Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2641-1687
Research Article(ISSN: 2641-1687) 
Effect of Tyrosine Kinase Inhibitors on Renal Functions Volume 1 - Issue 1
Özgür Can1*, Gülizar Manga Şahin2, Umut Kasapoğlu3,Süleyman Baş4 and Bala Başak Öven Ustaalioğlu5
- 1Department of Nephrology, Tatvan State Hospital, Turkey
- 2Department of Nephrology, University of Health Sciences, Sultan Abdülhamid Han Training and Research Hospital, Turkey
- 3Department of Nephrology, Ağrı State Hospital, Turkey
- 4Department of Internal Medicine, Haydarpaşa Numune Training and Research Hospital, Turkey
- 5Department of Oncology, Haydarpaşa Numune Training and Research Hospital, Turkey
Received: June 26, 2018; Published: July 02, 2018
Corresponding author: Özgür Can, Bitlis Tatvan Devlet Hastanesi. Bitlis Tatvan Karayolu Üç Yol Mevkii Tatvan, Bitlis/TÜRKİYE Postal Code: 06434
DOI: 10.32474/JUNS.2018.01.000106
Abstract
Background: We aim to document if any difference exists for renal functions between metastatic and non-metastatic patients.
Methods: The study population included 12 metastatic and 15 non-metastatic patients. Metastatic renal cancer patients using the TKIs were compared to non- metastatic patients.
Results:Preoperative estimated glomerular filtration rate (e-GFR) was significantly low in metastatic patients than nonmetastatic patients (p: 0.048). A trend toward increased acute kidney injury during hospital stay in the non-metastatic group was observed, but this fell just short of statistical significance (p: 0.109). Two groups did not differ significantly in terms of postoperative e-GFR (p: 0.256). No statistically significant differences were observed in actual e- GFR between two groups (p: 0.638). No statistically significant differences were found in pre-TKIs and post-TKIs e-GFR values (p: 0.735). Proteinuria was statistically more common in metastatic patients than non-metastatic patients (p<0.001). No statistically significant difference in age, sex, follow-up period, NSAIDs use, antihypertensive and ARBs/ACEIs use were documented between the two groups.
Conclusion: Increased risk for proteinuria was documented in metastatic patients with TKIs use. However, use of the TKIs had no effect on e- GFR. No statistically significant differences were observed in actual e- GFR between two groups.
keywords: Hypertension, Glomerular Filtration Rate, Proteinuria, Tyrosine- Kinase Inhibitors
Abbrevations: RCC: Renal cell carcinoma , TKIs: Tyrosine-Kinase Inhibitors, AKI: Acute Kidney Injury, NSAIDs: Non-Steroidal Anti-Inflammatory Drugs, ARBs: Angiotensin Receptor Blockers, ACEIs: Angiotensin Converting Enzyme Inhibitors, ARBs: Angiotensin Receptor Blockers, CCBs: Calcium Channel Blockers, e- GFR: Estimated Glomerular Filtration Rate
Introduction
Renal cell carcinoma (RCC) accounts for approximately 3% of adult malignancies [1]. Treatment options for the RCC include surgery, radiotherapy and immunotherapy [2]. Besides these options, molecular-targeted therapies in the form of tyrosinekinase inhibitors (TKIs) are commonly used. These agents are well tolerated compared to previously used options (interleukin 2 or interferon-α) and the use of these agents results in longer progression-free survival and increased response rates [3-5]. Despite the advances achieved with these agents, use of the TKIs results in side effects such as fatigue, nausea, diarrhea, gastrointestinal hemorrhage, dysphonia, and palmar-plantar erythrodysaesthesia [6]. Hypertension and proteinuria are also usually encountered after the start of these agents [7-9]. In this study, we compared clinical features between patients with renal cell carcinoma with and without the TKIs treatment and aim to document if any difference exists for renal functions between metastatic and non-metastatic patients during their follow up period.
Materials and Methods
The patients diagnosed with the RCC and followed up at Istanbul Health Sciences University Haydarpaşa Numune Education and Research Hospital oncology polyclinics between 2009 and 2016 were evaluated. Eligible 12 metastatic patients who received a nephrectomy and were treated with the TKIs were included in the metastatic group. Fifteen non-metastatic RCC patients who only received a nephrectomy formed the non-metastatic group. Metastatic patients who did not receive a nephrectomy, patients on dialysis, and patients with insufficient data were excluded. Patients’ demographics and laboratory data were collected from the hospital records and via telephone. The clinical, demographic, and laboratory parameters were compared between the two groups. The data collected included patient age, sex, follow-up time, nephrology follow-up, mortality, history of perioperative acute kidney injury(AKI), use of non-steroidal anti-inflammatory drugs (NSAIDs), use of antihypertensive medications including angiotensin converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs), calcium channel blockers(CCBs), and alpha and beta blockers. The data also included laboratory parameters such as estimated glomerular filtration rate (e-GFR) (calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation 2) and proteinuria (assessed using dipstick and 24-hour urine collection). Creatinine levels before the operations were used to calculate postoperative e-GFR, and postoperative creatinine levels taken just before hospital discharge were used to calculate postoperative e-GFR. Actual e-GFR was calculated using creatinine levels measured at the last hospital visit. Perioperative AKI was defined using the acute kidney injury network classification system. Increase in serum creatinine of 0.3 mg/dl or more within 48 hours or ≥ 1.5 times baseline within 7 days was used in order to define perioperative AKI. Urine volume was not used as a criterion because data for urine volume were not available.
The TKIs used as targeted therapy for our patients were sunitinib, pazopanib, and axitinib. Available creatinine measurements before and after the TKIs treatment were used to present the effect of the TKIs on renal function. Statistical analyses were performed using the MedCalc Statistical Software version 12.7.7 (MedCalc Software bvba, Ostend, Belgium; http://www. medcalc.org; 2013). The normality of continuous variables was investigated using the Shapiro-Wilk test. Descriptive statistics are presented using the mean and standard deviation for normally distributed variables and median (and minimum–maximum) for the non-normally distributed variables. Student’s t-test was used to compare two normally distributed groups. Non-parametric statistical methods were used for values with skewed distributions. The Mann-Whitney U test was used to compare two non-normally distributed groups. The χ² test was used for categorical variables and expressed as observation counts (and percentages). Statistical significance was accepted when the two-sided p value was lower than 0.05.
Results
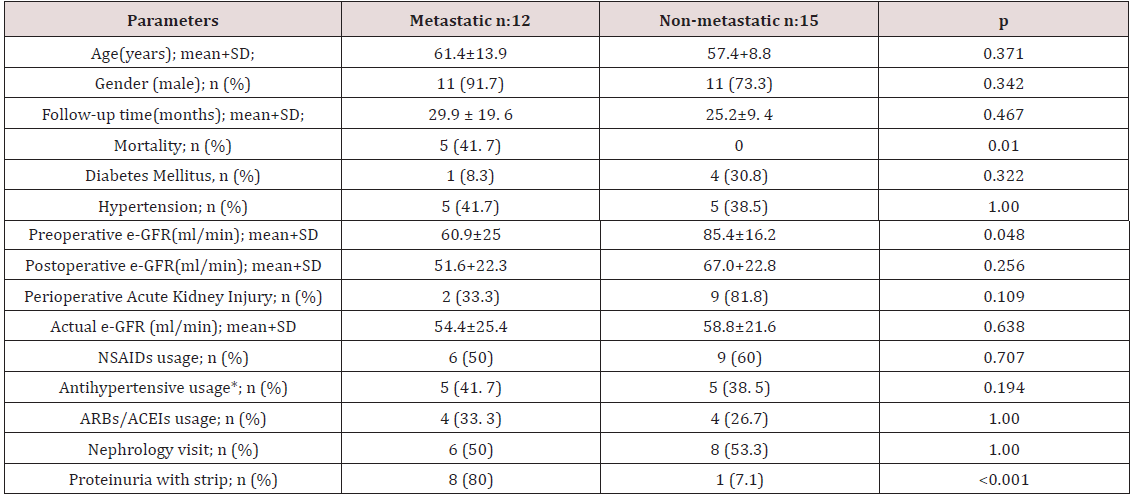
The study population consisted of 22 males and 5 female patients. The mean age of study population was 59.2±11.3 years. Clinical and laboratory parameters of metastatic and nonmetastatic patients are listed in Table 1. Preoperative e-GFR was significantly lower in metastatic patients when compared to nonmetastatic patients (p: 0.048). When two groups were compared, a trend toward the increased AKI during hospital stay in the nonmetastatic group was observed, but this fell just short of statistical significance (p: 0.109). There was no statistically significant result between two groups when two groups were compared for postoperative e-GFR (p: 0.256). The statistically nonsignificant result for postoperative e-GFR between two groups was also observed for actual e-GFR (p: 0.638) and no statistically significant differences were found between pre-TKI and post-TKI e-GFR values (p: 0.735). Proteinuria was more common in metastatic patients than non-metastatic patients (p<0.001). In metastatic group 8 patients had proteinuria. Four patients out of these 8 patients had 24-hour proteinuria measurements. One patient had nephrotic range proteinuria and the other three patients had non-nephrotic range proteinuria. No biopsies were performed on these patients with proteinuria. Two metastatic patients had negative results for proteinuria. Data for dipstick proteinuria were unavailable for the other two patients. Only one patient in the non-metastatic group had proteinuria and it was at a level of less than 1 gr/day. There was no statistically significant differences in age, sex, follow-up period, NSAIDs use, antihypertensive and ARBs/ACEIs use, and follow-up time between metastatic and non- metastatic patients. Follow-up times for metastatic and non-metastatic patients were 29.9±19.6 and 25.2±9.4 months; respectively. Three patients died during the follow-up period and all were in the metastatic group.
e-GFR: Estimated glomerular filtration rate; NSAİDs: Non-steroidal anti-inflammatory drugs; ARBs/ACEIs: angiotension receptor blockers/ angiotension converting enzyme inhibitors; * antihypertensive use rather than ARBs/ACEIs
Discussion
Nearly all patients taking the TKIs experience a rise in blood pressure. Systemic vasoconstriction and volume overload are parts of the mechanisms responsible for systemic hypertension caused by TKIs [7]. Despite the risk of hypertension occurrence, the development of hypertension is mostly a good prognostic sign because it is associated with longer progression free and overall survival and can be used as a biomarker for tumor responsiveness [10]. Regarding our study, there was no statistically significant results between groups for ACEIs/ARBs and other antihypertensive use. Proteinuria is another important side effect that can be encountered after the start of targeted agents. In the kidney, the vascular endothelial growth factor pathway is known to be responsible for proteinuria after the start of TKIs [11]. In the study of Baek et al. [12] initiation of sunitinib therapy was related to proteinuria and aggravation of preexisting proteinuria in 17.6% and 23.1% of patients; respectively [12]. Again in the COMPARZ study, discontinuation of treatment because of proteinuria was observed in 3% and 1% the of patients treated with pazopanib and sunitinib; respectively [13]. In our study, most of the patients treated with the TKIs had proteinuria. However, only one patient had nephroticrange proteinuria. We did not perform a renal biopsy on this patient but important data can be obtained through this procedure. Biopsyproven acute interstitial nephritis, thrombotic microangiopathy and acute tubular necrosis have also been encountered after the TKIs use [14-16]. In our study, there was no statistically significant difference between pre-TKIs and pre-TKIs e-GFR values. However, patients can develop renal insufficiency during treatment with targeted agents. A study by Zhu et al. [17] showed the development of renal insufficiency in the RCC patients receiving sunitinib [17]. In our study, despite the statistically significant result for preoperative e- GFR in favor of non- metastatic patients, there was no statistically significant results for postoperative e-GFR between two groups which may be due to the high percentage of patients with AKI in the non-metastatic group. Despite the tumor burden and use of the TKIs in metastatic patients, the non-significant result for postoperative e-GFR between two groups did not change at last hospital visit. The small sample size and retrospective nature of the study are important limitations that should be considered. However, renal side effects encountered after use of the TKIs were emphasized in this study with comparing metastatic and non-metastatic RCC patients. In conclusion, increased risk for proteinuria was documented in metastatic patients with TKIs use. However, use of the TKIs had no effect on e- GFR. There was also no statistically significant difference for the actual e-GFR between the two groups despite the higher preoperative e-GFR observed for non- metastatic patients.
Compliance with Ethical Standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Institutional approval has been obtained.
References
- Rini BI, Campbell SC, Escudier B (2009) Renal cell carcinoma. Lancet 373: 1119-1132.
- Ljungberg B, Bensalah K, Canfield S, Dabestani S, Hofmann F, et al. (2015) EAU Guidelines on Renal Cell Carcinoma: 2014 Update. Eur Urol 67: 913-924.
- Russo P, Jang TL, Pettus JA, Huang WC, Eggener SE, et al. (2008) Survival rates after resection for localized kidney cancer: 1989 to 2004. Cancer 113: 84-96.
- Ferry EK, Minnillo BJ, Maurice MJ, Abouassaly R, Zhu H (2015) Trends of Systemic Therapy Use for Renal Cell Carcinoma in the United States. Urology 85: 1399-403.
- Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, et al. (2007) Sunitinib versus interferon alfa in metastatic renal cell carcinoma. N Engl J Med 356: 115-124.
- Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, et al. (2009) Sorafenib for treatment of renal cell carcinoma: final efficacy and safety results of the phase III treatment approaches in renal cancer global evaluation trial. J Clin Oncol 27: 3312-3318.
- Ruiz JN, Belum VR, Creel P, Cohn A, Ewer M, et al. (2014) Current practices in the management of adverse events associated with targeted therapies for advanced renal cell carcinoma: a national survey of oncologists. Clin Genitourin Cancer 12: 341-347.
- Robinson ES, Khankin EV, Karumanchi SA, Humphreys BD (2010) Hypertension Induced by VEGF Signaling Pathway Inhibition: Mechanisms and Potential Use as a Biomarker Semin Nephrol 30(6): 591-601.
- Eremina V, QuagginSE, Biology of Anti-Angiogenic Therapy Induced Thrombotic Microangiopathy. Semin Nephrol 30: 582-590.
- Szmit S, Langiewicz P, Złnierek J, Nurzyński P, Zaborowska M, et al. (2012) Hypertension as a predictive factor for survival outcomes in patients with metastatic renal cell carcinoma treated with sunitinib after progression on cytokines. Kidney Blood Press Res 35(1): 18-25.
- Eremina V, Jefferson JA, Kowalewska J, Hochster H, Haas M, et al. (2008) VEGF inhibition and renal thrombotic microangiopathy. N Engl J Med 358: 1129-1136.
- Baek SH, Kim H, Lee J, Kim DK, Oh KH, et al. (2014) Renal adverse effects of sunitinib and its clinical significance: A single center experience in Korea. Korean J Intern Med 29(1): 40-48.
- Motzer RJ, Hutson TE, Cella D, Reeves J, Hawkins R, et al. (2013) Pazopanib versus sunitinib in metastatic renal-cell carcinoma. N Engl J Med 369: 722-731.
- Bollée G, Patey N, Cazajous G, Robert C, Goujon JM, et al. (2009) Thrombotic microangiopathy secondary to VEGF pathway inhibition by sunitinib. Nephrol Dial Transplant 24(2): 682-685.
- Winn SK, Ellis S, Savage P, Sampson S, Marsh JE (2009) Biopsy-proven acute interstitial nephritis associated with the tyrosine kinase inhibitor sunitinib: a class effect?. Nephrol Dial Transplant 24(2): 673-675.
- Chen YS, Chen CL, Wang JS (2009) Nephrotic Syndrome and Acute Renal Failure Apparently Induced by Sunitinib. Case Rep Oncol 2(3): 172-176
- Zhu X, Stergiopoulos K, Wu S (2009) Risk of hypertension and renal dysfunction with an angiogenesis inhibitor sunitinib: systematic review and meta analysis . Acta Oncol 48(1): 9-17.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...