Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1687
Research Article(ISSN: 2641-1687) 
Therapeutic Effect of Barley Grains powder (Hordeum vulgare L.) Among Sudanese Patients with Proteinuria Admitted at Ibn Sina Hospital in Khartoum State - Pilot study 2018 Volume 3 - Issue 1
Simon Robinson*, Ailsa Butler, Mufeed Ali and Hanif Motiwala
- FHFT Department Urology, Oxon dept epidemiology Oxford, Frimley health foundation trust, UK
Received: July 27, 2020; Published: August 07, 2020
Corresponding author: Simon Robinson, Frimley health foundation trust, FHFT Department Urology, Oxon dept epidemiology Oxford, FHFT Dept Pathology, UK
DOI: 10.32474/JUNS.2020.03.000151
Abstract
Objectives: There are well recognised differences in health outcomes generally in society due to a range of socio-economic factors. We have compared survival outcomes in patients undergoing radical cystectomy for aggressive bladder cancer in the national health service (NHS) and compared this with those with private health insurance (PHI).
Methods: This is retrospective study of 225 NHS and 32 PHI patients operated over 14 years by one surgeon. ASA scores were compared to approximate comorbidity status.
Results: There were significantly worse outcomes for all cause and disease specific mortality in the NHS group. There was no difference in ASA scores.
Conclusions: NHS patients fare worse than those with PHI. The reasons for this difference remain unclear. PHI patients have a more extensive lymphadenectomy and have fewer overall complications.
Keywords: NHS private insurance outcomes radical cystectomy bladder cancer
Introduction
We studied the outcome of patients with private health
insurance (PHI) compared to national health service (NHS) patients.
These patients had the same operation for muscle invasive cancer
of the bladder, and we looked to see whether there were significant
differences in both all-cause mortality and disease specific
mortality. We have attempted to see whether we can propose any
explanatory cause for such differences.
There are barriers in society that delay detection and the
treatment of cancers for those people with various social and
economic disadvantages [1]. This imbalance creates health
disparities such as differences in survival [2]. There are racial,
gender, ethnic and socioeconomic components which are reflected
in their insurance status [3].
Previous studies, mostly in the United States, although with
some English and Danish studies, have looked at the effect of
insurance status on outcomes of treatment for various cancers,
including urological cancers of the prostate, kidney and bladder
[4]. In the USA, the uninsured and those on Medicaid, have a worse
stage at presentation [3-7]and a lower all cause survival than those
with private insurance [2,1,8]. Worse disease specific mortality
has also been shown to be an independent risk factor with worse
outcomes for lower socio-economic status groups [9]. This finding,
however, was not confirmed by a British study specifically looking
at social deprivation [10]. Definitions of what constitutes adverse
social groups are complex and may affect robustness of data [11]. A
shift in classification of groups, pathological definitions and coding
of cause of death may explain the lack of improvement in survival
but not socioeconomic inequalities [11].
Many factors, including socio-demographic and clinical entities,
form a complex network influencing survival. In the USA Insurance
and socioeconomic status are predictors of mortality [1,8]. Uninsured patients are often treated at low volume centres[3].
Race and gender can also affect survival [12,13] as can communal
poverty [14,15].
It has been suggested that poorer health generally, as
demonstrated by more comorbidities and unhealthy lifestyle
behaviours account for this phenomenon [2,15]. The affordable care
act in the USA is an attempt to address the problem of the uninsured
presenting with more advanced disease and their subsequent lower
survival rates [16]. In addition, a negative association between
income and prevalence of physicians for bladder and renal cancer
mortality was found [17]. Those on Medicaid were less likely to
receive definitive treatment [18]. Similar effects of socioeconomic
status have been found in England [19]and Denmark [20].
These studies have all looked at the effect of all the range of
treatments, both curative and palliative, for all types of cancer,
including low- and high-grade disease which is of particular
importance when discussing bladder cancer.
For differences in outcomes after specific surgical treatments,
research has focused on radical prostatectomy. Insurance status is
associated with stage and cancer control [21]. Higher surgery rates
and lower radiotherapy rates were observed in private patients who
were more affluent [22]. We have shown previously that insurance
status is a protective risk factor with radical prostatectomy, but
wealth itself is not protective which may be due to different adverse
lifestyle behaviour[23].
There are few published studies pertaining to insurance
status and survival after a specific treatment for bladder cancer,
particularly radical cystectomy [24].
Effect of comorbidity and ASA score
Greater comorbidities as assessed by the ASA score, increase the likelihood of complications after radical cystectomy [25]. Patients with an ASA score of three or more have a greater complication rate [25]. Severe comorbidity is also associated with increased mortality [26,27]. The ASA score has been used to predict outcomes [28] and should be used for counselling and risk stratification [29,30]. In younger patients the ASA score can be useful despite EAU guidelines suggesting otherwise [31,32]. Neglect of comorbidities can cause misleading impressions of results [33] but the ASA score can be useful at to ascertain complication risk particularly with the type of urinary diversion and the time taken to do more complex surgery [34]. Comorbidity assessed using the Charlson score is useful for younger and older patients to predict toxicity of treatment and outcomes [35,36]. All-cause mortality has a stronger relation to comorbidity than disease specific mortality does [37].
Urinarydiversion
In general, the neobladder has a higher complication rate and longer stay in hospital [38] although it may be offered to most patients [39-41]. However, in practice, it still tends to be reserved for younger and fitter patients and thus may serve as a proxy for generally better health [42].
Methods
This is a retrospective study. The 257 radical cystectomy with
bilateral lymphadenectomy (with ileal conduit or neobladder)
operations were performed by one surgeon in both NHS and the
private sector in the same county of the UK, operated from 1999 to
2013 and followed up to the present day. There were 225 patients
from the NHS and 32 patients with private health insurance. These
were sequential patients who underwent potentially curative
operative and neoadjuvant/adjuvant treatments.
We compared various patient characteristics from health
records using Iqutopia for operation details, ICE, Bostwick
laboratories and Masterlab for pathology and PACS for imaging.
Medical statistics were done using Medcalc software for logistic
regression analysis to determine significant predictors of mortality
and progression. Fishers, Mann-Whitney, t tests and log rank tests
to compare Kaplan Meier. P values less than 0.05 were considered
significant.
The ASA score was documented by the attending anaesthetist
and retrieved from Iqutopia where available.
Results
(Table 1-4) (Graph1-6)
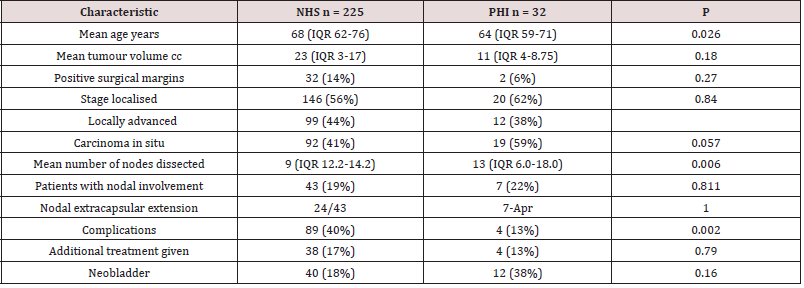
There is a significant difference between the ages of the two
populations with NHS patients being older by four years. Private
patients had a greater degree of nodal dissection and fewer
complications. However, there was no difference in tumour volume,
positive surgical margin status, advanced stage, carcinoma in situ,
nodal involvement, nodal extracapsular extension. Further, there
was no difference in rates of neo adjuvant or adjuvant treatment
or the use of a neobladder. Logistic regression showed private
health status to have highly significant negative coefficients which
are protective in all cause and disease specific mortality as well as
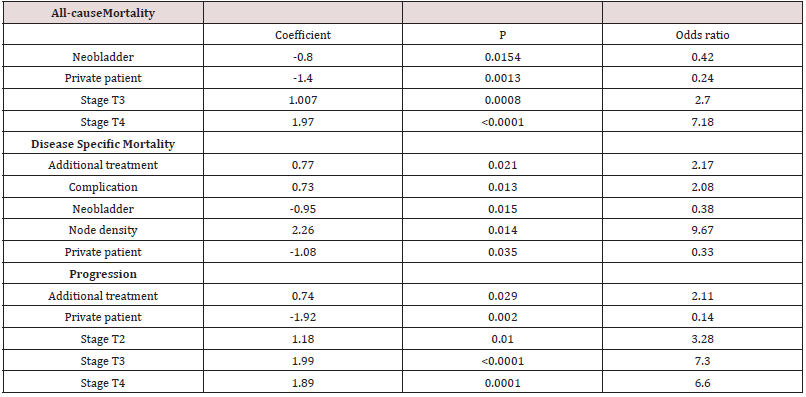
lower progression rates.
All-cause mortality was associated with negative coefficients
for neobladder and PHI. Locally advanced stage was predictive of a
deleterious outcome.
Disease specific mortality also showed adverse coefficients for
additional treatments (adjuvant and neoadjuvant), complications
and nodal density involvement.
Progression was adversely affected by additional treatment and
increasing stage.
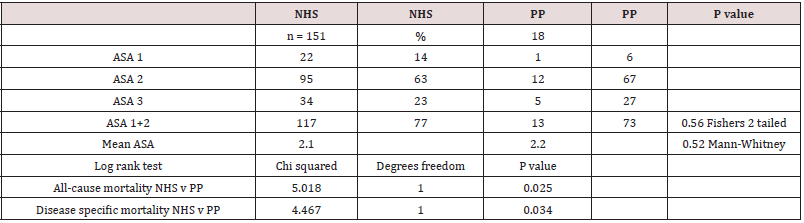
The ASA score was available in 169 of the 257 patients (66%).
There was no difference in the distributions of healthy and unhealthy patients as measured by this score. (ASA1 and 2 were
summated).
Log rank tests showed significant differences between the two
groups for mortality.
Discussion
This study has supported generally what is known about bladder cancer and the negative impact of lower socio-economic status. However former studies have looked at bladder cancer as a whole, which includes low grade and non-muscle invasive disease as well as advanced muscle invasive disease, inoperable cases, cases treated with radiotherapy and palliative cases all grouped together. This study looks at a subsection of these, those deemed appropriate for radical cystectomy. We have seen significant differences in both all-cause mortality and disease specific mortality. These differences have not been previously demonstrated. There are many gross and subtle differences that are difficult to detect with such a heterogenous disease in a heterogenous population. We propose that looking at a subgroup like this will decrease some of the obfuscating factors, by limiting geographical variation and the operation being performed by one surgeon only.
All-causemortality
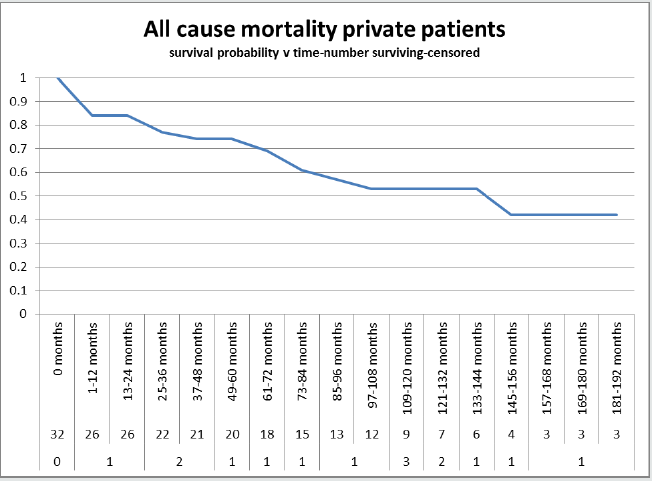
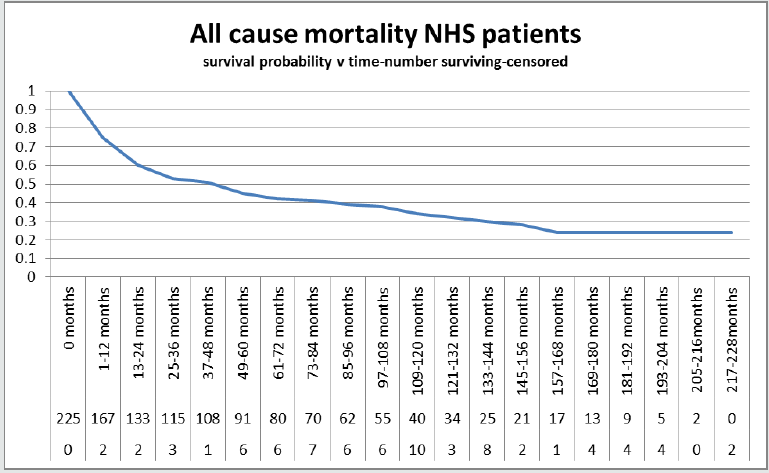
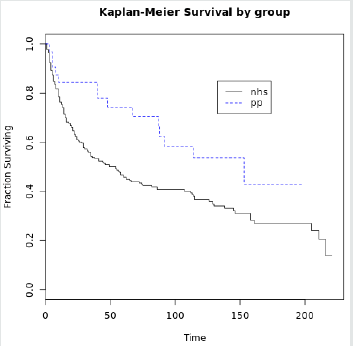
We have detected a significant difference in all-cause mortality
(Graph 5) with NHS patients (Graph 2)faring far worse (log rank
P = 0.025) and with an odds ratio 0.24 in favour of PP (Graph 1,
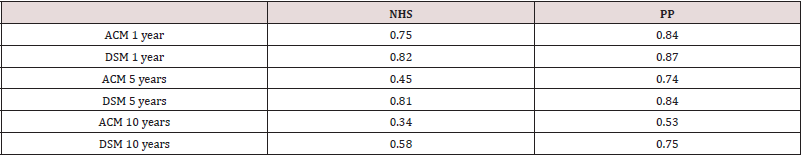
Table 2). Overall survival at one year is 0.75 for NHS and 0.84 for
PHI, and at five years it is 0.45 for NHS and 0.74 for PHI. Similarly,
at ten years for NHS patients it is 0.34 compared to 0.53 for PP
(Table 3). Numerous explanations have been proposed for the
worse prognosis overall for bladder cancer in the uninsured [1-
12]. Overall worse health is the proposed principal reason for a
worse all cause survival. We have tried to reflect potentially worse
comorbidities with the ASA score as an approximation to overall
health. There was, surprisingly, no significant difference detected
between the two groups (P = 0.56) (Table 4). Private patients were
four years younger on average and any chronic disease may not
have reached a critical point to effect outcome (P =0.026) (Table 1).
This was not a significant factor on regression analysis.
We do see that having a neobladder is a significant (P = 0.0154)
protective (negative coefficient -0.8) factor, perhaps standing as
a proxy for overall health [38,39], but there was no significant
difference in the use of this between the two groups (P=0.16) (Table
1). Locally advanced stage 3 and 4 were both significant as expected
[43, 44] with increasingly worse odds ratios (Table 2). The five-year
gold standard for radical cystectomy is about 50% [43-49], and our
population in the NHS reflects this. The patients with PHI fare much
better with a 5-year survival of 0.74.
Diseasespecificmortality
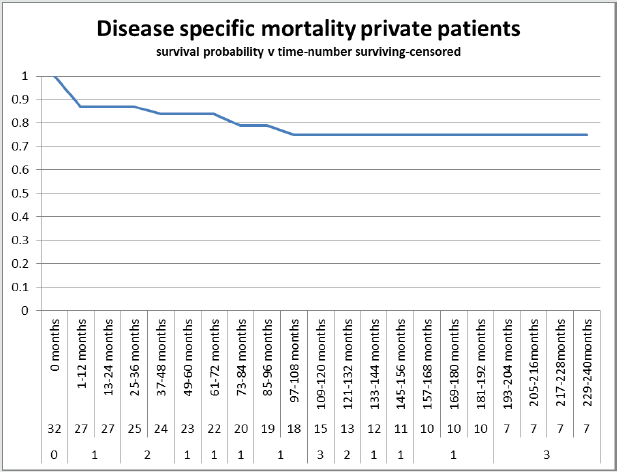
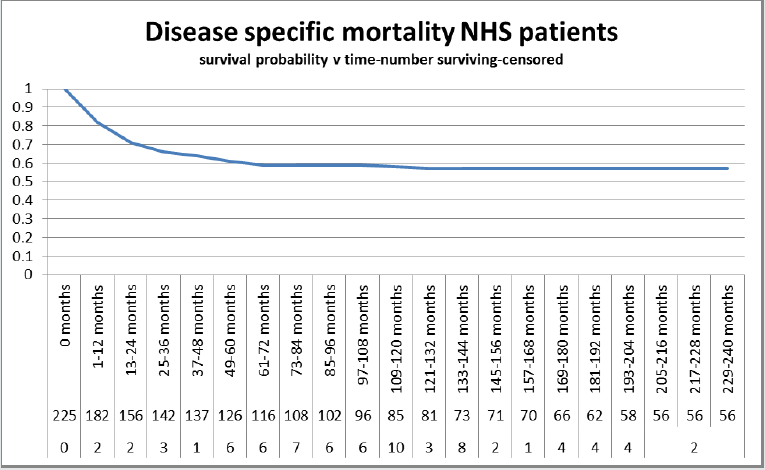
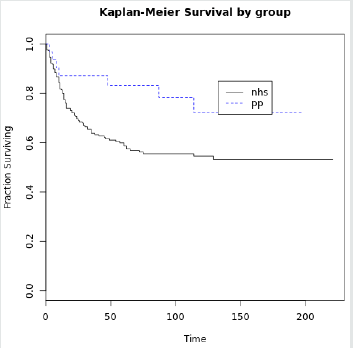
We found a significant protective coefficient and odds ratio of
0.33 in favour of private patients (Table2). There was a DSM of 0.87,
0.84 and 0.75 for PHI at one, five and ten years (Graph 3). This was
superior to NHS patients with DSM of 0.82, 0.81 and 0.58 (Table
3, Graph 4). This compares favourably with other series (49). The
log rank test comparing the two was significant (Graph 6, Table5).
Interestingly, the NHS mass of tumour was twice that of PHI, yet
there was no significant difference in tumour volume which one
might expect if there were a longer time to diagnosis in NHS patients
(11cc v 23cc P=0.18). Further, we did not detect a worse stage (NHS
56% localised, PHI 62% localised P=0.84) or a significantly greater
positive surgical margin rate in NHS patients (14% NHS v 6% PHI P
= 0.27). However, the trend was worse with all of these parameters
for NHS patients. There was no difference in rates of CIS between
the two.
The use of neobladder was, once again, a protective risk factor
(Table 2) which was used in 38% of PHI patients compared to 18%
in NHS patients, however this did not reach significance (P=0.16)
(Table 1).
Additional treatments, neo-adjuvant and adjuvant therapies,
were a significant detrimental risk factor (Table 2) but with no
significant difference between the two groups (NHS 17% and PHI
13% P=0.79).
The role of lymphadenectomy revealed no difference in
severity of extracapsular invasion (Table 1)(P=1.0) or in nodal
involvement with 19% of NHS patients having involved metastatic
deposits within at least one node, compared to 22% of PHI patients
(P=0.811). However, there was a significant difference in the
number of nodes retrieved at lymphadenectomy, with an average
of 9 for NHS and 13 for PHI patients (P=0.006). Node density is a
significant predictor of DSM with an odds ratio of 9.67 (Table2).
Complications, of all types, are a significant adverse predictor
for DSM. With a significantly worse rate in NHS, 40% compared to
PHI patients, 13% (P=0.002).
Progression
This also revealed PHI status to be protective and worse
outcomes were associated with need for additional treatment and
advancing stage.
Time to diagnosis and time to treatment from diagnosis may be
a factor, although studies are conflicting [50,51]. There is no lead
time bias (perceived increased survival with no effect on course of
cancer) as there is no screening protocol.
Conclusion
American studies have shown that insurance is important and
that even Medicaid patients do worse than private patients. We
concur that in the UK the NHS does not provide equal outcomes
compared with PHI when comparing radical cystectomy. There may
be social infrastructure issues involving hospital building capacity,
education, prevention, detection and treatment. Assistance to
patients to access and navigate healthcare systems and workplace
policies to encourage overall better health is needed. National
policies have been implemented to tackle this complex problem.
The NHS cancer plan 2000 in the United Kingdom (http.//www.doh.
gov.uk/cancer/pdfs/cancerplan.pdf) aims to reduce inequalities in
survival between rich and poor with a 62-day target to treatment.
The affordable care act in the USA highlights benefits of extending
health coverage to the uninsured [1].
We have found significant differences in outcomes for all cause
and disease specific mortality, yet the cause is elusive. We have
shown a direct comparison of NHS and PHI to reveal these outcomes
which can remain hidden in American studies as the SEER datebase
does not include uninsured patients by definition. There is no
difference in tumour volume, carcinoma in situ, advanced stage
or not, nodal involvement, extracapsular involvement of nodes,
rates of adjuvant and neo-adjuvant treatment. There are, however,
differences in complication rates and extent of lymphadenectomy
with favourable features pertaining to PHI.
We need to look for other ways to measure overall health that
include medical and lifestyle with socioeconomic components.
Limitations
The PHI cohort is small. We have not measured the time to diagnosis and to treatment. We have not been able to document the Charlson comorbidity score. We have not documented types of complications.
References
- Ward E, Halpern M, Schrag N, Cokkinides V, DeSantis C, etal. (2008) Association of insurance with cancer care utilization and outcomes. CA Cancer J Clin 58(1): 9-31.
- Niu X, Roche LM, Pawlish KS, Henry KA (2013) Cancer survival disparities by health insurance status. Cancer Med 2(3): 403-411.
- Jacobs BL, Montgomery JS, Zhang Y, Skolarus TA, Weizer AZ, etal. (2012) Disparities in bladder cancer. Urol Oncol30(1): 81-88.
- Ward EM, Fedewa SA, Cokkinides V, Virgo K (2010) The association of insurance and stage at diagnosis among patients aged 55 to 74 years in the national cancer database. Cancer J 16(6): 614-621.
- Halpern MT, Ward EM, Pavluck AL, Schrag NM, Bian J, etal. (2008) Association of insurance status and ethnicity with cancer stage at diagnosis for 12 cancer sites: a retrospective analysis. Lancet Oncol 9(3): 222-231.
- Urinary Bladder Cancer in Florida - Bureau of Epidemiology.
- Shackley DC, Clarke NW (2005) Impact of socioeconomic status on bladder cancer outcome. Curr OpinUrol 15(5): 328-331.
- Du XL, Lin CC, Johnson NJ, Altekruse S (2011) Effects of individual-level socioeconomic factors on racial disparities in cancer treatment and survival: Findings from the National Longitudinal Mortality Study, 1979-2003. Cancer 117(14): 3242-3251.
- Siddiqui MM, Heney NM, McDougal WS, Feldman AS (2015) Disparities in overall and urothelial carcinoma specific mortality associated with healthcare insurance status. Bladder 2(1): e10.
- Begum G, Dunn JA, Bryan RT, Bathers S, Wallace DM (2004) Socio-economic deprivation and survival in bladder cancer. BJU Int 94(4):539-543.
- A Shah, B Rachet, E Mitry, N Cooper,CM Brown, etal. (2008) Survival from bladder cancer in England and Wales up to 2001. Br J Cancer 99(Suppl 1): S86–S89.
- Mallin K, David KA, Carroll PR, Milowsky MI, Nanus DM (2011) Transitional cell carcinoma of the bladder: racial and gender disparities in survival (1993 to 2002), stage and grade (1993 to 2007). J Urol185(5):1631-1636.
- A Moran, AM Sowerbutts, S Collins, N Clarke, R Cowan (2004) Bladder cancer: Worse survival in women from deprived areas. British Journal of Cancer 90: 2142-2144.
- Brookfield KF, Cheung MC, Gomez C, Yang R, Nieder AM (2009) Survival disparities among African American women with invasive bladder cancer in Florida. Cancer 115(18): 4196-4209.
- Kaye DR, Canner JK, Kates M, Schoenberg MP, Bivalacqua TJ (2016) Do African American Patients Treated with Radical Cystectomy for Bladder Cancer have Worse Overall Survival? Accounting for Pathologic Staging and Patient Demographics Beyond Race Makes a Difference. Bladder Cancer 2(2): 225-234.
- Mahal BA, Aizer AA, Ziehr DR, Hyatt AS, Lago-HernandezC (2014) The association between insurance status and prostate cancer outcomes: implications for the Affordable Care Act. Prostate Cancer Prostatic Dis 17(3): 273-279.
- Janet Colli, Leah Grossman, Oliver Sartor, Benjamin R Lee (2011) 50 Urologic cancer mortality rates stratified by geographic region and physician prevalence in the united states. Journal of UrologyGeneral& Epidemiological Trends & Socioeconomics: Evidence-based Medicine & Outcomes 185(4s): e21-e22.
- Mahal AR, Mahal BA, Nguyen PL, Yu JB (2018) Prostate cancer outcomes for men aged younger than 65 years with Medicaid versus private insurance. Cancer 124(4): 752-759.
- MP Coleman, B Rachet, LM Woods, E Mitry, M Riga, etal (2004) Trends and socioeconomic inequalities in cancer survival in England and Wales up to 2001. British Journal of Cancer 90(7):1367-1373.
- Kirsten Thorup Eriksen, Anne Petersen, AslakHarbo Poulsen, Isabelle Deltour, Ole Raaschou-Nielsen (2008) Social inequality and incidence of and survival from cancers of the kidney and urinary bladder in a population-based study in Denmark, 1994-2003. European Journal of Cancer44(14):2030-2042.
- Andrea Gallina, Pierre I Karakiewicz, Felix K-H Chun, Alberto Briganti, Hartwig Huland, etal. (2007) Health-insurance status is a determinant of the stage at presentation and of cancer control in European men treated with radical prostatectomy for clinically localized prostate cancer. BJU Int 99(6): 1404-1408.
- Barbiere JM, Greenberg DC, Wright KA, Brown CH, Palmer C, etal. (2012) The association of diagnosis in the private or NHS sector on prostate cancer stage and treatment. J Public Health 34(1): 108-114.
- Simon Robinson, Marc Laniado, Assad Farooq, Hanif Motiwala, Mohamed Omar, etal. A comparison of clinical parameters at presentation, pathological outcomes and biochemical relapse between NHS and private patients undergoing radical prostatectomy at a single centre in the United Kingdom. Journal of clinical urology 9(5): 326-334.
- Robinson S, Rao A (2015) Differences in mortality and patient characteristics between national health service patients and those with private health insurance post radical cystectomy. Resource Centre - EMUC15.
- Novotny V, Zastrow S, Koch R, Wirth MP (2012) Radical cystectomy in patients over 70 years of age: impact of comorbidity on perioperative morbidity and mortality. World J Urol 30(6): 769-776.
- Fairey A, Chetner M, Metcalfe J, Moore R, Todd G, etal. (2008) Associations among age, comorbidity and clinical outcomes after radical cystectomy: results from the Alberta Urology Institute radical cystectomy database. J Urol 180(1): 128-134.
- Bartosz Dybowski, Krzysztof Ossoliński, Anna Ossolińska, Michal Peller, EwaBres-Niewada, etal. (2015) Impact of stage and comorbidities on five-year survival after radical cystectomy in Poland: single centre experience. Cent European J Urol 68(3): 278-283.
- Aziz A, May M, Burger M, Palisaar RJ, Trinh QD, etal. (2014) Prediction of 90-day mortality after radical cystectomy for bladder cancer in a prospective European multicenter cohort. Eur Urol 66(1): 156-163.
- Boorjian SA, Kim SP, Tollefson MK, Carrasco A, Cheville JC, etal. (2013) Comparative performance of comorbidity indices for estimating perioperative and 5-year all-cause mortality following radical cystectomy for bladder cancer. J Urol 190(1): 55-60.
- Williams SB, Kamat AM, Chamie K, Froehner M, Wirth MP, etal. (2018) Systematic Review of Comorbidity and Competing-risks Assessments for Bladder Cancer Patients. Eur Urol Oncol 1(2): 91-100.
- Froehner M, Koch R, Hübler M, Heberling U, Novotny V, etal. (2018) Predicting 90-day and long-term mortality in octogenarians undergoing radical cystectomy. BMC Urol 18(1): 91.
- Bokarica P, Hrkac A, Gilja I (2017) Re: J. Alfred Witjes, Thierry Lebret, Eva M. Compérat, et al. Updated 2016 EAU Guidelines on Muscle-invasive and Metastatic Bladder Cancer. Eur Urol 2017; 71: 462-75. Eur Urol 72(2): e45.
- Feinstein AR (1970) The pre-therapeutic classification of co-morbidity in chronic disease. J Chronic Dis 23(7): 455-468.
- Malavaud B, Vaessen C, Mouzin M, Rischmann P, Sarramon J, etal. (2001) Complications for radical cystectomy. Impact of the American Society of Anesthesiologists score. Eur Urol 39(1): 79-84.
- Extermann M, Overcash J, Lyman GH, Parr J, Balducci L (1998) Comorbidity and functional status are independent in older cancer patients. J Clin Oncol 16(4):1582-1587.
- Figueroa AJ, Stein JP, Dickinson M, Skinner EC, Thangathurai D, etal. (1998) Radical cystectomy for elderly patients with bladder carcinoma: an updated experience with 404 patients. Cancer 83(1):141-147.
- Miller DC, Taub DA, Dunn RL, Montie JE, Wei JT (2003) The impact of co-morbid disease on cancer control and survival following radical cystectomy. J Urol 169(1): 105-109.
- Evans B, Montie JE, Gilbert SM (2010) Incontinent or continent urinary diversion: how to make the right choice. Curr OpinUrol 20(5): 421-425.
- Autorino R, Quarto G, Di Lorenzo G, De Sio M, Perdonà S, etal. (2009) Health related quality of life after radical cystectomy: comparison of ileal conduit to continent orthotopic neobladder. Eur J Surg Oncol 35(8): 858-864.
- Lawrentschuk N, Colombo R, Hakenberg OW, Lerner SP, Månsson W, etal. (2010) Prevention and management of complications following radical cystectomy for bladder cancer. Eur Urol 57(6): 983-1001.
- Lee KS, Montie JE, Dunn RL, Lee CT (2003) Hautmann and Studer orthotopic neobladders: a contemporary experience. J Urol 169(6): 2188-2191.
- Saika T, Suyama B, Murata T, Manabe D, Kurashige T, etal. (2001) Orthotopic neobladder reconstruction in elderly bladder cancer patients. Int J Urol 8(10): 533-538.
- Bassi P, Ferrante GD, Piazza N, Spinadin R, Carando R, etal. (1999) Prognostic factors of outcome after radical cystectomy for bladder cancer: a retrospective study of a homogeneous patient cohort. J Urol 161(5): 1494-1497.
- Dalbagni G, Genega E, Hashibe M, Zhang ZF, Russo P, etal. (2001) Cystectomy for bladder cancer: a contemporary series. J Urol 165(4): 1111-1116.
- Shariat SF, Karakiewicz PI, Palapattu GS, Lotan Y, Rogers CG, etal. (2006) Outcomes of radical cystectomy for transitional cell carcinoma of the bladder: a contemporary series from the Bladder Cancer Research Consortium. J Urol 176(6 Pt 1): 2414-2422.
- Ghoneim MA, el-Mekresh MM, el-Baz MA, el-Attar IA, Ashamallah A (1997) Radical cystectomy for carcinoma of the bladder: critical evaluation of the results in 1,026 cases. J Urol 158(2): 393-399.
- Stein JP, Skinner DG (2006) Radical cystectomy for invasive bladder cancer: long-term results of a standard procedure. World J Urol 24(3): 296-304.
- Williams SB, Huo J, Chu Y, Baillargeon JG, Daskivich T, etal. (2017) Cancer and All-cause Mortality in Bladder Cancer Patients Undergoing Radical Cystectomy: Development and Validation of a Nomogram for Treatment Decision-making. Urology 110: 76-83.
- Taekmin Kwon, In Gab Jeong, Dalsan You, Bumsik Hong, Jun Hyuk Hong, etal. (2014) Long-Term Oncologic Outcomes after Radical Cystectomy for Bladder Cancer at a Single Institution. J Korean Med Sci 29(5): 669-675.
- Nielsen ME1, Palapattu GS, Karakiewicz PI, Lotan Y, Bastian PJ, etal. (2007) A delay in radical cystectomy of >3 months is not associated with a worse clinical outcome. BJU Int 100(5): 1015-1020.
- Gore JL, Lai J, Setodji CM, Litwin MS, Saigal CS (2009) Mortality increases when radical cystectomy is delayed more than 12 weeks: Results from a Surveillance, Epidemiology, and End Results-Medicare analysis. Cancer 115(5): 988-996.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...