Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2637-4749
Research Article(ISSN: 2637-4749) 
Histochemical and Histomorphometrical Studies on the Digital Cushion of Heifers and Dairy Cows with Claw Horn Lesions Volume 3 - Issue 1
Celal İzci1, İlhami Çelik2*, Muharrem Erol3 and Ebru Gökşahin4
- 1Department of Surgery, Faculty of Veterinary Medicine, University of Selçuk, Konya, Turkey
- 2Department of Histology and Embryology, Faculty of Veterinary Medicine, University of Selçuk, Konya, Turkey
- 3Department of Surgery, Faculty of Veterinary Medicine, Balıkesir University, Balıkesir, Turkey
- 4Department of Surgery, Faculty of Veterinary Medicine, University of Selçuk, Konya, Turkey
Received: August 20, 2019; Published: August 29, 2019
Corresponding author: İlhami Çelik, Department of Histology and Embryology, Faculty of Veterinary Medicine, University of Selçuk, Konya, Turkey
DOI: 10.32474/CDVS.2019.03.000151
Abstract
Background: The ruminant claw horn lesions seriously diminish histological structure and functional efficiency of the digital cushion of the dairy cows.
Objective: To investigate histological and histochemical properties of the digital cushion in the dairy cow with claw horn lesions, such as white line disease, sole damage and hemorrhage.
Results: The results showed that digital cushion loses its normal histological structure in claw horn lesions.
Conclusion: Further biochemical, physiological and feeding studies on the experimentally induced claw horn lesions are necessary to understand the nature of the events playing roles involving animal feeding in the development of the lesions, to develop new animal feeding strategies aiming to protect and maintain the normal structure of the digital cushion.”
Keywords:Protein Degradability; Goats; Intensive Feeding; Nitrogen Balance; Digestibility
Introduction
Lameness is one of the most important problems negatively affecting milk yield and animal welfare in modern dairy farming in high-performance animals. The cows with laminitis produce lower amount of milk [1-5]. Moreover, cyclic reproductive changes of these cows slow down [6] or cease because of anoestrus [7] or cystic ovarian disease [8]. Lameness is a major animal welfare problem because that it is a painful condition and causes to behavioral changes of the affected cow. A lame cow tends to display low mobility [9], to spend decreased daily feeding time and dry matter intake [10] and lying behaviour may be indicative [11]. In the dairy cow, claw horn lesions [CHL] constitute 65% of the total lesions causing to lameness [12,13]. Although possible roles of risk factors, such as claw conformation and trimming, animal welfare, management, feeding and housing have been evaluated in detail by previous researchers [14-17], pathophysiology of CHL is still unclear. Previous researchers [18,19] have suggested that CHL is arisen from traumatic lesions of the supportive tissues of the claw. Around the time of calving, increase of hoofase enzyme, oestrogen and relaxin secretions results in loosening and increased mobility of suspensor apparatus of third phalanx [19]. The bovine digital cushion locating under the third phalanx has a complex structure, which major part is constituted of fat tissue. The cushion comprises three cylindrical segments [axial, medial and abaxial segments] of adipose tissue surrounded by a thick connective tissue trabecules, those arranged in parallel to each other [20,21]. The pads of the bovine digital cushion play crucial roles in reducing the body load transferred to surface of sole via absorbing of substantial forces acting within the claw [22]. The biomechanical function of the digital cushion in distributing and attenuation of the load transferred to the base of flexor process of the distal phalanx have been well appreciated by previous researchers [21,23,24]. The aim of the present study was to determine histological and histochemical properties of the digital cushion in the heifers and primiparous, second and third parturitions, and multiparous dairy cows managed under similar feeding, housing and welfare conditions. The foot was chosen from the animals with CHL, such as white line disease, sole damage and haemorrhage, in order to define the histology and histochemical changes in the digital cushions of the animals.
Material and Methods
Animals
This experiment was approved by the Ethical Committee of
Experimental Animal Production and Research Centre (2011/023)
of Veterinary Faculty of Selçuk University. In the study, the
120-cadaver foot (totally 240 hooves) of Holstein heifers and
cows were used as materials. The animals were culled because
of moderate to severe CHL and lameness in one or more hooves.
The claws of the animals with similar feeding, housing and welfare
conditions were subjected to a detailed clinical examination prior
to slaughter. Information on the age, number of parturitions, calving
history and stage of lactation were and recorded, in addition to
the clinical findings. Among the reasons given for culling were
insufficient milk yield, lameness and infertility. The animals were
divided into 5 groups, each consisted of 6 animals, as
group I: heifers,
group II: primiparous cows,
group III: cows with second parturition,
group IV: cows with third parturition,
group V: multiparous cows with more than three calving.
The first group animals were 20 months old averagely; other
groups were between 2.6-6.2-year-old. After slaughtering, all the
foot was cleaned, and lesions, such as discoloration, sole ulcer and
hemorrhages were recorded, frozen at -20°C and kept until use for
histological procedure.
Dissection of the Hooves and Obtaining Tissue Samples
Each foot was thawed. The soft tissues of the sole and heel of each hoof were separated following the horn had been removed, in order to fully expose the axial, middle and abaxial pads of the digital cushion (Figures 1A-B & Figures 2A-C) [18]. Axial pad of the cushion was totally removed, fixed in 10% phosphate buffered (0.1M pH 7.4) formal-saline and used for histological procedures.
Figure 1: The study area, green - places of mass appearance of painted lady butterflies (Vanessa cardui L.) in 2019.
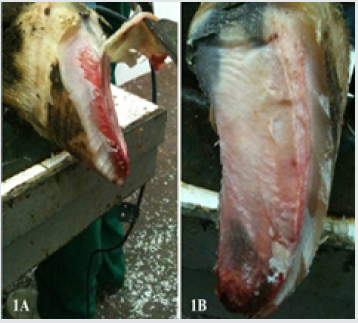
Figure 2: Separating the corium (2A), dissecting pads of the digital cushion (2B) and totally removing the pads (2C).

Histology and Histomorphometry
The tissue samples were subjected to routine histological tissue processing. Briefly, the tissue samples were washed with tap water overnight, dehydrated in ethylene series, cleared in xylene and immersed in paraffin. The sections taken at 6μm were stained as follows; hematoxylin-eosin for investigating general histology and histological changes [25], safranin-O (SO) for histochemical properties of ground substance, alcian blue at pH 2.5 (AB 2.5) for staining acid mucins and mucosubstance, alcian blue at pH 4.0 (AB 4) for demonstrating carboxylated glucose aminoglycans in the ground substance, trichrome stain for Type I collagen fibres (Coll I) and other histological detail, Verhoef’s elastic fibre (EL) stain for EL fibers [26]. The specimens were investigated under light microscope equipped with digital imaging system and digital images were recorded. The images were analyzed with digital image analysis software (BS200 PRO-2005).
Statistical Analysis
The data obtained from the digital image analyses were analyzed statistically with one-way variance analysis (ANOVA) and Tukey’s pair wise comparison tests by using SPSS software 14.01 (Release 14.01 Licence No: 9869264).
Results
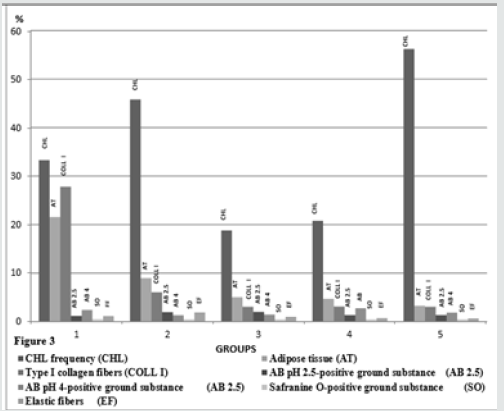
Clinical findings of the animals prior to slaughter and results of postmortem gross examination of the foot Welfare and management of the animals included into the study were far from than satisfactory. The animals were housed on the concrete floor, under poor hygiene conditions, and claw trimming was irregular. The animals were mainly fed with corn silage, alfalfa, vetch and hay. Findings of the postmortem examinations of the foot showed that the animals suffered from CHL, such as sole ulcers, hemorrhage and folding, white line disease and sole erosions in one or more hooves. The highest percentage of the claws with CHL was in the multiparous cows, in the group 5. The groups 2 and 1 the group followed it respectively. The lowest lower CHL frequencies were found in the groups 3 and 4, and the frequencies of these groups were quite similar (P>0.05, Figure 3).
Gross Macroscopic Findings of the Cushion Segments
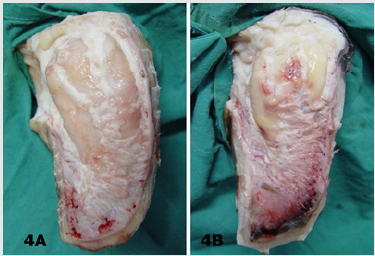
In most of the claws with CHL, segmental structure of the digital cushion was diminished. The digital cushion tissues of the heifers (Group 1) were pale and softer, whereas in the cows with the increasing the parturition number, the tissues gained yellowish colour and relatively more brittle and fragile. The cushion segments were also smaller in the animals of the groups 4 and 5, aged and multiparous cows (Figures 4A & 4B).
Figure 4: Macroscopic appearance of digital cushion tissues of a heifer from the group 1 (4A) and a cow from the group 5 (4B). The cushion pads of the group 5 animal are smaller and yellowish in colour.
Histological Findings
In general, the digital cushion pads of the animals were mainly formed of a loose connective tissue and unilocular adipose tissue. Adipocytes appeared as large cells with unstained cytoplasm in all of the used staining procedures (Figures 5-9). Fibrous connective tissue septa surrounded the loose connective and adipose tissues. There were striking structural differences in the histology of the digital cushion of heifers and calved cows. Loose connective tissue in the heifers, was replaced with adipose tissue in the primiparous cows, and fibrous connective tissue rich in collagen fibres in the third lactation cows. In the heifers and primiparous cows, adipocytes were seen as groups, whereas in the cows of the groups 3, 4 and 5, the cells were observed as individual cells and the cell groups consisting of small numbers of the adipocytes. Adipose tissue percentages of the groups are given in the Table 1. The highest adipose tissue percentage (21.55%) was in the heifers, group 1, and the value was significantly (P<0.001) higher than those of the other groups (Figure 3). The groups 3 and 4 displayed similar (P>0.05) adipose tissue percentages. The multiparous animals (group 5) had the lowest adipose tissue percentage (Figure 3). Type I collagenous fibres were seen as thick undulating bundles of the fibres darkly stained with aniline blue in trichrome stained sections (Figures 5A- 5E).
Figure 5: Collagen fibre bundles (arrows) and adipocytes (asterisks) are seen in the sections from lateral claw cushion of the right forefoot of the heifer from the group 1 (5A), in the medial claw cushion of the left hindfoot of the animal from the group 2 (5B), in the lateral claw cushion of the left forefoot of the animal from the group 3 (5C), medial claw cushion of the left hindfoot of the animal from the group 4 (5D) and lateral claw cushion of the left hindfoot of the animal from the group 5 (5E) are seen. In contrast to a definite decrease in the adipose tissue rate, the connective tissue gradually increased from the group 1 to the group 5. Crossmon’s trichrome. Magnification bar: 100μm.
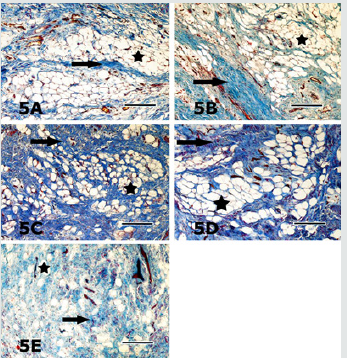
Figure 6: Sections of the digital cushion stained with AB pH 2.5 from the medial claw of the left forefoot of the animal from the group 1 (6A), the lateral claw of the right forefoot of the animal from the group 2 (6B), the lateral claw of the right hindfoot of the animal from the group 3 (6C) are seen. Staining intensity of the ground substance (arrows) in different sections is quite similar. Asterisks depict the adipocytes. AB pH 2.5 stain. Magnification bar: 100μm.
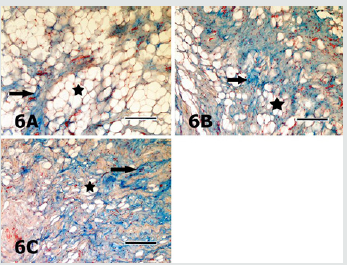
Figure 7: Sections of the digital cushion stained with AB pH 4 from the medial claw of the left hindfoot of the animal from the group 1 (7A), the medial claw of the left hindfoot of the animal from the group 4 (7B), the medial claw of the left hindfoot of the animal from the group 4 (7C) and the medial claw of the right hindfoot of the animal from the group 5 (7D) are seen. Slight differences in staining intensities of the ground substance (arrows) in different sections are seen. Asterisks depict the adipocytes. AB pH 4 stain. Magnification bar: 100μm.
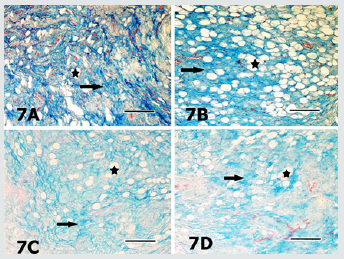
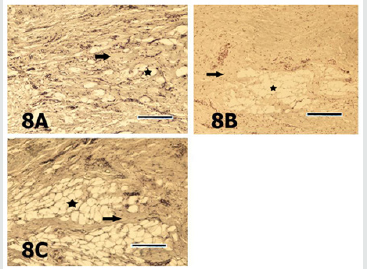
Figure 8: Sections of the digital cushion stained with safranin O from the lateral claw of the left hindfoot of the animal from the group 1 (8A), the lateral claw of the left hind foot of the animal from the group 2 (8B) and the lateral claw of the right fore foot of the animal from the group 3 (8C) are seen. Staining intensities of the ground substance (arrows) in different sections are quite similar. Asterisk depicts the adipocytes. SO stain. Magnification bar: 100μm.
The heifers had significantly (P<0.001) higher type I collagen fibre percentage (27.82%) than those of the other groups those had quite similar (P>0.05) percentages (Figure 3). In AB pH 2.5 stained specimens, the AB positivity was mainly located in the ground substance around the connective tissue fibres, embedded in-between the adipocyte groups (Figures 6A-6C). The positivity was also stronger around the individual adipocytes and the groups constituted of small numbers of these cells. Percentage of the AB pH 2.5 stained ground substance was highest (2.08%) in the group 3 and group 2 followed it (1.96%), whereas the groups 1, 4 and 5 had quite similar (P>0.05) and significantly (P<0.01) lower values than those of the groups 2 and 3 (Figure 3). AB pH 4.0 mainly stained the similar ground substance regions with AB pH 2.5, around the connective tissue fibres, located around the adipocyte groups (Figures 7A-7D). Results of the histometrical investigations of the AB pH 4.0 stained specimens showed that the group 1 and group 4 had similar (P>0.05), and significantly higher (P<0.01) positivity percentages than those of the groups 2, 3 and 5 (Figure 3). In SO stained specimens, the positivity was very weak (Figures 8A-8C) in all groups. The positivity percentages of the groups were quite similar and there were no significant differences (P>0.05) between the groups (Figure 3). Elastic fibres were mainly found in-between the connective tissue trabecules surrounding adipocyte groups and tunica adventitia of the blood vessels, and seen as brownishblack, branching coarse and gently undulating fine fibres, in the sections stained with Verhoeff’s elastic fibre stain (Figures 9A- 9E). The results of digital image analyses showed that the group 2 had the highest (1.89%) elastic fibre percentage and the value was significantly (P<0.05) higher than those of the other groups. The groups 1and 3, and 4 and 5 had quite similar elastic fibre percentages (P>0.05, Figure 3).
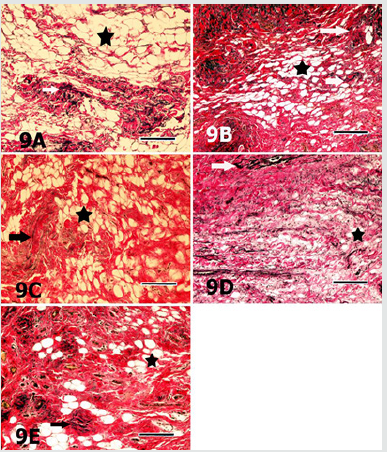
Figure 9: Sections of the digital cushion stained with Verhoef’s elastic fibre stain from the medial claw of the right hindfoot of the animal from the group 1 (9A), the medial claw of the left hindfoot of the animal from the group 2 (9B), the lateral claw of left forefoot of the animal from the group 3 (9C), medial claw of the left hind foot of the animal from the group 4 (9D) and the lateral claw of the left hind foot of the animal from the group 5 (9E) are seen. Elastic fibres (arrows) are mainly located in connective tissue trabecules surrounding the adipocyte groups (asterisks). Verhoef’s elastic fibre stain. Magnification bar: 100μm
Discussion
In the present study, statistically significant (P<0.01) differences were found between the CHL frequencies of the groups. Particularly, high incidences of CHL in the primiparous cows (45.80%, group 2) and multiparous (56.20%) cows (group 5) were striking. The differences possibly have arisen from higher incidence of CHL in more than one or all foot of the animals in these groups. Previous researchers [27,28] have reported that the histological development and functional maturation of the digital cushion are not completed before the age of three, and also incidence of sole lesions, such as sole ulcerations and white line disease are relatively higher in the first lactation because that the cushion tissues is less elastic and rich in saturated fatty acids. In accordance with the previous researchers [27,28] Incidence of CHL was higher in primiparous cows in the present study. The roles of the additional loads and forces exposed at late pregnancy, and structural and biochemical changes in the regional tissues, in addition to trauma of both corium of sole and the cushion tissues in the pathogenesis and occurrence of CHL have been discussed in detail by previous researchers [17-19,29,30]. Moreover, collaborative effects of improper, inadequate feeding and housing practices are mentioned among significant disposing factors. In coordination of anatomic structures of the hoof [bones, joints, ligaments and bursae] leads to CHL, mainly sole ulcers and white line disease. Thus, hoof problems occur more commonly among the cows kept in cubicles or concrete yards or straw yards than in the cows kept in cowsheds. Previous researchers [18,31,32] have reported that lameness incidence increases in multiparous cows with increase of ageing and calving, especially after third lactation, due to increasing the amount of the loose connective tissue while reducing adipose tissue, decreasing body condition score [BCS], thinning of the cushion segments. In the present study, a gradually decline was found in adipose tissue percentages of the groups, with ageing and increasing parturitions. Loosening of suspensor ligament system of the hoof results in increasing in mobility of bones, and this situation disposes the sole corium and sole horn to traumatic lesions, and consequently plays significant roles in the development of CHL, such as sole ulcers, white line disease [19,33]. Loosening of suspensor system of the hoof also increases the sensitivity of the claw to the CHL depending on increase of loads applied to the foot by increase in mammary gland and foetus weights, especially during first lactation [34]. Clinical findings and histometrical results of the hooves and higher CHL incidence of the groups 2 and 5 are consistent with previous findings [18,31,32]. Moreover, higher CHL incidence of the group 5, in which significant shrinkage of the cushion tissue was observed, supports the hypothesis suggesting the decline in load absorbing capacity of the cushion increases risk of injury of sole corium. Thus, environmental factors, such as long time standing on the concrete floor, unsuitable housing conditions and in proper claw trimming are effective in embodiment of the sole trauma and haemorrhages in addition to increase of coffin bone motility in late pregnancy and parturition. Unsuitable housing and hygiene conditions in the animals of the present study were in accordance with the high CHL incidence of the animals.
The digital cushion, which is a specified connective tissue, is constituted of connective tissue cells and intercellular substance [ICS]. The ICS contains collagenous, elastic and reticular fibres embedded in an amorphous ground substance. The amount of the amorphous ground substance in young animals is higher than fibrous elements. With ageing, the amount and rate of fibrous elements increase and the rate reverses in older animals. The digital cushion, a member of supporting system of the ruminant hoof locating in subcutis, is a complex structure, composed of white and yellow adipose tissues. There are significant differences in histological structure and organization of the digital cushion among the ungulate animal species. The digital cushion of the ruminant hoof is closely resembled with the cushion of the elephant’s cushion [18]. Equine digital cushion has relatively different structural features. In the horse, the cushion is composed of small number of elastic fibres and closely packaged interlacing collagenous fibres dispersed in acidic mucous matrix, rich in hyaluronic acid. Large spaces between the cells and fibres occupied with mixoid tissue and islets of fibro cartilage tissue [35,36]. Collagen fibres, especially type I fibres are the dominant fibre type of the connective tissues. Although the collagenous fibres have limited elasticity, their tensile strength is enough to meet the high tensile forces of the foot. In the present study, axial pad of the digital cushion constituted of white, unilocular adipose tissue lobuli surrounded by connective tissue trabecules. Fibro cartilage was not observed in the specimens. In some of the groups, trabecules surrounding adipose tissue lobuli constituted of loose connective tissue and contained less amount of type I collagen fibres, the others were rich in the collagenous fibres. Group 1 contained significantly (p<0.001) higher collagen (27.82%) when compared with the other groups. A gradual decline of type I collagenous fibre percentage was typical in the remaining, older and multiparous groups (groups 3, 4 and 5). Nevertheless, they contained quite similar (P>0.05) type I collagen fibre rates. Gradual reduction of type I collagenous fibre rate with the increase of age and calving might arisen from the structural and functional changes occurred in aged animals and during the late pregnancy and post parturition. It might be predicted that shock absorbing capacity of the digital cushion considerably reduced with declining of collagen fibre rate. In accordance with this prediction, the highest (56.20%) CHL incidence was found in the group 5, which had relatively low rate of type I collagen fibres. Moreover, in accordance with the prediction above, previous researchers [19,33,37], also suggested that loosening of the suspensor system of the hoof during pregnancy and post parturition increases sensitivity of sole corium located between coffin bone and sole to trauma, and consequently playing significant roles in the pathogenesis of the CHL, such as sole ulcers and white line disease.
Although the information on the types and distribution of adipocytes in ruminant digital cushion is limited, the heifer cushion contains lesser amount of the fat cells. The digital cushion of the heifers is a whitish and gelatinous in nature containing high amounts of ground substance and lipid content gradually increases through 2-3 parturitions, and gradually decrease starting from 3 years of age [18,21,32]. Because that white fat tissue is an energy source, many metabolic and hormonal mechanisms, such as pancreatic hormones and glucocorticoids are efficient on this fat tissue. Thus, feeding and other metabolic factors are likely to efficient on both amount and composition of fat of the digital cushion. Moreover, it is well known that the fatty acid composition and amounts of the cow’s digital cushion are seriously affected by metabolic disorders, mainly by ketosis. Similarly, previous researchers [38] emphasized that the topic needs detailed evidences. In the presented study, the digital cushion of the dairy cattle comprised of islets of large unilocular adipocytes. In the digital cushion with low levels of fat tissue, individual adipocytes were frequently seen. The animals in the group 1 contained significantly (P<0.001) higher adipocyte rates (%21.55) and the group 2 it, with a significant decline. However, the group 2 contained significantly (P<0.05) higher fat tissue percentage than those of the groups 3, 4 and 5. Rastani [39] suggested that lipids are mobilized from fat tissues in order to be used in milk synthesis. Lipids in white fat depots in the digital cushion are also consumed. Considering the data obtained in this study, it is possible to assume that white adipose tissue in the ruminant digital cushion also can be used as an energy source in ketosis, which is a condition resulting in negative energy balance [NEB], and thus CHL increase post parturition period. It is well known that fat and elastic fibres of the digital cushion play significant roles in shock absorbing mechanism of the cushion via expanding sideways until their viscoelasticity is limited by stretched fibrous connective tissue septa rich in collagen fibres. Elastic fibres support expansion by flattening their undulations and support reversing to the situation before when the applied force is expired. Significant decreases in both elastic fibre and adipose tissue rates result in loss of elasticity of the digital cushion.
There is limited information on the histochemical properties of ground substance of the cow’s digital cushion, and also histometrical results evaluation of these parameters have not been documented previously. In the present study, the results of the AB pH 2.5, AB pH 4.0 and SO stains showed that ground substance of the cow’s digital cushion was rich in hyaluronic acid (hyaluronan) and other proteoglycans, although there were some differences between the groups. Because that hyaluronic acid is a macromolecule increasing the viscosity of the tissue fluid, it consequently augments resilience of the tissue by binding water molecules. Other proteoglycans stained with SO might support the digital cushion in resisting to pressure, since the proteoglycans also show high water binding affinity and tend to gelation [40]. Although the results of the present study indicated that the ground substance of cow’s digital cushion was rich in hyaluronic acid, the hoof material was obtained from the animals with varying degree of CHL lesions. The authors strongly stress that a detailed comparison should be carried out between the findings of healthy and CHL animals. The elastic fibres, which relatively thinner when compared to the collagen fibres, are able to compensate the forces applied between elasticity limits. Tissues gain elasticity and stress bearing features by their elastic fibre content. Elastic fibres are found in hoof soft tissues as in other elastic tissues. Information on distribution and localization of these fibres in the ruminant hoof soft tissues is insufficient. In a previous histological study [38], elastic fibres were found between the collagen fibre bundles in the digital cushion of African elephant. In this study, elastic fibres were more peculiarly observed in-between the collagenous fibre bundles and in blood vessel walls. Morphometrical analyses evidenced that the group 2 displayed significantly (P<0.05) higher elastic fibre percentage (1.89%) than the other groups. Although there was a declining tendency with increase of the parturition number, the other groups had relatively similar elastic fibre ratios. Besides, striking decline of the elastic fibre percentage was determined in the groups 4 and 5 in concomitant with higher CHL lesion incidence. Although further studies are needed, the results of the present study imply that elastic fibre content of the cow digital cushion is quite limited and might play only small role in the functions of the digital cushion and in the development CHL lesions.
Conclusion
Based on the results, it was concluded that CHL seriously affected both histology and functional efficiency of the digital cushion of dairy cows. Results of the present study, which show strong evidence between histological, histochemical and histometrical findings and CHL lesions are noteworthy. However, the results should be compared with those of the healthy animals in further experiments those are involving experimental models. Further studies necessary to understand the nature of the events playing roles involving animal feeding in the development of the lesions, to develop new animal feeding strategies aiming to protect and maintain the normal structure of the digital cushion, in order to maintain and augment its supportive and protective roles, to reduce incidence of laminitis.
Grant Support
This work was supported by a grant from The Scientific and Technological Research Council of Turkey with project no: TOVAG 112O332.
References
- Green LE, Hedges VJ, Schukken YH, Blowey RW, Packington AJ (2002) The impact of clinical lameness on the milk yield of dairy cows. Journal of Dairy Science 85: 2250-2256.
- Amory JR, Kloosterman P, Barker ZE, Wright JL, Blowey R W, et al. (2006) Risk factors for reduced locomotion in dairy cattle on nineteen farms in the Netherlands. Journal of Dairy Science 89(5): 1509-1515.
- Amory JR, Barker ZE, Wright JL, Mason SA, Blowey RW, et al. (2008) Associations between sole ulcer, white line disease and digital dermatitis and the milk of 1824 dairy cows on 30 dairy cow farms in England and Wales. Preventive Veterinary Medicine 83(3-4): 381-391.
- Archer SC, Green MJ, Huxley JN (2009) The dynamics of dairy herd mobility. In: Proceedings of the Cattle Lameness Conference, University of Nottingham, England, pp 49-50.
- Archer SC, Green MJ, Huxley JN (2010) The association between milk yield and seriallocomotion score assessments in UK dairy cows. Journal of Dairy Science 93(9): 4045-4053.
- Garbarino EJ, Hernandez JA, Shearer JK, Risco CA, Thatcher WW (2004) Effect of lameness on ovarian activity in postpartum Holstein cows. Journal of Dairy Science 87(12): 4123-4131.
- Hultgren J, Manske T, Bergsten C (2004) Associations of sole ulcer at claw trimming withreproduction and performance, udder health, milk yield and culling Swedish dairy cattle.Preventive Veterinary Medicine 62(4): 233-251.
- Melendez P, Bartolome J, Archbald LF, Donovan A (2003) The association betweenlameness, ovarian cysts and fertility in lactating dairy cows. Theriogenology 59(3-4): 927-937.
- O’Callaghan KA, Cripps PJ, Downham DY, Murray RD (2003) Subjective and objective assessment of pain and discomfort due to lameness in dairy cattle. Animal Welfare 12(4): 605-610.
- Bach A, Binares M, Devant M, Carre X (2007) Associations between lameness and production, feeding and milking attendance of Holstein cows milked with an automatic milking system. Journal of Dairy Research 74(1): 40-46.
- Cook NB, Bennett TB, Nordlund KV (2004) Effect of free stall surface on daily activity patterns in dairy cows with relevance to lameness prevalence. Journal of Dairy Science 87(9): 2912-2922.
- Bicalho RC, Vokey F, Erb HN, Guard CL (2007) Visual locomotion scoring in the firstseventy days in milk: impact on pregnancy and survival. Journal of Dairy Science 90(10): 4586-4591.
- Bicalho RC, Cheong SH, Cramer G, Guard CL (2007) Association between a visual and an automated locomotion score in lactating Holstein cows. Journal of Dairy Science 90(7): 3294-3300.
- Dippel S, Dolezal M, Brenninkmeyer C, Brinkmann J, March S, et al. (2009) Risk factors for lameness in freestall housed dairy cows across two breeds, farming systems, and countries. Journal of Dairy Science 92(11): 5476-5486.
- Ito K, von Keyserlingk MAG, LeBlanc SJ, Weary DM (2010) Laying behaviour as an indicator of lameness in dairy cows. Journal of Dairy Science 93(8): 3553-3560.
- Chapinal N, Barrientos AK, Von Keyserlingk MAG, Galo E, Weary DM (2013) Herd-level risk factors for lameness in free stall farms in the north-eastern United States and California. Journal of Dairy Science 96(1): 318-328.
- Räber M, Letter V, Chassot A, Geyer H, Scheeder MRL (2015) The influence of feeding regimen on the composition of the fat pads in the bovine digital cushion. Agricultural Sciences 6: 889-899.
- Räber M, Lischer CH, Geyer H, Ossent P (2004) The bovine digital cushion-a description and anatomical study.The Veterinary Journal 167(3): 258-264.
- Tarlton JF, Holah DE, Evans KM, Jones S, Pearson GR, et al. (2002) Biomechanical and histopathological changes in the support structures of bovine hooves around the time of first calving. The Veterinary Journal 163(2): 196-204.
- Ossent P, Greenbush PR, Vermunt JJ (1997) Laminitis. In: Lameness in cattle. GreenoughPR & WeaverA.D. eds. Saunders Company USA, pp. 277-292.
- Räber M, Scheeder MRL, Ossent P, Lischer CH, Geyer H (2006) The content andcomposition of lipids in the digital cushion of the bovine claw with respect to age and location -A preliminary report. The Veterinary Journal (1): 173-177.
- Bicalho RC, Machadon VS, Caixeta L (2009) Lameness in dairy cattle: A debilitating disease or a disease of debilitated cattle? A cross-sectional study of lameness prevalence and thickness of the digital cushion. Journal of Dairy Science 92(7): 3175-3184.
- Logue DN, Offer JE, McGoandrn RD (2004) The bovine digital cushion-how crucial is itto contusions on the bearing surface of the claw of the cow? The Veterinary Journal 167(3): 220-221.
- Van Amstel SR, Shearer J (2006) Manual for treatment and control of lameness in cattle. Blackwell Publishing, USA, Ames, p. 22-30.
- Culling CFA, Allison RT, Barr WT, Bradbury P, Gordon, KC (1992) Connective tissues and stains. In: Theory and practice of histological techniques. Bancroft JD, Stevens A eds. (3rdedn). Churchill Livingstone UK, pp. 119-141.
- Bradbury P, Gordon K (1977) Connective tissues and stains. In: Bancroft DJ, Stevens A, eds. Theory and Practice of Histological Techniques. Edinburgh: Churchill Livingstone, pp. 95-112.
- Webster AJF (2001) Effects of housing and two forage diets on the development of claw horn lesions in dairy cows at first calving and in first lactation. The Veterinary Journal 162(1): 56-65.
- Maierl J, Böhmisch R, Dickomeit M, Liebich HG (2002) A method of biomechanicaltesting the suspensory apparatus of the third phalanx in cattle: a technical note. AnatomiaHistologiaEmbryologia31(6): 321-325.
- Thoefner B, Pollitt CC, Van Eps AW, Milinovich GJ, Trott DJ, et al. (2004) Acute bovine laminitis: A new induction model using alimentary oligofructose overload. Journal of Dairy Science 87(9): 2932-2940.
- Vermunt JJ (2007) One step closer to unravelling the pathophysiology of claw horn disruption: For the sake of the cows’ welfare. The Veterinary Journal 174(2): 219-220.
- Lischer CH, Ossent P, Raber M, Geyer H (2002) Suspensory structures and supporting tissues of the third phalanx of cows and their relevance to the development of the suspensory structures and supporting tissues of the third phalanx of cows and their relevance to the development of typical sole ulcers (Rusterholz ulcer). Veterinary Record 151(23): 694-698.
- Räber M, Scheeder MRL, Geyer H (2002) The influence of load and age on the fat content and the fatty acid profile of the bovine digital cushion In: Proceedings of the 12th International Symposium on Lameness in Ruminants. Orlando, Florida, pp. 194-198.
- Ossent P (1999) Subclinical bovine laminitis. Cattle Practice (7): 193-195.
- Alsleben B, Russke A, Wrede J, Hamann H, Distle O (2002) Messung der druckandrteilungunter den klauenbeirindern der rassedeutschesbraunvien in den erstenzweilebensjahren. TierӓrztlicheUmschau (57): 657-666.
- König HE, Macher R, Polsterer-Heindl E (2003) StoßbrechendeEinrichtungen am Zehenendorgan des Pferdes. Wiener TierärztlicheMonatsschrif (90): 267-273.
- Egerbacher M, Helmreich M, Probst A, König H, Böck P (2005) Digital cushions in horses comprise coarse connective tissue, myxoid tissue, and cartilage but only little unilocular fat tissue. AnatomiaHistologiaEmbryologia34(2): 112-116.
- Leach KA, Logue DN, Kempson SA, Offer JE, Ternent HE, et al. (1997) Claw lesions in dairy cattle: development of sole and white line haemorrhages during the first lactation. The Veterinary Journal 154(3): 215-225.
- Weissengruber GE, Egger GF, Hutchinson JR, Groenewald HB, Elsässer L, et al. (2006) The structure of the cushions in the feet of African elephants (Loxodonta africana). Journal of Anatomy 209(6): 781-792.
- Rastani RR, Andrew SM, Zinn SA, Sniffen CJ (2001) Body composition and estimated tissue energy balance in Jersey and Holstein cows during early lactation. Journal of Dairy Science 84(5): 1201-1209.
- Lodish H, Berk A, Matsudaira P Matsudaira P, Kaiser CA, et al. (2004) Biomembranes and cell architecture. In: Molecular Cell Biology. (5thedn). Freeman USA, New York, pp.147-196.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...