Lupine Publishers Group
Lupine Publishers
Menu
Review ArticleOpen Access 
Sustainable Desalination of Seawater via Reverse Osmosis using Polymeric Membranes Volume 1 - Issue 2
Hemangi Kabra1, Shubham Joshi1, Saurabh Nigade1, Sagar Chaudhari1 and Balasubramanian Kandasubramanian2*
- 1Department of Mechanical Engineering, Shrimati Kashibai Navale College of Engineering, India
- 2Nanosurface Texturing Laboratory, Department of Metallurgical & Materials Engineering, Defence Institute of Advanced Technology (DU), Ministry of Defence, India
Received: February 17,2020 Published: March 05, 2020
Corresponding author: Balasubramanian Kandasubramanian, Nanosurface Texturing Laboratory, Department of Metallurgical & Materials Engineering, Defence Institute of Advanced Technology (DU), Ministry of Defence, India
DOI: 10.32474/JBRS.2020.01.000108
Abstract
In today’s world water scarcity is a curse for humanity which can even be leading us to extension in future. Desalination is one of the solutions to nullify this problem up to some extent which is now-a-days a growing trend. Desalination professionally was implemented in the 1960’s with its first official plant in Freeport, Texas. Since then, much advancement was made in desalination. According to the recent market condition reverse osmosis is the known process for desalination. But lately, pervaporation has also grabbed attention owning to its characteristics of removal not only salt but also metal ions. Majorly, reverse osmosis employs cellulose acetate and polyvinylidene fluoride for membrane material. The gulf countries top the desalination market with 55% of global water desalination plants in world. Currently, the dependent world population for daily needs on desalinated water is 0.1%. Countries like Saudi Arabia, Kuwait, Iraq uses 100% desalinated water for their daily needs. As there are advantages there are some flaws too like hot brine management is the biggest problem faced by desalination today.
Keywords: Desalination; reverse osmosis; seawater; keratin; polymeric membrane
Introduction
In recent years, the massive issue of water scarcity led to
researchers’ focus on the furtherance of the desalination process.
The origin of desalination belongs to ancient Greece, where
Aristotle and other intellectuals performed distillation and
filtration using soil deposits [1]. Aristotle observed in his Metrology
that “saltwater, when evaporates, becomes sweet and when it
condenses again salt water is not formed [1]. Evaporation-based
desalination was introduced a long time ago and was commonly
used before the Second World War. The United States found the
saltwater office in the Department of the Interior in 1952, which
was later merged in 1974 with the water resource research office.
Desalination was developed at a large extension during the latter
half of the 20th century and continuously improving to date. The
foremost commercial desalination plant was built in the United
States at Freeport, Texas, in 1961, which produced 1 million gallons
per day of clean water with an infrastructure cost of $1.2 million
[2].
Later, in the late 1970’s reverse osmosis overtook thermal
techniques like Multi Flash Distillation. The exceptional mechanical
and chemical stability along with the low price of RO membranes,
making them favourable for industrial level use. Therefore, much
research was carried out with the RO membrane to increase its
permeability, which led to a decrease in the cost of production
about 10 times since 1978. The maximization of flux permeated
through membrane and energy recovery resulted in a reduction of
energy consumption, therefore, making RO membranes preferable
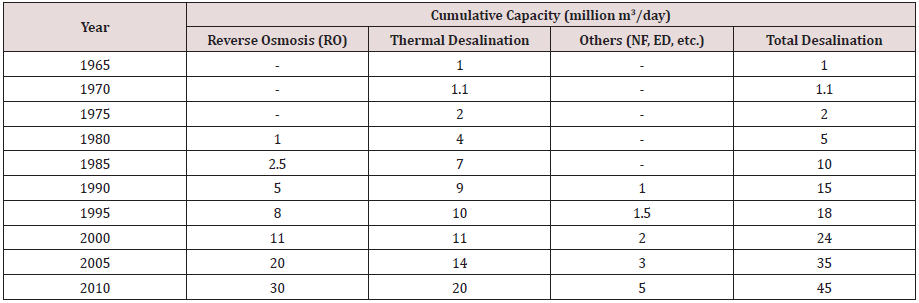
in terms of cost-cutting also [3] (Table 1).
Desalination rapidly increased worldwide from 1945 with 326 m3/d to 1980 with 50, 00,000 m3/day in 1980 and to moreover in 2004 it reached 35,000,000 m3/day. In 2008, 52,333,950 m3/d was the entire daily capacity, from approximately 14,000 plants functioning globally. Later, 67,000,000 m3/d was the whole capacity in 2011, but concerning 2012, from an extraordinary 16000 plants it was adjudged at approximately 79,000,000 m3/d [1]. The gulf countries top with a leading number of Desalination plants
Desalination is fundamentally classified into two disparate methods known as Multi-Stage Flash Distillation and Membrane Filtration, amid MSF is a thermal process in which we tend to seethe the water and then cool the vapour down to detach the salt. Despite that, in the membrane filtration, a reticulated structure is employed called a membrane through which water is swept with pressure beyond the reverse osmosis pressure which refines the water. Desalinated water can be utilized in tremendous applications such as human consumption, irrigation, marine applications like ships and submarines. A massive part of desalinated water focuses on economic the provision of potable water for human consumption along with conventional reserves.
Reverse osmosis membranes
Today, Thin Film Composite (TFC) polyamide membranes are leaders in the field of reverse osmosis which consist mainly of three films: a polyester band that acts as a structural reinforcement (120- 150μm), a micro-intermediate porous film (about 40μm) and an ultra-thin barrier (0.2μm) [4].
Fluoro-graphite modified PVDF membranes
Recently it is distinguished that the surface hydrophobicity influences membrane distillation of seawater. Recognizing this, Zhao et al., synthesized fluorographite which is extremely hydrophobic microcrystalline powder utilized in lubricants and lithium batteries with Polyvinylidene Fluoride. The hydrophobicity expands with an increase in fluorine content in fluoro graphite was analysed [5]. The hydrophobicity of the PVDF membrane by dispersing fluoro graphite particles employing the filtration method was built, these particles randomly adhere the surface via hydrophobic-hydrophobic forces concluded membrane surface with enhanced roughness. The dry-jet-spinning process was employed to forge a single-bore hollow fibre PVDF membrane and by liquefying fluoro graphite powder with a concentration of 0.01mg/ml in hexane forms a blanketing solution [5]. The SEM analysis revealed presence of sieves which function as the outer surface morphology prior to succeeding fluorographite blanketing. The volume of filtration solution is increased by unsystematically allocated fluorographite particles and an expansion in a number of particles. It was analysed that mechanical properties like tensile stress is increased with an increase in fluorine content. This led to the modification of PVDF membranes with rising in anti-wetting properties and surface hydrophobicity.
Graphene oxide-cellulose acetate nanocomposite membrane
Graphene oxide (GO)/Cellulose acetate (CA)/ nanocomposite membranes are manufactured with the sight of enhancing mechanical and thermal stability. Thin-film composite (TFC) consisting of two or further films accommodating asymmetric cellulose variation are mostly employed in business whose major disadvantage compared to CA membrane is low chlorine tolerance, low availability [3]. However, CA membranes still can’t compete with the TFC membrane in case of mechanical robustness, thermal soundness, salt abandonment and permeability [6]. Recently, a significant potential is manifested an atomic sheet of discarded graphene oxide to empower RO membrane implementation, owning to its inflated mechanical robustness, surface area and hydrophilicity [7]. Oxygenated functional groups of GO such as carboxyl, epoxide and hydroxyl groups are used for finer dispersion of GO in a variety of polymers [8]. Phase inversion method was employed to assemble GO/CA membrane [9]. Ghaseminezhad et al. [9] analysed using field emission scanning electron microscopy the porosity structure and morphology of membrane accompanied by a functional group of the membrane with or without GO were appreciated [9]. According to the seawater range of osmotic pressure and saline concentration, water with 25000 ppm of salt was passed through a lab-scale reverse osmosis setup with a membrane diameter of 5cm and pressure of 5-35 bars. Compared to pristine CA membrane porosity structure of membrane was altered by Inclusion of GO appraised CA matrix to sponge-like from finger-like with amplification in surface roughness. From the analysis, it was concluded that the addition of GO up to 1wt% amplifies membrane properties, which when increases further result in a negative effect. The combination of the CA membrane with 1wt% of GO increases salt rejection up to 90% and flux decreases up to 30% [9]. Figure 2a shows the ECR-1 zeolite membrane with the flux of the simulated seawater under disparate negative pressures. Figure 2b unveils dismissive pressure of 0.08 MPa at which disparate ions are refused by seawater after salvaging 3 times. The removal of Na+ ion can be spotted as 99.99%, 100%, 100%, respectively, while the abandonment of Ca2+ and Mg2+ ions are inevitably 100% in all recurring experiments. 97.1%, 97.64%, 97.23% are dismissal of K+ ion and for Cl- ion are 96.54%, 96.77%, 97.14%, respectively.
ECR-1 a zeolite membrane based on silica fume
Silica fume is a waste material from the fusion process when ferrosilicon and silicon alloys are produced which have a scandalous pozzolanic activity due to their maximum fineness; they also have the ability to respond with prolonged calcium hydroxide while hydrating cement. The consolidated zeolite comprising strongly alternating mordenite and mazzite layers forms an ECR-1 zeolite accompanied by 3.5-4.5 as a Si / Al molar ratio. Yao Jun Zhang et al. [10] proposed a seed-induced synthesis. For the ECR-1 zeolite membrane consisting of SF and MK geopolymers activated with alkali for polycondensation, accompanied by this hydrothermal crystallization assisted by amorphous geopolymer seeds [10]. This perspective leads to two extraordinary supremacies, one is an organic model unleashed to avoid surrounding contamination and the model's expulsion agents, and the other is the valuable use of large-scale commercial waste for the green synthesis of ECR a zeolite membrane -1. Yao Jun Thang et al. [10]has passed a hybrid ion K+, Na+, Ca2+, Mg2+ and Cl- of its CaCl2, NaCl, MgCl2.6H2O and KCl chlorides at room temperature with downstream pressure of 0.05 ⁓ 0.08 MPa and pressure upstream of the atmospheric condition
Challenges Faced by Reverse Osmosis
Figure 1: Various Volumes of the Fluor graphite Solution by FESEM results of the Cross-Section and Outside Surfaces prior to and succeeding the Filtration [5].
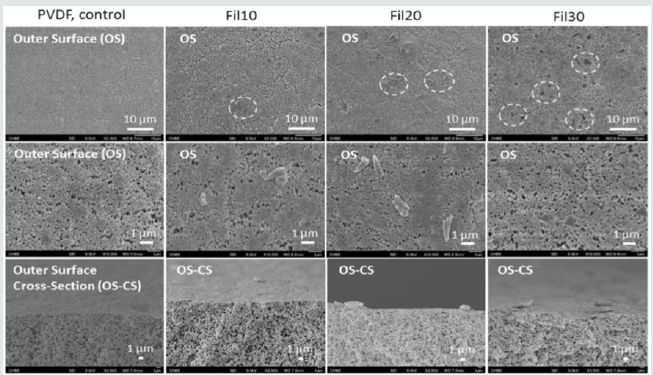
Sheikholeslami recently assumed that the dispute from now on with desalination production includes process improvement, feed water energy, material improvement, renewable energy source, brine management and a strict standard of water [11]. Immediately, Ashkelon, in Israel, is the largest plant in the world that works with reverse osmosis with a production rate of around 110 million m3 / year. In general, the average water consumption per capita of 1243 m3 / year all over the world (85% for agricultural floods, 5% for domestic use and 10% for commercial use) [12]. This plant can provide a small population of 100,000 people with fresh water. Therefore, monstrous desalination plants should be encouraged if we want to allocate new supplies of pure water to billions of people. In this framework, poor countries should have access to RO desalination. Investments, funds and operational expenses impossible for RO plants should be reduced. About 87% of all RO expenses consist of chemical composition and labour [3]. The occurrence of the optimization of the module and of the membrane material can provide the exhaustion of three wires. Furthermore, low molecular weight compounds, boron species, have brought about a considerable improvement. It is reported that 99% of boron rejection is crucial in the Middle East region so that the one-step RO procedure meets the WHO standard for drinking water, but the optimal membrane provides only 93% of the waste of boron. Furthermore, high salt depletion can feasibly reduce the RO steps needed to achieve the water quality of the applicable product. The improvement of chlorine-tolerant membranes causes the exhaustion of the scale, which reduces the costs of replacing the membrane, energy to gain further osmotic pressure and backwashing chemicals. While the operating pressure in contemporary systems is so close but so far from the thermodynamic limit and a further reduction would have a limited impact on performance [13], the reduction in energy consumptio would be substantial, since energy expenditure represents half of the total expenditure for water production. Furthermore, greater permeability would lead to a decrease in the membrane area and inevitably to a reduction in membrane replacement costs, a reduced implant footprint and a reduced use of chemical purifiers [3]. Escorted by these, the transcendental problem with desalination is the manipulation of hot brine, which is a by-product of desalination; this hot brine cannot be taken into the sea without care. As seen above, the massive desalination plants emit an enormous amount of brine every day; this brine must be managed properly as it can destroy marine life if carelessly handled (Figure 1).
Market Survey Analysis
Market survey of desalination of seawater
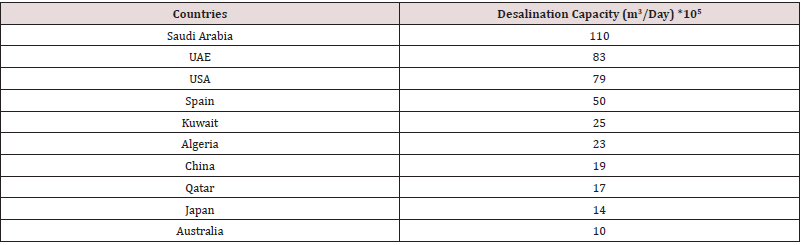
According to the survey over global countries versus desalination capacity, the existence of countries such as Saudi Arabia, the United Arab Emirates and Kuwait among others depends deliberately on seawater desalination to quench their thirst for freshwater. Middle East Asia leads and governs the global desalination market with almost 55% of the global water desalination plants available in this territory. The merged desalination capacity of all the above three country crosses 20 Mn m3 of water per day, which is approaching double the global capacity [14] (Table 2).
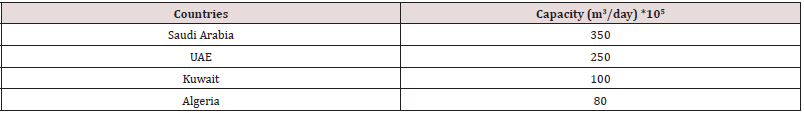
Above data is according to a 2008 survey, after that between 2011 -2016 another survey manages according to that leading four countries and their capacities are specified in the beneath table (Table 3):
Market survey of desalination method
Reverse osmosis and thermal processes such as multi-stage flash (MSF) and multi-effect distillation (MED) are mass frequently used desalination technologies. According to the International Desalination Association, the supreme process for desalination with regard to installed capacity and annual flourishing is reverse osmosis (Figure 2). As per the survey conducted in 2015, regarding installed capacity and annual growth reverse osmosis is foremost. Also, in terms of energy cost, reverse osmosis is one of the economical methods amid all methods. Comparison of Energy cost vs. desalination methods designated beneath. Due to this justification, Reverse osmosis is the most used method behind its thermal processes such as MSF and MED chiefly used [15].
Market survey of reverse osmosis membrane for desalination of seawater
A thin-film composite membrane is used in reverse osmosis is
embraced the thin scented polyamide film. This polyamide film gives
the membrane its transport characteristics, while the remains of
thin-film composite membranes assign mechanical reinforcements.
The polyamide film is a turbid and arid polymer with a high surface
that allows its high-water permeability.
Figure 3 shows the decoupling powers of the membrane and
the separation processes of the pressure membrane and of the
integrators used in water treatment, ultra-filtration, nanofiltration
and microfiltration. While ultra-filtration (UF) and microfiltration
(MF) membranes are used to confiscate bacteria (MF / UF), fine
colloidal particles (MF / UF), larger molecules (UF) such as proteins
and viruses (UF), plus Small compounds such as dissolved salts are
removed by reverse osmosis and nanofiltration. Nanofiltration and
practically all compounds dissolved in water are removed by reverse
osmosis are discarded molecules with a molecular weight greater
than 200 g/mol. The United States UU Environmental Protection
Agency [16]. Indicates RO as the best technology for eliminating
harmful compounds such as barium, arsenic and nitrate.
Future Prospective
The fabrication of slits instead of pores in membrane
The holes in membranes can model the membrane weak by truncating its strength as high stresses are evolved around holes and this can escort to the destruction of the membrane due to the high pressure of water. The stress induced in membrane due to holes can be given by following equations:
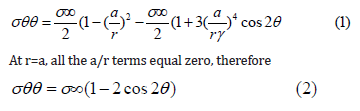
Where, σθθ is the hoop stress, σ∞ is the uniaxial tension and θ
is the loading direction [17].
These slits can provide improved permeability, selectivity, and
controllability in the desalination process. As discussed previously,
depletion in the membrane area will result due to towering
permeability, and inevitably a depletion in membrane substitution
price, a minute plant footprint and lessen use of purifying chemicals
[3]. Therefore, instead of holes, slits can be used as shown in
(Figures 4-7) which will guide smooth transit of flow and will
work as a non-return valve for the membrane. These slits can be
manufactured in any shape and size with simple manufacturing
techniques like lasers.
The PVDF membrane with Keratin (Human Hair fibre)
PVDF is a remarkable plastic used in the use that requires maximum purity, in addition to resistance to solvents, acids and hydrocarbons. PVDF has a truncated density (1.78 g/cm3) balanced with other fluoropolymers, such as polytetrafluoroethylene (Teflon). PVDF can be combined with the human hair fibre, which is hydrophilic and contains keratin in many quantities, which in turn has an affinity with soaps, i.e. fatty acids such as capric, caprylic, myristic, lauric, myristoleic, palmitoleic, palmitic acid, stearic, linoleic, oleic and linolenic. Since keratin has a characteristic to absorb, it can be used as an additive for membranes. It can also further increase the resistance of the membrane. The hair is a protein filament that grows from the follicles present in the dermis. It has been analysed that KAP 10, KAP 5, KAP 12 and KAP 17.1 transpire in the enormous proportion of the human hair cuticle, while KAP 19.2, KAP 19.1, KAP 9, KAP 7, KAP4, KAP 3, KAP 2 and KAP 1 in bark with the highest amounts. Metals such as Cu and Zn have been verified to exist in human hair with little data on Fe, Ca and other metals. The group studied that the Ca affinity for carboxylic acid and suffocate groups is greater than Cu, furthermore the scientists analysed the Cu (II) propensity to the union with primary amino groups Cu (I) and {NH2} has an affinity inflated by the thiol groups {SH}. Soaps do not form when Cu and Zn react with lipids in the hair. Carboxylic acids and sulfonic acids in hair have a greater affinity with Ca, but carboxylic acids form “soaps” when they react with Cu, but the presence of Ca decreases their degree [17]. The tensile properties of capillary fibres at 100% relative humidity compared to 55-65% relative humidity (in water) show a lower extensibility at low humidity and a higher elastic modulus and higher work and resistance values in general. Changes in human resources have an effect similar to the dynamic elastic modulus of human hair. Water, wool and hair with a virtually identical quantitative union depending on human resources. Therefore, a similar stress / strain ratio of HR was concluded [17].
Conclusion
To conclude, this work illustrates a review of various desalination processes and modern techniques towards the design of desalination membranes. Moreover, the authors of this paper propose a new combination of several materials for membrane fabrication. By examining various processes, reverse osmosis and thermal processes such as Multi-stage flash and multi-effect distillation are the frequently used desalination technologies. According to the International Desalination Association, in terms of installed capacity and yearly growth, the leading process for desalination is reverse osmosis. However, the new approach concerns the fabrication of Slits Instead of Pores in the membrane. This proposed work has the potential to give great permeability, selectivity, and controllability in the desalination performance. It reveals that a slit membrane can accomplish a water permeability rate 3.5 times larger while still maintaining perfect salt rejection. Another proposal is a combination of PVDF membrane with Keratin (Human Hair fibre) in which PVDF can be consolidated with human hair fibre which is hydrophilic and contains keratin in sufficient amount which in turn has an affinity towards soaps. As keratin has a property to absorb, it can be used for membrane additive. It can even additionally increase the strength of the membrane. However, advancements are still possible in many regions including pretreatment, integration with renewable energy, brine management, and development of membranes targeting chlorine resistance, fouling, and specific contaminants.
References
- Zotalis K, Dialynas E G, Mamassis N, Angelakis A N (2014) Desalination Technologies: Hellenic Experience. Water 6(5): 1134-1150.
- Krishna H (1961) Introduction to Desalination Technologies. p. 1-7.
- Lee KP, Arnot TC, Mattia D (2011) A Review of Reverse Osmosis Membrane Materials for Desalination-Development to Date and Future Potential. J Memb Sci 370(1-2): 1-22.
- Petersen RJ (1990) JEC Thin Film Composite Reverse Osmosis Membrane. In handbook of industial membrane technology (Ed), Noyes publication, New Jersey, USA.
- Zhao D, Zuo J, Lu K, Chung T (2017) Fluorographite Modi Fi Ed PVDF Membranes for Seawater Desalination via Direct Contact Membrane Distillation. Desalination 413: 119-126.
- De Moraes ACM, Andrade PF, De Faria AF, Simões MB, Salomão FCCS (2015) Fabrication of Transparent and Ultraviolet Shielding Composite Films Based on Graphene Oxide and Cellulose Acetate. Carbohydr Polym.
- Chae H R, Lee J Lee, C H Kim I C, Park P K (2015) Graphene Oxide-Embedded Thin-Film Composite Reverse Osmosis Membrane with High Flux Anti-Biofouling and Chlorine Resistance. J Memb Sci.
- Chang X, Wang Z, Quan S, Xu Y, Jiang Z, et.al. (2014) Exploring the Synergetic Effects of Graphene Oxide (GO) and Polyvinylpyrrodione (PVP) on Poly (Vinylylidenefluoride) (PVDF) Ultrafiltration Membrane Performance. Applied Surface Science 316: 537-548.
- Ghaseminezhad S M, Barikani M, Salehirad M (2018) Development of Graphene Oxide-Cellulose Acetate Nanocomposite Reverse Osmosis Membrane for Seawater Desalination. 161: 32-327.
- Zhang Y J, Chen H, He P Y, Li C J (2018) Developing Silica Fume-Based Self-Supported ECR-1 Zeolite Membrane for Seawater Desalination. Materials Letters 236.
- Sheikholeslami R (2009) Strategies for Future Research and Development in Desalination - Challenges Ahead. Desalination 248(1-3): 218-224.
- Hoekstra AY, Chapagain AK (2008) Globalization of Water: Sharing the Planet’s Freshwater Resources. Wiley-Blackwell, New Jersy, USA, pp. 220.
- Cho C H, Oh K Y, Kim S K, Yeo J G, Sharma P (2011) Pervaporative Seawater Desalination Using NaA Zeolite Membrane: Mechanisms of High Water Flux and High Salt Rejection. J Memb Sci 371(1-2): 226-238.
- Fritzmann C, Löwenberg J, Wintgens T, Melin T (2007) State-of-the-Art of Reverse Osmosis Desalination. Desalination 216 (1-3): 1-76.
- Culp TE, Shen Y, Geitner M, Paul M, Roy A, et al. (2018) Electron Tomography Reveals Details of the Internal Microstructure of Desalination Membranes. Proc Natl Acad Sci 115 (35): 8694-8699.
- Kirsch EG (1898) Die Theorie Der Elastizität Und Die Bedürfnisse Der Festigkeitslehre. Zeitschrift des Vereins Dtsch.
- Robbins C R (2012) Chemical and Physical Behavior of Human Hair. 5th Edition, Springer, Germany.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...