Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2690-5760
Research ArticleOpen Access 
Stem Cells Therapeutics for Infertility: The Future Perspectives in African Countries Volume 1 - Issue 2
Dalhatu A1*, Umar LB1, Alhasan AW3, Danlami S2 Hadad MM1, Rabi MR1, Yunusa U1 and Jari S4
- 1Department of Nursing Sciences, Bayero University, Kano, Nigeria
- 2College of Nursing and Midwifery, Kadun, Nigeria
- 3Department of Medicine, Ahmadu Bello University, Zaria, Nigeria
- 4College of Nursing and Midwifery, Katsina, Nigeria
Received: December 14, 2019 Published: January 10, 2020
Corresponding author: Adamu Dalhatu, College of Health Sciences, Bayero University, Kano, Nigeria
Abstract
Background: Globally infertility is evidently recognized as of public health concern especially in African countries. In African societies human reproduction is highly valued and inability to conceive is considered a personal tragedy and a curse for couples, impacting on the entire family and local community. Stem cells are the new technique in this era against many diseases and disorders.
Aim: The paper reviewed scientific related literature on the effectiveness of stem cells therapeutics in the management of infertility and its associated challenges especially in African society.
Methods: Various studies employed nonrandomized clinical trials on evaluating the effectiveness of stem cells therapy in the management of infertility and its associated challenges.
Results: The effectiveness of stem cell therapy for infertility management is becoming more important and some stem cells have been proven to have the potential for improving the damaged ovarian tissue in vitro.
Conclusion: The paper concludes that management of infertility with stem cells transplantation remains a challenge for greater clinical application especially in African countries. Recommendation: It is therefore, recommended that Stem cells therapy requires a great deal of research to treat infertility and this may be impeded by ethical issues in African countries because the use of human pluripotent stem cell is driven from human embryo or cadaveric fetal tissue.
Keywords: Stem Cells; Infertility; Therapy; Africa
Introduction
Infertility is a major problem globally affecting 8-12% couples. Studies have shown that the population of the world had been on the increase, evident by high fertility trends. The joy of every marriage is for the couple to procreate and raise children of their own. Globally, infertility affects at least about one couple in six, and affecting the developing countries than the developed ones [1,2]. In Sub-Saharan Africa, the prevalence of infertility is as high as 30% and the male contribution in most countries including Nigeria had been variously estimated to be between 30 and 50% [1].
In Nigeria, it has been described as an important reproductive health concern of women and a common reason for gynecological clinic consultations. It is a common knowledge that having children is a social obligation, parenthood is culturally mandatory, childlessness is socially unacceptable, and childlessness stigmatized; all due to a high societal value placed on children [1]. It has been hypothesized that female infertility produces social consequences for African women and these consequences are particularly profound for women as compared to men, regardless of the causes of infertility [1].
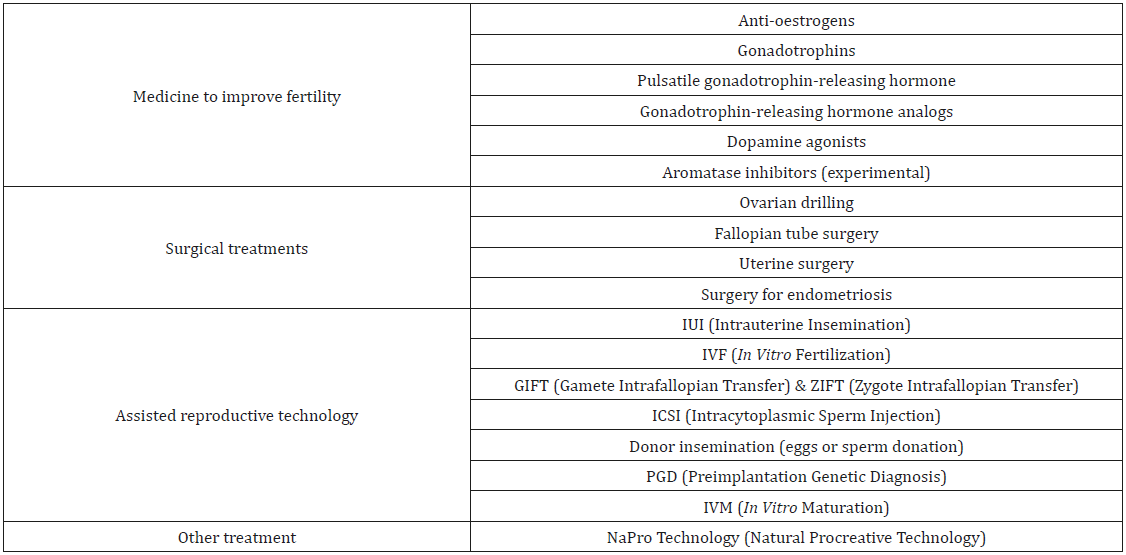
There are limited treatment options currently available for infertile couples. Individuals affected with infertility are wary of choosing adoption as a way of resolving infertility because of cultural factors and the non-specific provisions for adoption in the Nigerian legal system. This quest of infertile couples to resolve the problem of infertility has resulted in the patronage of various treatment centers with different treatment options [1].
The prevalence of primary and secondary infertility in the world range from 5-30% with variations from country to country [3]. Reports have shown that, secondary infertility in developing countries increases exponentially with age, from about 5% at ages 20-24 to about 62% at ages 45-49 years. In Sub-Saharan Africa, infertility rates are as high as 30%. This high infertility rate is due to infection, either from sexually transmitted infections, after child birth or abortion.
In African societies human reproduction is highly valued and inability to conceive is considered a personal tragedy and a curse for couples, impacting on the entire family and local community. In addition, childless women are frequently stigmatized resulting in isolation, neglect, domestic violence and polygamy [4,5].
In Kenya, infertility is a major reproductive health concern as reflected in a national infertility survey. The survey highlighted infertility related consultations to be as follows; at teaching and referral (tertiary) hospitals 30%, in provincial hospitals 27%, in district hospitals 15%, in health centers 4% and in dispensaries to be 2% [4]. Earlier infertility studies conducted in Kenya had almost similar findings. Infertility in Kenya is a significant family problem in all communities across the country, most people interviewed during the 2005/6 infertility survey cited infertility as woman problem since its’ difficult to involve the men in fertility assessment.
The National reproductive health policy (2007) recognizes infertility as an important public health concern in African countries. It prioritizes the following actions; improving access to quality infertility services to all levels, promoting community awareness especially among men and encouraging all aspects of research. The pattern of infertility varies from population to population and is influenced by social cultural differences, level of sexual promiscuity, prevalence of sexually transmitted diseases and reproductive behavior. There is also evidence that a woman with high risk sexual history, multiple marital unions, and sexual initiation before puberty has a higher risk for infertility [3,6].
The National reproductive health policy (2007) recognizes infertility as an important public health concern in Nigeria. It prioritizes the following actions; improving access to quality infertility services to all levels, promoting community awareness especially among men and encouraging all aspects of research.
The pattern of infertility varies from population to population and is influenced by social cultural differences, level of sexual promiscuity, prevalence of sexually transmitted diseases and reproductive behavior. There is also evidence that a woman with high risk sexual history, multiple marital unions, and sexual initiation before puberty has a higher risk for infertility [7,8].
Causes of Infertility
Reports have shown that genetic background of infertility in Nigeria have indicated that about 39.5% of the normozoospermic population has reduced CAG repeats and a lower percentage of the infertile population (23%) has shown moderately increased repeats [9]. This information have serious implications for making decisions on treatment strategy, especially hormonal replacement therapy in hypogonadism. Individuals affected by some forms of genetic alterations produce a higher frequency of sperm with aneuploidies; hence, an increased risk of congenital abnormality in the fetus exists as produced by in vivo fertilization or intracytoplasmic sperm injection (IVF/ICSI) [10]. There is increasing evidence that genetic conditions may also be transmitted to the offspring, thereby creating transgenerational infertility or other health problems. The increase rate of infertility in sub-Saharan Africa may be unfamiliar to foreigners, as they are accustomed to reports of ubiquitous poverty and high rates of morbidity and mortality from infectious and parasitic disease [11]. Evidently there is also growing prevalence of chronic degenerative diseases, and this compounded by severe malnutrition and basic impediments due to inferior environmental quality.
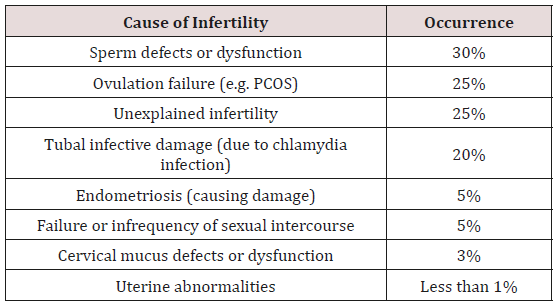
The causes of infertility are widely classified in four major categories: the female factor; the male factor; combined factors; and unexplained infertility. It is generally reported that in approximately 35% of cases, infertility is mainly due to a female factor, in 30% to a male factor, in 20% to abnormalities detected in both partners, and in 15% of cases no diagnosis can be made after a complete investigation. A report by World Health Organization on infertility shows female factor infertility to be due to the following; Ovulatory disorders 25%, endometriosis 15%, pelvic adhesions 12%, tubal blockage 11%, other tubal abnormalities 11% and hyperprolactinemia 7%. Locally a study by Gachara & Manina found that 92% of women and 76% of men among infertile couples were between the ages 21-35 years at GNH, amongst these 35% had primary infertility while 65% had secondary infertility. Mati & Mulani found that in 61.9% of couples in Ghana with infertility, the cause of infertility could be attributed to female factor, 20.1% to male factor and 18% to both male and female. Minor degrees of fertility impairment are not necessarily associated with couple infertility when present in only one partner but may reduce the couple’s fertility when present in both partners. In the US, 15- 30% of infertile couples have unexplained infertility [4]. Ovulatory disorders can be as a result of infrequent ovulation or absent ovulation. The World Health Organization has classified anovulation into three main groups and recognizes hyperprolactinemia as an additional etiology. WHO class 1 is defined in women who have low or low- normal serum follicle stimulating hormone (FSH) concentration and low serum estradiol concentrations due to decreased hypothalamic secretion of gonadotropin -releasing hormone (GnRH) or pituitary unresponsiveness to GnRH. WHO class 2 is normogodotropicnormoestrogenic anovulation, while class 3 is hyper gonadotropichypo estrogenic ovulation. This system is useful for defining and treating anovulatory disorders according to the underlying endocrine dysfunction [12,13].
Age is also an important characteristic affecting a woman’s fertility. The decrease in fecundability with aging is likely due to a decline in both the quantity and quality of the oocytes. The germ cell complement of the ovary reaches its apex of 6 to 7 million follicles in the mid gestation female fetus, followed by a steady attrition from 1 to 2 million follicles at birth to 300,000 follicles at the onset of puberty. This rate of follicle loss accelerates after the woman reaches her mid-thirties [12,13].
Patent fallopian tubes, functional uterus and cervix are also essential for conception to take place. Tubal disease and pelvic adhesions can lead to infertility by preventing normal transport of the oocyte and sperm through the fallopian tube. The primary cause of tubal factor infertility is pelvic inflammatory disease caused by pathogens such as chlamydia or gonorrhea. Other conditions that may interfere with tubal transport include severe endometriosis, adhesions from previous surgery or nontubal infection (e.g. appendicitis, inflammatory bowel disease), and pelvic tuberculosis. Uterine abnormalities due to fibroid or anomalies can cause infertility by impaired implantation, either by mechanical or due to reduced endometrial receptivity. Müllerian anomalies are a significant cause of recurrent pregnancy loss, with the septate uterus associated with the poorest reproductive outcome. Other structural abnormalities associated with infertility include endometrial polyps, and synechiae from prior pregnancy-related curettage. However, data establishing a causal link between these uterine abnormalities and infertility are lacking [12,13].
Endometriosis is also thought to decrease infertility by anatomic distortion from pelvic adhesions, damage to ovarian tissue by endometrioma formation and surgical resection, and the production of substances such as cytokines and growth factors which impair the normal processes of ovulation, fertilization, and implantation [12,13].
Male factor infertility can be caused by numerous factors which can be categorized into pre-testicular, testicular and post- testicular.Pretesticular factors includes; Firstly, hypogonadotropichypogonadism which may be as a result of elevated prolactin, medications. illicit drugs like marijuana, pituitary damage and Kallmann syndrome; Secondly, hypergonadotropichypogonadism whose most common cause include Klinefelters syndrome, which alter spermatogenesis; and lastly, other genetic disorders of gonadotropin secretion like multiorgan genetic syndromes such as the Laurence-Moon-Biedl syndrome, Prader-Willi syndrome, Lowe oculo-cerebral syndrome, and familial cerebellar ataxia.
Testicular factors include the following; Firstly, Varicocele whose association with infertility is not clear-cut, as it is also observed in about 10% of normozoospermic men. Though, it has also been related to semen abnormalities, decreased testicular volume and decline in leydig cell function; Secondly, Cryptochidism which affects 1-3% of full term newborns with a higher prevalence in premature boys, is also associated with a 13% incidence of azoospermia if unilateral and up to 98% if bilateral and untreated; Thirdly, testicular cancer can lead to infertility by destroying and compressing the healthy testicular tissue and also from treatment for the testicular cancer. fourthly, environmental factors like, hyperthermia is considered a major contributor of infertility in men with varicocele and cryptochidism; Lastly, other causes of male infertility include, testicular injury by direct trauma or indirect through infections like mumps ,kartageners syndrome, antisperm antibodies due to trauma and infection ,DNA damage can be caused by radiation, toxins, genital tract infections, varicocele, advanced paternal age and testicular hyperthermia, sertoli cell only syndrome, Ionizing radiation, chemotherapy and tobacco.
Post testicular characteristics include; congenital bilateral absence of the vas deferens, Young’s syndrome, ejaculatory duct obstruction /seminal vesicle dysfunction commonly caused by cystic fibrosis spectrum disease, woffian or mullerian original cyst ,calcifications, tuberculosis, genitourinary infections, calculi and urinary instrumentation ,nerve injury ,medication,resection of the prostate causing retrograde ejaculation and lastly ,coital problems like erectile dysfunction and penile abnormalities such as hypospadias and chordee may interfering with semen deposition.
Causes of Female Infertility
According to the center for Disease Control (CDC), the causes of female infertility can be classified into three broad categories including:
a) Defective ovulation
b) Transport
c) Implantation
Defective ovulation occurs because of the following causes:
Endocrine disorders
The dysfunction of hypothalamus and pituitary gland can lead to an excess amount of prolactin, this may prevent ovulation. Moreover, other endocrine glands including adrenal and thyroid may also delay ovulation. When the corpus luteum, fails to produce enough progesterone required to thicken the uterine lining, the fertilized egg may not be able to implant, thus leading to infertility.
Physical disorders
Certain physical disorders such as obesity, anorexia nervosa, and excessive exercise may lead to overweight or malnutrition, and later the menstrual cycle, thus make the couple infertile.
Ovarian disorders
polycystic ovarian disease (PCO) can lead to infertility because of an increased amount of testosterone and LH and decrease uptake of glucose by muscle, fat and liver cells resulting in the production of large amount of insulin by the pancreas. Low FSH levels also hinder the production of eggs from the ovarian follicles, and lead to form fluid- filled ovarian cysts that eventually cover the whole ovaries and prevent conception.
Endometriosis
This refers to a condition in which sections of the uterine lining implant in the vagina, ovaries, oviducts or pelvis. These implants form a fluid –filled cysts that grow with each menstrual cycle, and eventually turn into blisters and scars then block the passage of egg and block pregnancy (Table 1).
Stem Cell
Stem cells are biological cells that can differentiate into other
types of cells and can divide to produce more of the same type of
stem cells. They are found in multicellular organisms. In mammals,
there are two broad types of stem cells: embryonic stem cells,
which are isolated from the inner cell mass of blastocysts, and adult
stem cells, which are found in various tissues. In adult organisms,
stem cells and progenitor cells act as a repair system for the body,
replenishing adult tissues. In a developing embryo, stem cells can
differentiate into all the specialized cells-ectoderm, endoderm and
mesoderm (see induced pluripotent stem cells)-but also maintain
the normal turnover of regenerative organs, such as blood, skin, or
intestinal tissues.
There are three known accessible sources of autologous adult
stem cells in humans:
a) Bone marrow, which requires extraction by harvesting,
that is, drilling into bone (typically the femur or iliac crest).
b) Adipose tissue (fat cells), which requires extraction by
liposuction.
c) Blood, which requires extraction through apheresis,
wherein blood is drawn from the donor (similar to a blood
donation) and passed through a machine that extracts the stem
cells and returns other portions of the blood to the donor.
Stem Cell History
Stem cells were first recognized experimentally in 1945 and the properties of stem cells have been discussed for more than 40years. In 1998, James Thomson and his group managed, for the first time, to isolate and culture human embryonic stem cells using blastocyst obtained (Table 2).
Stem Cell Therapy
The use of conventional treatment to treat infertility and many other disorders aren’t very effective, however, stem cell therapy has been showing a lot of promising results all over the world [14]. The treatment is safe, speedy and often completed within a day or two [15]. It was shown that, the results are far better than the long and ineffective conventional therapies. It is however noted that therapy using stem cells from various sources, including those from bone marrow and umbilical cord, has been successfully used to treat a number of life-threatening diseases with good results, since 1988[16]. History have shown that the past few years, with increased research and development activities, a growing number of successful stem cell treatments have emerged. It has become one of the most exciting area of medicine, encompassing all areas of current medical science [17].
Stem Cells Therapy in Infertility
Stem cells are the new devices in the period of technological advancement against many diseases and disorders [18]. Many researchers worldwide are working on the problem of infertility. Stem cells seem to be an important mode of therapy for this problem [19]. Infertility is the term labeled if a couple fails to achieve pregnancy even after 1 year of unprotected intercourse. Being infertile has major on the overall psychological as well as social profile of an individual. Broadly divided as ‘male factor’ and ‘female factor’ infertility has stem cell therapy as an emerging modality of treatment.
Evidently spermatogenesis has gone awry in male factor infertility. Spermatogenesis takes place in seminiferous tubules. Therefore, spermatgonial stem cells which are key to the sperm production, have an important role. Some structural and numerical chromosomal abnormalities along with Y-chromosome deletions, are associated with a severe male factor infertility [20]. In addition to this sometimes it has been noted that certain gonadotoxic drugs, radiations and chemotherapy as a mode of treatment for some other diseases interferes with spermatogenesis.
Stem cell-based strategies have also been tried for ovarian regeneration and oocyte production. Researchers have evidently proposed that stem cell-base therapy can be used as future clinical therapies for treating infertility in women. Some workers have demonstrated mitotically active germ cells in human ovaries that can be purified and cultured in vitro to spontaneously form oocytes [21]. This proves unique potential to generate oocytesin vitro from isolated cells in reproductive-aged women who may have a depleted follicle pool from such genetic defects.
Stem Cell Therapies for Infertile Women
Until recently, most of the major advances involving germ cell differentiation into haploid cells have been in male stem cells. To date, work in human pluripotent stem cells has generated haploid female cells but nothing that resembles an oocyte nor is predicated to possess a functional ooplasm capable of being fertilized [16]. However, the recent work by Hayashi et al. [22] showed that mouse stem cells could be differentiated in an in-vitro/in-vivo system into oocyte-like cells that are capable of being fertilized by spermatozoa and generating normal progeny. This outstanding advancement further shows the ability of pluripotent stem cells to differentiate into all cells of the adult organism. Whether the work by Hayashi and colleagues can be adapted for human stem cells remains to be seen, but this advancement is a critical step forward in generating functional de-novo oocytes from human iPSC from female patients rendered sterile by medical interventions, exposure to toxicants or by premature ovarian failure.
Challenges Related to Stem Cell Therapy in Africa
a) Lack of research about stem cell therapy
b) Challenge for clinical application.
c) Lack of promising healthcare policy
d) Lack of political will and professionalism
e) Educational drawbacks faced by African nations
f) Lack of biomedical researches
g) Lack of funding agencies
h) Ethical issues
Conclusion
Stem cells therapy in infertility provides a prosperous foundation to a long-lasting effect and when use provides promising results in most high income setting but this may not be feasible in most low income setting particularly in African society. Management of infertility with stem cells transplantation remains a challenge for greater clinical application especially in African countries.
References
- Olugbenga BA, Adebimpe WO, Olarewaju SO, Babatunde OA, Oke OS (2014) Prevalence of Infertility and Acceptability of Assisted Reproductive Technology among Women Attending Gynecology Clinics in Tertiary Institutions in Southwestern Nigeria. Gynecol Obstet (Sunnyvale) 4: 210.
- Blair RM, Jason CP, Darren KG, William BS, et al. (2017) Infertility diagnosis has a significant impact on the transcriptome of developing blastocysts. Molecular Human Reproduction, 23(8): 549-556.
- Ronald S Swerdloff, Christian W (2011) Causes of male infertility. Up to Date version 19. 3.
- Wiser Herbe J, Jay sandlow, Tobias S Kohler (2012) Causes of male infertility Male infertility p. 3-14
- White YA, Woods DC, Takai Y, Ishihara O, Seki H, et al. (2012) Oocyte formation by mitotically active germ cells purified from ovaries of reproductive-age women. Nat Med 18(3):413-21.
- Yamauchi K, Hasegawa K, Chuma S, Nakatsuji N, Suemori H (2009) In vitro germ cell differentiation from cynomolgus monkey embryonic stem cells. PLoS One 4: e5338.
- Zhang Z, Shao S, Meistrich ML (2007) The radiation-induced block in spermatogonial differentiation is due to damage to the somatic environment, not the germ cells. J Cell Physiol 211:149-58.
- Zhao Xy, Li W, Lv Z, Liu L, Tong M, et al. (2010) Viable fertile mice generated from fully pluripotent iPS cells derived from adult somatic cells. Stem Cell Rev 6: 390-397.
- Akinloye O, Grommoll J, Nieschlag E, Simoni M (2009) Androgen receptor gene CAG and GGN polymorphisms in infertile Nigerian men. J Endocrinol Invest 32(10): 797-804.
- Engel W, Adham IM, Nayernia K, Neesen J (2004) Genetic causes of male infertility. Verh Dtsch Ges Pathol 88: 130-135.
- Oluemi A, Ernest JT (2011) A review of management of infertility in Nigeria: Framing the ethics of a national health policy. Int J Womens Health 3: 265-275.
- WHO (2005) Health and millennium development goals. World Health Organization, Geneva, Switzerland.
- WHO (1991) Infertility: A Tabulation of Available Data on Prevalence of Primary and Secondary infertility. Programme on Maternal and Child Health and Family Planning, Division of Family Health, Geneva, Switzerland.
- Geijsen N, Horoschak M, Kim K, Gribnau J, Eggan K, et al. (2004) Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature 427: 148-1454.
- Eguizabal C, Montserrat N, Vassena R, Barragan M, Garreta E, et al. (2011) Complete meiosis from human induced pluripotent stem cells. Stem Cells 29 (8): 1186-1195.
- Hayashi K, Ogushi S, Kurimoto K, Shimamoto S, Ohta H, et al. (2012) Offspring from oocytes derived from in vitro primordial germ cell-like cells in mice. Science 338(6109): 971-975.
- Zegers-Hochschild F, Adamson GD, de Mouzon J, Ishihara O, Mansour R, et al. (2009) International Committee for Monitoring Assisted Reproductive Technology, and World Health Organization. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology. Fertil Steril 92(5): 1520-1524.
- Mulder CL, Zheng Y, Jan SZ, Struijk RB, Repping S (2016) Spermatogonial stem cell autotransplantation and germline genomic editing: A future cure for spermatogenic failure and prevention of transmission of genomic diseases. Hum Reprod Update 22(5): 561-573.
- Jahnukainen K, Ehmcke J, Hou M, Schlatt S (2011) Testicular function and fertility preservation in male cancer patients. Best Pract Res Clin Endocrinol Metab 25(2): 287-302.
- Van Voorhis Bj, Ryan Gl (2010) Ethical obligation for restricting the number of embryos transferred to women: Combating the multiple-birth epidemic from in vitro fertilization. Semin Reprod Med 28(4): 287-294.
- Volarevic V, Bojic S, Nurkovic J, Volarevic A, Ljujic B, et al. (2014) Stem cells as new agents for the treatment of infertility: Current and future perspectives and challenges. Biomed Res Int 2014: 507234.
- Hayashi K, Ohta H, Kurimoto K, Aramaki S, Saitou M (2011) Reconstitution of the Mouse Germ Cell Specification Pathway in Culture by Pluripotent Stem Cells. Cell 146(4): 1-14.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...