Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4749
Research Article(ISSN: 2637-4749) 
Presence of Beneficial Bacteria In Kefir-Enriched Ice Cream During Shelf Life Volume 5 - Issue 1
Tuğba Kök Taş*, Saliha Bahar and Büşra Sapmaz
- Department of Food Engineering, Faculty of Engineering Suleyman Demirel University, Turkey
Received: July 04 ,2022; Published: July 15, 2022
Corresponding author: Tuğba Kök Taş, Department of Food Engineering,Faculty of Engineering Suleyman Demirel University, Isparta, Turkey
DOI: 10.32474/CDVS.2022.05.000203
Abstract
Kefir is a magnificent source of beneficial bacteria; it also contains a variety of nutrition components. The aim of study was determined microbiology, and sensory properties during 60 days storage period of ice-cream produced using kefir. The aim of the study is primarily to determine the presence of beneficial bacteria for shelf life. In addition, its chemical and sensory properties were studied. Ice-cream produced using kefir (rate 50% or 75%) were determined the counts of total aerobic mesophilic bacteria, lactic acid bacteria, lactococcus, probiotic bacteria (L. acidophilus, Bifidobacterium spp.); chemical parameters (pH and total solids levels); some physical properties (colour, viscosity); scores for texture, odour, taste, and over-all sensory properties in ice-cream samples during storage time. The results suggested that the growth of benefical bacteria in kefir-enriched ice cream, which resulted in an improved viability of these organisms during storage. The influence of using different ratios of kefir was not apparent in relation to other physico-chemical properties and sensory attributes of the product. According to the results of this experiment, a product with a very high content of beneficial bacteria was produced as an alternative to ice cream.
Keywords:Kefir; ice-cream; benefical bacteria; physical ;sensory properties
Introduction
Kefir is a dairy fermented drink. Traditional kefir produces with kefir grains and it provides benefical lactic acid bacteria include Lactobacillus acidophilus, Bifidobacterium spp., and some yeasts [1]. Content of benefical microorganisms in the kefir has microorganisms of probiotic properties which is generate functional fermented diary foods. Therefore kefir has therapeutic and nutritional properties. Kefir has both therapeutic and nutritional attributes, such as high contents of probiotic microoganisms and LAB in all seeking to develop functional fermented dairy products. Bifidobacterium species and lactic acid bacteria, especially Lactobacillus species are used widely as probiotics in foods and beverages. They known as safe (GRAS) [2].Probiotics has various benefits, include prevention of constipation, colon cancer, lactose intolerance, infantile diarrhoea, travellers’ diarrhoea, antibiotic induced diarrhoea, hypercholesterolaemia, vaginitis and intestinal infections [3]. The production of ice cream including number of probiotic bacteria connecting with survival of probiotics from during this proccessing as well as frozen storage [4]. Ice cream has been shown as an sufficient food matrix that can combine with probiotic bacteria but the information about the effect of being exceed normal levels on the survival bacteria during production of ice cream restricted as well as the sensory admission of this type of product [5]. The aim of this study was to detemined presence of beneficial bacteria ın kefir-enriched ice cream during storage.
Materials and Methods
Materials
The pasteurised milk and pasteurised cream (13,5% milk fat) used in ice cream mix were supplied from Unsüt Co (Isparta, Turkey). The solid ingredients used in the experiment included nonfat milk powder (Enka Dairy Inc., Konya, Turkey), sugar (Dogus sugar, Gebze, Kocaeli,Turkey). Guar Gum, Locust Bean Gum, CMC, Carragenan, Sodium Alginate as stabiliser (Maymix DIC 20K, Maysa Food Inc., Tuzla, Istanbul, Turkey) were blended for artisanal style ice-cream application. Kefir was used as probiotic in ice cream mix that it was obtained from Danem Dairy Product Co(Tecnopark,Isparta,Turkey).
Kefir processing
Kefir have produced from natural kefir grains (2%) at 24 ᵒC for approximately 22 h. The fermentation was ended at pH 4.9±0.2 for this research at Danem Dairy Product Co. (Tecnopark, Turkey). Natural kefir contains probiotic microorganism, lactic acid bacteria and yeast.
Production of ice-cream
Batches of ice cream were manufactured in the pilot plant of Kulah ice cream factory (Isparta,Turkey). The ice cream mix formula consisted of 13.5% fat, 4% (wt/wt) nonfat milk powder, 18% (wt/ wt) sugar and 0.6% (wt/wt) stabilizer–emulsifier. The increased fat of milk was heated to 40–45°C before being mixed with solid ingredients. The mixture was homogenized at a temperature close to 75 °C, at a pressure close to 1.33×107 Pa, and was subsequently subjected to heat treatment at 85° C for 30 min and was then cooled to 4° C. The ice cream mix was matured for overnight after being cooled to 4°C. The mixture was subdivided into three portions. The different ratios of kefir was determined by preliminary studies. The treatments were performed ice cream without kefir (IC-P),ice cream added 50% kefir (IC-LK), or ice cream added 75% kefir (ICHK).
The kefir was added in order to obtain a high initial lactic acid bacteria count of 107–108 cfu⁄ml and probiotic bacteria count 106–107 cfu⁄ml. Kefir was mixed in the matured ice cream mix. A Gram brand FH 151 scraped-surface freezer (Gram Equipment A/S, Denmark), with a capacity of 50 L/h and a draw temperature of –10 °C, was used to prepare the ice cream. Following aging at 4 °C for 24 h to ensure complete hydration of all ingredients, the mix was frozen in a Mantematic KEL 60S batch freezer (Cattabriga S.r.). The ice cream was packaged into 100 mL plastic lid containers and then at the end of 15, 30, 45 and 60 days of storage at −25°C. The described procedure was repeated three times.
Microbiological analyses
Viable microbial counts were determined in the ice creams at 1, 15, 30, 45, and 60 d of frozen storage. Lactic acid bacteria counts were performed on MRS agar (de Man, Rogosa, and Sharpe, Merck, Darmstadt, Germany) after incubation at 37 ᵒC under anaerobic conditions (6% CO2) for 72 h. Lactic streptococ bacteria were enumerated on M17 medium (Merck, Darmstadt, Germany) at 37°C under anaerobic conditions (6% CO2) for 48 h. Yeasts were grown on potato dextrose agar (Merck, Darmstadt, Germany) with addition of 0.14% lactic acid at 25°C for 5 d [6]. The viability of Lactobacillus acidophilus were determined on MRS with addition of 10% (wt/vol) sorbitol [7] and the viability of Bifidobacterium spp. were performed on MRS agar supplemented with neomycin, nalidixic acid, lithium chloride, and paromomycine sulfate [7] at 37°C under anaerobic conditions (6% CO2) for 72 h.
Chemical analyses
The pH values of ice cream sample was determined by Beckman Zeromatic SS-3, USA. Determination of fat content was carried out aqccording to Gerber Method. 3g ice cream sample was placed in a butyrometer (butyrometer for cheese, 40%) and 10 ml sulfuric acid (d=1,55, 90%) was added than butyrometer plug was closed. Sample was dissolved in water bath (68 ). 1 ml amyl alcohol (d=0,812-0,818) was added in dissolved sample and up to 40% of butyrometer was filled with sulfiric acid, subsequently butyrometer’s plug was closed again. The mixture was centrifuged (1200 rev/min, 5min). Determination of total solid was carried out according to electrical conductance method. 3g ice cream sample was placed in device, 5-10 min later, dry matter of labne sample was read on device.The color of ice cream samples were determinated by Konica Minolta Color Reader and the results were obtained in the form of two parallel.The viscosity of ice cream samples were determinated by Brookfield Viscosimeter with using LV4 spindle and the results were obtained in the form of two parallel.
Sensory evaluation
Twelve panelists (four male and eight female) working in the Department of Food Engineering at Suleyman Demirel University (Isparta, Turkey). Panelists were selected from volunteer graduate students and academic staff of the Department of Food Engineering. The panelists (n = 12) received a 2h training session on descriptive analysis technique, including basic tastes and flavor identification, and using 7-point product specific scale with references [8]. Ice cream samples (50 g; two scoop) were served and presented to the panelists in cups situating a random 3-digit number by styrofoam plates; each sample was scored individually. Samples were presented to the panelists in individual plastic cups, and bread sticks and a glass of water were given between each sample. Sensory evaluation was based on 7-point scales from 1 = absence of the attribute, to 7 = extremely high intensity of the attribute. Each sample was coded using a three digit random number and served successively to the panelists in individually partitioned booths. Serving order was completely randomized. The sensory attributes appearance, odor, color, texture, fermented taste, and overall acceptability were evaluated [9]. In order to achieve homogeneous conditions during all sensory analysis sessions, samples were tempered at first day and sixtyth day.
Statistical analysis
All analyses were replicated 3 times and performed at five times of storage (0, 15, 30, 45 and 60 days) in duplicate. The data were analyzed using SPSS Base 16.0 (SPSS Inc., Chicago, IL). Results for the analyses of rheological parameters and EPS content of samples were statistically evaluated by repeated-measures variance analyses technique. Differences within groups were determined by Tukey test, a multiple comparison test was used to compare means when the effect was significant (P<0.05). In addition, statistical significance was given in terms of P values, with differences at the 95% confidence interval (P<0.05) being considered statistically significant.
Results and Discussion
The chemical characteristics of the ice cream products were given in Table 1. The pH values of IC-P, IC-LK, and IC-HK were 6.82, 6.90, and 6.71, respectively. Our results were in deal with prior records demonstrated lower pH values in probiotic inoculated ice cream samples [10-13]. The total solid results showed that fortification with natural kefir decreased the total solid of the samples compared with the control (P < 0.05). . In terms of total solid content of ice cream products, [13] reported that inoculation of S. boulardii decreased the total solid content of ice cream products whereas Pandiyan et al. (2012) reported that inoculation of S. boulardii increased the total solid content of ice cream products. Color feature of foods is one of the most critical quality indicators accepted by consumers [14]. Table 1 shows the color values (L* (brightness), a* (redness) and b* (yellowness)) of ice cream samples. L* values of ice cream samples were 85.48, 86.83, and 87.85, respectively. For ice cream samples, addition of kefir (50%) and kefir (75%) resulted in higher L* values compared to control (P ≤ 0.05). The viscosity values for all the ice cream samples containing different concentrations of kefir were significantly (P < 0.05) (Table 1). The viscosity of the ice cream samples without kefir recorded the highest viscosity value (34.85 Pa.s). Then, the viscosity was lowered, followed by ice cream, which incorporated 75% kefir (19.10 Pa.s), that with 75% kefir (27.95 Pa.s).
a,bMeans with different letters in the same column are significantly different (P < 0.05).
Treatments: IC-P= control, ice cream without kefir, IC-LK= ice cream added 50% kefir, IC-HK= ice cream added 75% kefir.
The viability of lactic acid bacteria from natural kefir on day 1, day 15, day 30, day 45, and day 60 was found to be the highest in ice cream without kefir (4.76, 4.55, 4.15, 4.02 and 3.89 log CFU/mL, respectively), followed by 50% kefir (7.50, 6.91, 6.80, 6.67, and 5.54 log CFU/mL, respectively) and 75% kefir (7.74, 7.39, 6.88, 6.74, and 6.60 log CFU/mL, respectively; Table 2).
Table 2: Lactic acid contents and mesophilic aerobic bacteria (g/100 g; means ± SD) of probiotic ice creams during storage (n= 3).
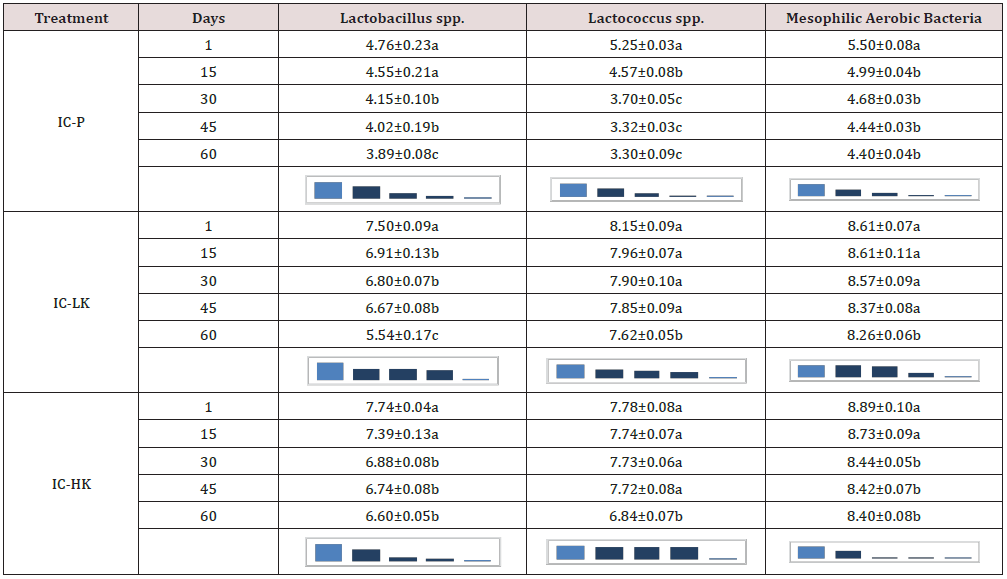
a,bMeans with different letters in the same column are significantly different (P < 0.05).
Treatments: IC-P= control, ice cream without kefir, IC-LK= ice cream added 50% kefir, IC-HK= ice cream added 75% kefir.
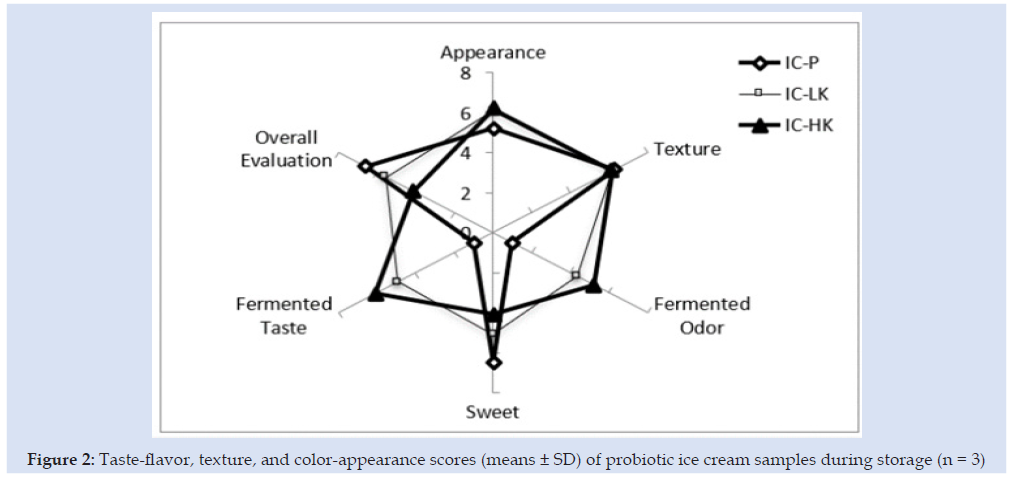
Probiotic products should contain viable probiotic microorganisms at numbers ranging from 106 to 109 CFU/g during their shelf life forbetter functionality [15]. L acidophilus and Bifidobacterium spp. content of ice cream production with kefir (50 %) were determined at level of 7.41 and 6.36 log CFU/g, respectively at the first day. L acidophilus and Bifidobacterium spp. content of ice cream production with kefir (75%) were determined at level of 7.24 and 6.66 log CFU/g, respectively at the first day. Also, decreasing of viable counts during storage period could be due to the storage at low temperature (-18 °C). It should be also noted that following the 60 d of storage time, viable counts of L acidophilus and Bifidobacterium spp. of ice cream production with kefir (50 %) were at 7.35 and 3.15 log CFU/g levels, respectively (Figure 1). Viable counts of L acidophilus in the IC-LK was maintained during storage. Sensorial characteristics of the products critically affect the acceptance of the newly developed product and it has become a big challenge for the food industry. The overall acceptability of the prepared ice cream in terms of color, texture, and taste of free and encapsulated probiotic ice cream was noted (Figure 2) From a sensory analysis score, it was concluded that the without kefir and with kefirs had significantly affected the sensory. The overall evaluation of the ice cream samples without kefir recorded the highest value (6.60). The overall evaluation of the IC-LK and IC-HK were determined scores as the 5.55 and 4.14.
Figure 1: Survival of natural kefir containing Lactobacillus acidophilus and Bifidobacterium spp. (log CFU/g) in the ice cream samples during 60 day of storage period
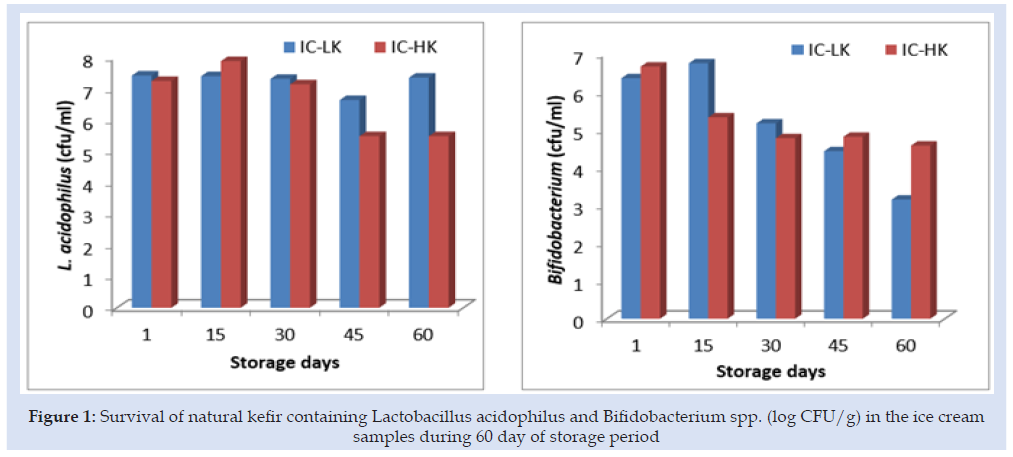
Figure 2: Taste-flavor, texture, and color-appearance scores (means ± SD) of probiotic ice cream samples during storage (n = 3)
Conclusion
The addition of kefir into ice cream mixes yielded greater improvement in microorganism content compared with control and other experimental samples. However, overall scores were lower in samples with high percentage kefir. In the manufacture of probiotic ice cream, natural kefir has potential to improve benefical microorganisms content while maintaining sensory properties and probiotic viability.
References
- Erdogan FS, Ozarslan S, Seydim ZG, Kök Taş T (2019) The effect of kefir produced from natural kefir grains on the intestinal microbial populations and antioxidant capacities of Balb/c mice. Food Research International 115: 408-413.
- Nyanzi R, Jooste PJ (2012) Cereal-based functional foods. Rigobelo EC (Eds.), Probiotics InTech pp. 161-197.
- Rousseau V, Lepargneur JP, Roques C, Remaud-Simeon M, Paul F (2005) Prebiotic effects of oligosaccharides on selected vaginal lactobacilli and pathogenic microorganisms. Anaerobe 11(3): 145-153.
- Mohammadi R, Mortazavian AM, Khosrokhavar R, Cruz AG (2011) Probiotic ice cream: Viability of probiotic bacteria and sensory properties. Annals of Microbiology 61: 411- 424.
- Song D, Khouryieh H, Abughazaleh AA, Salem MME, Hassan O, et al. (2011) Sensory properties and viability of probiotic microorganisms in chocolate ice cream supplemented with omega-3 fatty acids. Milchwissenschaft 66(2): 172-175.
- Mossel D AA, Mossel JEL, Corry CB, Struijk RM (1995) Baird Essentials of the Microbiology of Foods: a textbook for advanced studies, Wiley, England pp. 175-214.
- Shah N P (2000) Probiotic bacteria: Selective enumeration and survival in dairy foods. J Dairy Sci 83(4): 894-907.
- Meilgaard M, Civille G, Carr B T (1999) Sensory Evaluation Techniques, CRC Press, Boca Raton, FL and New York, NY, USA.
- Soukoulis C, Tzia C (2010) Response Surface Mapping of The Sensory Characteristics and Acceptability of Chocolate Ice Cream Containing Alternate Sweetening Agents. Journal of Sensory Studies,25(1):50-75.
- Abghari A, Sheikh-Zeinoddin M, Soleimanian-Zad S (2011) Nonfermented ice cream as a carrier for Lactobacillus acidophilus and Lactobacillus rhamnosus. International Journal of Food Science and Technology 46(1): 84-92.
- Alamprese C, Foschino R, Rossi M, Pompei C, Corti S (2005) Effects of Lactobacillus rhamnosus GG addition in ice cream. International Journal of Dairy Technology 58(4): 200-206.
- Niamah AK, Al-Manhel AJ, Al-Sahlany STG (2018) Effect Microencapsulation of Saccharomyces boulardii on Viability of Yeast in vitro and Ice Cream. Carpathian Journal of Food Science & Technology 10(3): 100-107.
- Sarwar A, Aziz T, Al-Dalali S, Zhang J, Ud Din J, et al, (2021) Characterization of synbiotic ice cream made with probiotic yeast Saccharomyces boulardii CNCM I-745 in combination with inulin. LWT 141.
- Chranioti C, Nikoloudaki A, Tzia C (2015) Saffron and beetroot extracts encapsulated in maltodextrin, gum Arabic, modified starch and chitosan: Incorporation in a chewing gum system. Carbohydrate Polymers 127: 252-263.
- Casarotti SN, Penna ALB (2015) Acidification profile, probiotic in vitro gastrointestinal tolerance and viability in fermented milk with fruit flours. International Dairy Journal 41: 1-6.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...