
Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4595
Mini Review(ISSN: 2637-4595) 
Advances in Halogen-Free Flame Retardants Volume 1 - Issue 4
Asim Kumar Roy Choudhury*
- KPS Institute of Polytechnic, India
Received: February 02,2018; Published: February 15,2018
*Corresponding author: Asim Kumar Roy Choudhury, Principal, KPS Institute of Polytechnic, Belmuri, Ex-professor, Govt. College of Engineering and Textile Technology, Serampore, Dt. Hooghly (W.B.) India
DOI: 10.32474/LTTFD.2018.01.000117
Abstract
The challenges of finding substitutes for banned halogen-containing flame-retardants (HFFRs) more specifically bromine-based FRs (BFRs) include lower flame retardancy, high cost, serious melt-dripping and deterioration in mechanical properties of halogen-free products. Phosphorous-based FRs (both additive and reactive types), silicon FRs, intumescent flame-retardants (IFRs), nano-materials including nano composites, bio macromolecules, use of catalysts provide some alternatives to halogen-based FRs.
Keywords: Halogen-free flame retardants; Phosphorous-based flame-retardants; Intumescent flame-retardants; Nano composites; Bio macromolecules; Silicon flame-retardants; Catalysts
Introduction
Types of Flame-retardants (FRs)
The main families of flame retardants are
a. Halogens (Bromine and Chlorine)
b. Phosphorus
c. Minerals
d. Nitrogen
e. Intumescent
f. Nano material based FRs e.g. Nanocomposites.
Halogen-based flame retardants (HFFRs)
The Stockholm Convention (2001) listed 23 organo-halogen chemicals (including all BFRs) to be banned globally. The challenges of finding substitutes for banned halogen-containing flame- retardants include:
A. Lower flame retardancy: Only 10 wt% of BFRs is equivalent to about 30-50% by wt of inorganic FRs
B. High cost: Inorganic FRs is low-cost but require high loading. New HFFRs, on the other hand, are mostly costlier. The cost ratios (£) of BF-FR with phosphorous based and non- phosphorous based HFFR are about 1:6 and 1:2 respectively.
C. Higher thermal stability: Many HFFRs decompose at higher temperature (about 400°C) than their halogenated counterparts (about 3300C).
D. Melt-dripping: Melt dripping occurs with most polymers e.g. polyethylene, polypropylene, PET, ABS and so on. Flammable drips act as secondary ignition sources. FRs should make drips non-flammable.
E. Deterioration in Mechanical Properties: The deterioration of mechanical properties increase proportionately with the amount of FR. Therefore, the deterioration is more with less efficient FRs [1].
Phosphorous based FRs (PFRs)
Phosphorus flame retardants (PFRs) (organic and inorganic) are more environmentally friendly, good thermally stable and have superior performance, resulting from the synergistic effect of P-N. They do not tend to form toxic gases since phosphorus is mostly locked into the char. Phosphorous FRs may be reactive (chemically bound) and additive (physical mixing) [2]. Lanxess [3] has developed is a halogen-free phosphate ester. The product that combines plasticising and flame retardant properties has a low odour and can be used in many plastics, coated textile fibres, tarpaulins, furniture and automotive interiors etc. It was found that non-halogenated phosphorus flame retardants have an environmentally friendly profile e.g. 9, 10-dihydro-9-oxa-10- phosphaphenanthrene-10-oxide (DOPO), (Figure 1).
Figure 1: Chemical Structure of DOPO.
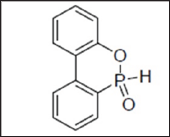
However, the chemical addition of DOPO to the epoxy resin significantly decreases the Tg of the cured epoxy resin. Hence, new multifunctional reactive DOPO derivatives, phosphorus containing oligomers and phosphorus-based curing agents that would not have a detrimental effect on the Tg of the cured resin are subject to extensive research [4]. Similar phosphorus contents are required for DOPO-based additives and for reactive DOPO derivatives to reach UL 94-V0 rating. Even though the reactive approach has yet to be adopted in industry, it has been subject to an ever-growing interest from the academic community. Indeed, the reactive P-H bond of hydrogen phosphonates or phosphinates enables to covalently bind the flame retardant to the polymer chain by reaction with the epoxy functionality [4].
Nitrogen FRs
This class consists of pure melamine, melamine derivatives, i.e. salts with organic or inorganic acids such as boric acid, cyanuric acid, phosphoric acid or pyro/poly-phosphoric acid, and melamine homologues. Combinations of nitrogen-based flame retardants such as melamine polyphosphate and metal phosphinates also demonstrated flame retardancy of composites that has repercussions on printed wiring boards (PWB) [4].
Mineral FRs
Mineral FRs includes aluminium trihydroxide (ATH), Magnesium hydroxide (MDH), talcum or chalk (calcium carbonate). They are inexpensive, but require loadings in polymers about 60% or more.
Intumescent FRs
Intumescence is the result of a combination of charring and foaming at the surface of the burning polymer, which protects the underlying material from the action of the heat or flame. Intumescent FRs (IFRs) is often used for applications requiring high levels of flame retardancy. They are highly efficient and low toxic. They provide very robust fire safety and flame resistance performance. The carbon agent forms multicellular charred layers, the char may be soft or hard [5]. Soft char IFRs: composed of a carbon source pentaerythritol (PER), acid source (ammonium polyphosphate) and a gas-blowing additive (melamine) harder char IFRs: composed of sodium silicates and graphite. These are suitable for use in plastic pipe firestops as well as exterior steel fireproofing.
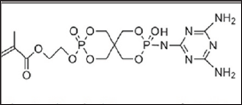
PER is quite costly. A possible substitute is green carbon agent is chitosan (CS), obtained by the alkaline deacetylation of abundantly naturally occurring chitin. A good synergistic effect observed when chitosan/urea compound based phosphonic acid melamine salt (HUMCS), was added to an IFR system for polypropylene (PP) [5]. A reactive, intumescent, HFFRs 2-({9-[(4,6-diamino-1,3,5-triazin-2- yl)amino]-3,9-dioxido-2,4,8,10-tetraoxa-3,9-diphosphaspiro[5.5] undecan-3-yl}oxy) ethyl methacrylate (EADP), synthesized from phosphorus oxychloride, pentaerythritol, hydroxyethyl methacrylate, and melamine. EADP (Figure 2) exhibited excellent thermal stability and char-forming ability without affecting tensile strength of polypropylene [6].
Figure 2: Chemical Structure of EADP.
IFRS for Polyurethane Foam
Figure 3: Chemical Structure of TSPB.
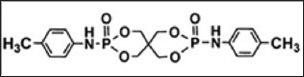
Figure 4: Chemical Structure of ODDP.
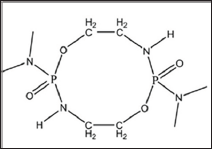
Rigid polyurethane foam (RPUF) is very flammable and releases toxic gases while burning. Traditional IFRs are mixtures, and are not compatible with the RPUF matrix [7]. A phosphorous- nitrogen (P-N) containing IFR, toluidine spirocyclic pentaerythritol bisphosphonate (TSPB), (Figure 3) exhibited better compatibility with RPUF and lesser negative influence on the mechanical properties of TSPB-RPUF forming compact and smooth char [8]. Waterborne polyurethane (WPU) has good applicability including fabric coating as against conventional solvent borne polyurethanes. Octahydro-2,7-di(N,N-dimethylamino)-1,6,3,8,2,7-dioxadiazadiphosphecine (ODDP) (Figure 4), with bi-phosphonyl in a cyclic compound, was synthesized. ODDP reacted phosphorus- nitrogen synergistic HFFR waterborne PU (DPWPU) has excellent flame retardancy due to the presence of ODDP. The LOI value of DPWPU is 30.6% with UL-94, V-0 classification obtained at 15 wt % ODDP [9].
Nano materials
Inexpensive nanoclays form 'nano-network' which spread out and dispersed through the host material forming a sort of gauze. The breakdown of material and the release of gas phase combustible molecules are slowed down or even prevented. Nano materials can be used as:
a) Nano polymer composites
b) Nanoparticles in traditional back-coatings
c) Deposition of nano coatings on the fabric substrates.
The DEROCA project (EU) studies the synergic effect of carbon nanotube (CNT) with phosphorus based flame retardants and other new additives in intumescent or carbon crust formation systems [10]. However, with any nanotechnology the potential for harm is associated with the size and shape of the particles [11].
Nanocomposites
Nanocomposites is a multiphase solid material where one of the phases has one, two or three dimensions of less than 100 nanometres, or structures having nanoscale repeat distances between the different phases that make up the material. Polymer layered-silicate nanocomposites (PLSNs) with silicate enhance mechanical properties, increase heat-distortion temperatures, improve thermal stability, decrease gas/vapour permeability and reduce flammability [12]. The use of P-N flame-retardant with montmorillonite (MMT) as a flame-retardant synergist for flame-resistant thermoplastics such as PP, PA6, and PA66 has been reported [13]. Chemically, montmorillonite is hydrated sodium calcium aluminium magnesium silicate hydroxide (Na,Ca)0.33(Al,Mg)2(Si4O10)(OH)2•nH2O. Montmorillonites swell or expand considerably more than other clays due to water penetrating the interlayer molecular spaces and concomitant adsorption. By exchanging sodium cations for organic cations the surface energy of MMT decreases and the interlayer spacing expands. The resulting material is called organoclay [14].
Thermoplastic poly(ester ether) elastomers (TPEEs) are easily ignited and rapidly burned. TPEE nanocomposites with phosphorus-nitrogen (P-N) flame retardants and organic montmorillonite (o-MMT) were prepared by melt blending. A significant fire-retardant effect was induced in TPEE and rendered a V-0 classification in the UL 94 test [15]. Organically treated layered silicates (clays), carbon nano-tubes/nano-fibres, or other submicron particles at low loading (1-10wt%) are used for polymer nanocomposites. The polymer nanocomposites greatly lower the base flammability of a material. Recent research has suggested that combining nanoparticles with traditional fire retardants (e.g., intumescent) or with surface treatment (e.g., plasma treatment) effectively decreases flammability [16].
When polymer nanocomposite is used, very little additive is needed (no great cost increase) and polymer dripping/flow while burning is reduced. The composite may have multifunctional performance (ex: electrical conductivity from carbon nanotubes) and a balance between flammability and mechanical properties may be maintained. However, careful design and analysis is required for set up of a polymer nanocomposite structure. Many aspects of nanocomposite technology (long-term ageing, environmental hazards, conservation of fire safety principles etc.) are not known.
Silicon FRs
Silicone is eco-friendly, widely available in nature and is easy to prepare flame retardant from it. The silica ash layer can also prevent oxygen from reaching the matrix. A novel, simple and low cost flame retardant containing phosphorus, nitrogen and silicon was synthesized [17]. LOI was improved from 18.0 to 27.1, wrinkle recovery angle increased from 800 to 980. A novel phosphorous-silicone-nitrogen ternary flame retardant (FR), [(1,1,3,3-tetramethyl-1,3-disilazanediyl)di-2,1-ethanediyl] bis(diphenylphosphine oxide) (PSiN) was synthesized. An UL 94 V-0 rating was achieved with 20 wt % PSiN in the epoxy composites. Silica tended to migrate to the surface of polymer for its low surface energy.
Nitrogen components turned into gas in the combustion process. Phosphorus components played a role of FR in both condensed phase and gas phase. All the results indicated that current P-Si-N ternary FR might be a good candidate for epoxy flame retardancy [18]. Three novel phosphorus-based flame retardant monomers were successfully prepared by reacting 1-(acryloyloxy)-3-(methacryloyloxy)-2-propanol (AHM) with hexachlorocyclotriphosphazene. With the aid of a photo initiator and a UV flood curing system, these monomers were cured onto cellulosic substrates to generate flame retardant coatings. All three monomers formed coatings under UV conditions in the presence of the photo initiator that were durable to extraction. Only 15 % of these monomers were required to prevent the substrates from combusting in the horizontal, 450 and vertical orientations [19]. Recently silicon-containing monomers have successfully used as reactive-type HF FRs, especially polyhedral oligomeric silsesquioxanes (POSS), due to their notable reinforcing effect [20].
Bio macromolecular FRs
Very recently, bio macromolecules such as proteins (whey proteins, caseins, hydrophobias) and deoxyribonucleic acid (DNA) have shown unexpected flame retardancy on cellulosic or synthetic substrates [21]. These proteins are waste or by-products from the cheese and milk industry, DNA is costly, but DNA was extracted and purified from salmon milt. The mechanism is still under investigation -a stable and protective char (i.e. a carbonaceous residue) formed. Compared to proteins, DNA shows unique behaviour, since it contains the three main ingredients of an intumescent formulation all in one molecule. Phosphate groups able to produce phosphoric acid, the deoxyribose rings act as a carbon source and blowing agents (upon heating they may dehydrate forming char and releasing water) and the nitrogen-containing bases may release ammonia, DNA-treated cotton fabrics showed outstanding self-extinguishment features [21].
Possible new technologies
So far a very small amount of the periodic table has been utilised as FRs. New undiscovered chemistries may be far more effective and useful than halogen, phosphorus, or any existing technology [22].
Catalysts
Some divalent and multivalent metal compounds on the flame retardancy of intumescent FRs - LOI increased by 7-9 units and grade in vertical burning test (UL-94) improved from V-2 to V-0. Various organic and inorganic have been chosen as catalysts with conventional FRs or alone. Metal compounds which convert the flammable polymer to graphite under fire conditions.
Low-melting glasses and ceramics
Such material can set up an inorganic (cannot be oxidized or burned further) protective layer.
New vapour phase combustion inhibitors
Oxygen scavengers or even more potent free-radical inhibitors can be used as FRs. The persistence, bioaccumulation, and toxicity (PBT) of a selection of HFFRs are to be studied. Some preliminary studies have already been made [23].
Microencapsulation
Melamine-formaldehyde polymer-shell microcapsules, containing resorcinol bis(diphenyl phosphate)) as the core substance, were coated by an outer thermoplastic wall (polystyrene (PS) or poly(methyl methacrylate)), before being applied to a core/sheet-type bi-component PET/co-PET spunbond nonwoven substrate using impregnation. The presence of microcapsules has a low impact on the air permeability and thickness of the nonwoven samples, even if they are more impregnated on the surface than in the inner structure. The flame spread rate was relatively low for all of the tested formulations [24]. Ethylene vinyl acetate (EVA) is an essential polymer in HFFR compounds used in thermoplastic and crosslinked wire and cable applications, with higher VA levels improving filler loading, flame retardancy and flexibility [25].
Future Trends
The market size of HFFRs was USD 3.36 Billion in 2015 and is projected to reach USD 5.38 Billion by 2021, registering a Compound Annual Growth Rate of 8.4% between 2016 and 2021. The most preferred HFFR is ATH followed by organo-phosphorus chemicals, and others. The factors driving the ATH market are its low cost, easy availability, and high use in all the sectors [26]. Future FR materials should be [22]:
a) Passes new flammability Test (based upon fire risk scenarios and fire safety principles).
b) Environmental friendly, recyclable and sustainable.
c) Recyclable could mean energy recovery until we can truly separate out the additives and recycle them.
d) Inexpensive.
The replacement of halogen-containing FRs by halogen-free FRs is a long-term process. The complete replacement of halogen-based FRs will be faced with many challenges. A feasible and practical strategy is to follow two routes. Firstly, the existing halogen-based FRs which cannot be replaced should be used selectively and in reduced quantities. Secondly, novel HF FRs are to be developed which may be based on phosphorous and/or nitrogen organic compounds, inorganic compounds, especially nano-materials.
References
- http://www. davidsuzuki.org/issues/health/science/ envirohealthpolicy/toxicflameretardantsareaburningissue/accessed on 23.3.16.
- Schartel B (2010) Phosphorus-based Flame Retardancy Mechanisms- Old Hat or a Starting Point for Future Development? Materials 3(10): 4710-4745.
- http://www.mrcplast.com/, Lanxess developed new flame retardants, August 16/2016.
- Rakotomalala M, Wagner S, Döring M (2010) Recent Developments in Halogen Free Flame Retardants for Epoxy Resins for Electrical and Electronic Applications. Materials 3(8): 4300-4327.
- Xiao Y, Zheng Y, Wang X, Chen Z, Xu Z (2014) Preparation of a Chitosan- Based Flame-Retardant Synergist and Its Application in Flame-Retardant Polypropylene. Journal of Applied Polymer Sciences 1-8.
- Li G, Wang W, Cao S, Cao Y, Wang Y (2014) Reactive, Intumescent, Halogen-Free Flame Retardant for Polypropylene. Journal of Applied Polymer Science.
- Kim J, Nguyen C (2008) Thermal stabilities and flame retardancies of nitrogen-phosphorus flame retardants based on bisphosphoramidates. Polymer Degradation and Stability 93(6): 1037-1043.
- Wu DH, Zhao PH, Liu YQ, Liu XY, Wang XF (2014) Halogen Free Flame Retardant Rigid Polyurethane Foam with a Novel Phosphorus-Nitrogen Intumescent Flame Retardant. J Appl Polym Sci.
- Gu L, Ge Z, Huang M, Luo Y (2015) Halogen-Free Flame-Retardant Waterborne Polyurethane with a Novel Cyclic Structure of Phosphorus- Nitrogen Synergistic Flame Retardant. J Appl Polym Sci 1-9.
- DEROCA Project, http://cordis.europa.eu/project/rcn/105644_en.html, accessed on 10.8.16 2016.
- Kellyn SB (2008) New Thinking on Flame Retardants. Environ Health Perspectives 116(5): A210-A213.
- Sinha SR, Masami O (2003) Polymer/layered silicate nanocomposites: a review from preparation to processing. Progress in Polymer Science 28(11): 1539-1641.
- Gilman JW (1999) Flammability and thermal stability studies of polymer-layered-silicate(clay) nanocomposites. Applied Clay Science 15(1-2): 31-49.
- Singla P, Mehta R, Upadhyay SN (2012) Clay Modification by the Use of Organic Cations. Green and Sustainable Chemistry 2(1): 21-25.
- Zhong Y, Wu W, Lin X, Li M (2014) Flame-Retarding Mechanism of Organically Modified Montmorillonite and Phosphorous-Nitrogen Flame Retardants for the Preparation of a Halogen-Free, Flame-Retarding Thermoplastic Poly(ester ether) Elastomer. Journal of Applied Polymer Science 131: 41094.
- Bourbigot S, Duquesne S, Jama C (2006) Polymer Nanocomposites: How to Reach Low Flammability? Macromol Symp 233(1): 180-190.
- Zhou T, He X, Guo C, Yu J, Lu D, et al. (2015) Synthesis of a novel flame retardant phosphorus/nitrogen/siloxane and its application on cotton fabrics. Textile Research Journal 85(7): 701-708.
- Li ZS, Liu JG, Song T, Shen DX, Yang SY (2014) Synthesis and characterization of novel phosphorous-silicone-nitrogen flame retardant and evaluation of its flame retardancy for epoxy thermosets. Journal of Applied Polymer Science 40412.
- Edwards B, Hauser P, El-Shafei A (2015) Nonflammable cellulosic substrates by application of novel radiation-curable flame retardant monomers derived from cyclotriphosphazene. Cellulose 22(1): 275-287.
- Bourbigot S, Le Bras M, Flambard X, Rochery M, Devaux E, et al. (2005) Polyhedral oligomeric silsesquioxanes: Application to flame retardant textiles. Fire Retardancy of Polymers: New Applications of Mineral Fillers. (In Eds Le Bras M, Bourbigot S, Duquesne S, Jama C, Wilkie, CA, Royal Society of Chemistry (Pub)) pp. 189-201.
- Alongi J, Bosco F, Carosio F, Di Blasio A, Malucelli G (2016) A new era for flame retardant materials? Materials Today 17(4): 152-153.
- Alexander BM Polymer Flame Retardant Chemistry, FR Chemistry
- Waaijers SL, Kong D, Hendriks HS, de Wit CA, Cousins IT, et al. (2013) Persistence, bioaccumulation, and toxicity of HF-FRs. Rev Environ Contam Toxicol. 222: 1-71.
- Butstraen C, Salaün F, Devaux E, Giraud S, Vroman P (2016) Application of Flame-Retardant Double-Layered Shell Microcapsules to Nonwoven Polyester. Polymers 8(7): 267.
- Formulations for halogenfree flame retardant (HFFR) cables.
- Markets and Markets, Report Code: CH 4218, Publishing Date: April 2016.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...












