Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2638-5945
Case Report(ISSN: 2638-5945) 
Recurrent Ischemic Stroke as a First Manifestation of Poorly Differentiated Carcinoma (PDC) of Unknown Etiology Volume 3 - Issue 2
Dani Choufani1, Elena Schmidt2 and Julius Gene Latorre2*
- 1Department of Neurology, University of Mississippi Medical Center, Syracuse, NY, USA
- 2Neurocritical Care, Department of Neurology and Neurosurgery, SUNY Upstate Medical University Hospital, USA
Received:October 31, 2019 Published: November 06, 2019
Corresponding author: Julius Gene Latorre, Neurocritical Care Service, Department of Neurology, SUNY Upstate Medical University Hospital, 750 East Adams Street, Syracuse, NY, USA
DOI: 10.32474/OAJOM.2019.03.000160
Keywords:Paraneoplastic Syndrome; Acute Sschemic Stroke; Cryoptogenic Stroke; Embolic Stroke; Carcinoma
Introduction
The association between cancer and thrombosis has been known for a long time, and the majority of the thromboembolic events associated with cancer are usually diagnosed after the cancer being identified [1]. However, a thromboembolic event may precede the identification of a cancer and maybe the first manifestation of the underlying malignancy [2]. Malignant cancers promote hypercoagulable state and increases the risk of thromboembolism. Carcinoma is the most common cause of ischemic stroke associated with malignancy [3] with lung cancers being the most common [1,4] while adenocarcinoma confers a higher risk of recurrent venous thromboembolism (VTE) [5]. We report a case of recurrent embolic strokes resistant to anticoagulation as the first manifestation of poorly differentiated carcinoma (PDC) of unknown etiology. It is the first case reported in the literature.
Case Report
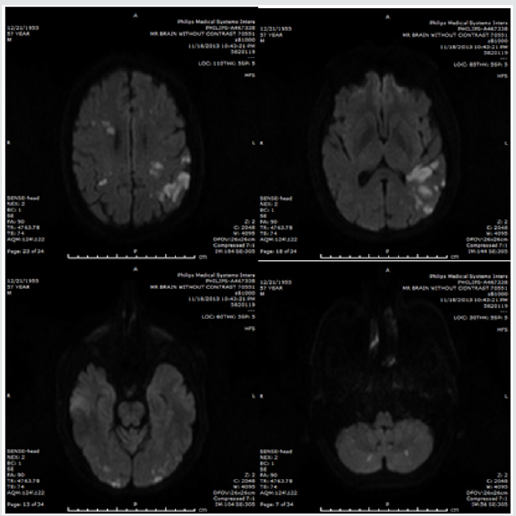
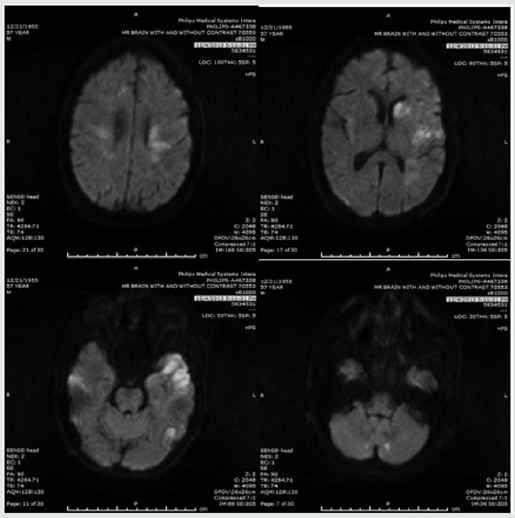
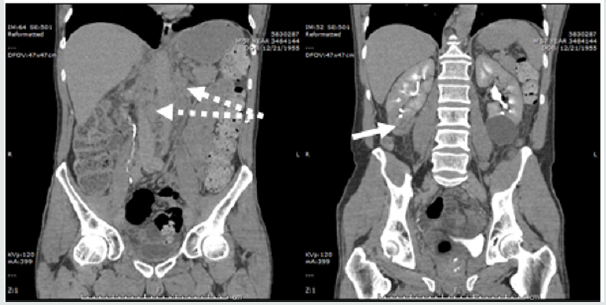
A 57 years old male patient presented to our hospital with nonfluent aphasia. There was no significant past medical history and he denied smoking, alcohol or illicit drug use. Clinical examination revealed a right inferior quadrantanopsia, a right upper extremity weakness with strength 4/5 according to Medical Research Council Scale. There were difficulties in word-finding, naming and reading with intact repetition. His admission National Institute of Health Stroke Scale (NIHSS) was 6. He was deemed ineligible for thrombolysis due to unclear onset time and was started on aspirin and high intensity statin. The CT angiography of the head and neck showed no evidence of aneurysm, dissection, significant flow stenosis, or arteriovenous malformations. A Transesophageal Echocardiography (TEE) showed normal left ventricular function with no evidence of a thrombus or a Patent Foramen Ovale (PFO). A Magnetic Resonance Imaging (MRI) of the brain showed acute infarcts within multiple vascular territories, involving the bilateral cerebellum, left frontal, temporal, parietal and occipital lobes, consistent with embolic infarcts (Figure1). A four-vessel digital subtraction cerebral angiogram did not show evidence of vasculitis or vessel malformation. A hypercoagulable panel did not reveal evidence of hematologic hypercoagulability. The patient was treated with warfarin with low molecular weight heparin bridging due to the highly embolic nature of the MRI findings, and was discharged to the acute inpatient rehabilitation facility. While at the rehab, the patient started complaining of back and abdominal pain. A CT abdomen and pelvis identified heterogeneously enhancing lesion within the lower pole of kidney with extensive lymphadenopathy (Figure 2). While waiting for biopsy to be done, the patient had worsening of aphasia and dysarthria prompting readmission. Repeat MRI brain showed new scattered areas of acite infarcts in different vascular territories (Figure 3). Anticoagulation was continued. Two days later, he had worsening mental status and repeat imaging showed more acute infarction. Biopsy of the abdominal lymph node was facilitated, and result showed poorly differentiated carcinoma of unknown origin. The family opted for comfort measures only and the patient expired shortly.
Figure 2: CT abdomen showing extensive mesenteric and retroperitoneal lymphadenopathy (dashed arrow) and heterogeneously enhancing R lower kidney mass (solid arrow). A benign left lower kidney cyst is also seen.
Discussion
Hypercoagulable state is a common finding in patients with malignancy due to the production of substances with procoagulant activity. Different mechanisms have been implicated in the cerebrovascular complications of cancer patients and includes the following: [6]
I. Cerebral infarction from nonbacterial thrombotic endocarditis (NBTE).
II. Thrombosis due to compression or infiltration of cerebral vessels by tumor or coagulopathy induced by chemotherapy.
III. Cerebral venous sinus thrombosis due to hypercoagulable state or chemotherapy.
IV. Cerebral infarction due to tumor emboli or septic thrombi.
V. Intracranial hemorrhage which is more common in patients with hematological malignancies.
Our patient presented with ischemic stokes involving multiple vascular territories. A complete workup for ischemic stroke was performed, including Brain MRI, TEE and cerebral angiogram that ruled out the presence of brain tumors, atherosclerosis, vasculitis, cardiac embolic source as well as sinus thrombosis. The results were in favor of embolic strokes. The physical examination did not reveal any signs of sepsis or infection. The conventional etiologies of stroke could not be identified. Despite appropriate therapeutic anticoagulation the patient had recurrent ischemic strokes. The commonest cause of stroke in cancer patients is embolic due to hypercoagulation [1,4]. Recent study showed that infarction in multiple vascular territories in stroke patients associated with cancer is significantly elevated (34%), compared to 15% of stroke patients not associated with cancer [5], so it is important to consider the existence of a concealed cancer in stroke patients with multiple vascular territories involvement and conventional mechanisms for stroke etiology not identified. A retrospective study showed that in patients with ischemic stroke associated with cancer, the cumulative rates of recurrent ischemic stroke were 7 % at 1 month, 15 % at 3 and 6 months [3]. In our case the patient experienced recurrent events within a two-week period and continued despite adequate anticoagulation. It is unclear whether initiation of aggressive cancer treatment may have improved patient outcome as the family opted for withdrawal of care. Further study on the early initiation of cancer therapy in malignancy-associated ischemic strokes even during the acute phase may be warranted.
References
- DM Cestari, DM Weine, KS Panageas, AZ Segal, LM DeAngelis (2004) Stroke in patients with cancer Incidence and etiology. Neurology 62: 2025-2030.
- HM Kwon (2007) Stroke as the first manifestation of concealed cancer. Journal of the Neurological Sciences 258: 80-83.
- Merkler Alexander, Singer Samuel, Cheng Natalie T, Stone Jacqueline B, Kamel Hooman, et al. (2013) Recurrent Thromboembolic Events After Ischemic Stroke in Patients with Cancer.[Abstract] International Stroke Conference Poster Abstracts 44(2).
- Christopher J Schwarzbach, Anke Schaefer, Anne Ebert, Valentin Held, Manuel Bolognese, et al. (2012) Stroke and Cancer: The Importance of Cancer-Associated Hypercoagulation as a Possible Stroke Etiology.Stroke 43(11): 3029-3034.
- Martha L Louzadaa, Habeeb Majeedb, Vi Daoc, Philip S, Wellsb D (2011) Risk of recurrent venous thromboembolism according to malignancy characteristics in patients with cancer-associated thrombosis: a systematic review of observational and intervention studies. Blood Coagulation and Fibrinolysis 22: 86-91.
- Rogers LR (2004) Cerebrovascular complications in patients with cancer. Semin Neurol 24: 453-460.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...