.
- Boniel S, Szymańska K, Śmigiel R, Szczałuba K (2021) Kabuki Syndrome—Clinical Review with Molecular Aspects. Genes 12(4): 468.
- Tanaka R, Takenouchi T, Uchida K, Sato T, Fukushima H, et al. (2012) Congenital corneal staphyloma as a complication of Kabuki syndrome. Am J Med Genet A 158A(8): 2000-2002.
- Nobili V, Marcellini M, Devito R, Capolino R, Viola L, et al. (2004) Hepatic fibrosis in Kabuki syndrome. Am J Med Genet A 124A(2): 209-212.
- Ito H, Mori K, Inoue N, Kagami S (2007) A case of Kabuki syndrome presenting West syndrome. Brain Dev 29(6): 380-382.
- Lodi M, Chifari R, Parazzini C, Viri M, Beccaria F, et al. (2010) Seizures and EEG pattern in Kabuki syndrome. Brain Dev 32(10): 829-834.
- Akin Sari B, Karaer K, Bodur Ş, Soysal AŞ (2007) Case Report: Autistic Disorder in Kabuki Syndrome. J Autism Dev Disord 38(1): 198-201.
- Mervis CB, Becerra AM, Rowe ML, Hersh JH, Morris CA (2005) Intellectual abilities and adaptive behavior of children and adolescents with Kabuki syndrome: A preliminary study. Am J Med Genet A 132A(3): 248-255.
- Boisgontier J, Tacchella JM, Lemaître H, Lehman N, Saitovitch A, et al. (2019) Anatomical and functional abnormalities on MRI in kabuki syndrome. Neuroimage Clin 21:101610.
- Emmert-Buck LT, Preslan MW, Kathuria SS (2004) Jaw-winking ptosis in a patient with Kabuki syndrome. J Pediatr Ophthalmol Strabismus 41(6): 369-372.
- Cuesta L, Betlloch I, Toledo F, Latorre N, Monteagudo AF (2011) Kabuki syndrome: a new case associated with Becker nevus. Dermatol Online J 17(8): 1.
- Speirs JN, Morris SC, Morrison III MJ (2019) Slipped capital femoral epiphysis in an adult patient with Kabuki syndrome. J Am Acad Orthop Surg Glob Res Rev 3(10): e19.00084.
- Tuna EB, Marşan G, Gençay K, Seymen F (2012) Craniofacial and dental characteristics of Kabuki syndrome: nine years cephalometric follow-up. J Clin Pediatr Dent 36(4): 393-400.
- Utine GE, Alanay Y, Aktas D, Boduroglu K, Alikasifoglu M, et al. (2008) Kabuki syndrome and trisomy 10p. Genet Couns 19(3): 291-300.
- Haanpää M, Schlecht H, Batra G, Clayton-Smith J, Douzgou S (2017) Interrupted/bipartite clavicle as a diagnostic clue in Kabuki syndrome. Am J Med Genet A 173(4): 1115-1118.
- Yoshioka S, Takano T, Matsuwake K, Sokoda T, Takeuchi Y (2011) A Japanese patient with Kabuki syndrome and unilateral perisylvian cortical dysplasia. Brain Dev 33(2): 174-176.
- So PL, Luk HM, Cheung KW, Hui W, Chung MY, et al. (2021) Prenatal phenotype of Kabuki syndrome: A case series and literature review. Prenat Diagn 41(9): 1089-1100.
- Benjamin JS, Pilarowski GO, Carosso GA, Zhang L, Huso DL, et al. (2017) A ketogenic diet rescues hippocampal memory defects in a mouse model of Kabuki syndrome. Proc Natl Acad Sci U S A 114(1): 125-130.
- Schott DA, Gerver WJM, Stumpel CTRM (2017) Growth hormone therapy in children with Kabuki syndrome: 1-year treatment results. Horm Res Paediatr 88(3-4): 258-264.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...