Lupine Publishers Group
Lupine Publishers
Menu
ISSN: 2690-5787
Research Article(ISSN: 2637-4692) 
Chediak-Higashi Syndrome Volume 1 - Issue 1
N Khelafi*, MS Ladj, Y Ferhani, R Belbouab, S Sokhal, A Hadji, N Benali Khodja, N Baghdali and R Boukari
- Pediatrics Department CHU Mustapha, Algiers, Algeria
Received: June 17, 2019; Published:June 27, 2019
Corresponding author: N Khelafi, Pediatrics Department CHU Mustapha, Algiers, Algeria
Abstract
Chediak-Higashi syndrome (SCH) is a rare autosomal recessive genetic disorder characterized by oculo-cutaneous albinism, immunodeficiency responsible for recurrent infections, predisposition to bleeding, And late neurological deterioration. The pathognomonic sign is the presence of giant intracytoplasmic granules in most of the cells of the organism but often they are identified in peripheral blood. In 85% of cases, CHS patients develop the accelerated phase characterized by an Hemophagocytic Lymphohistiocytosis syndrome (HLH) responsible for a high mortality rate. The only current effective treatment of haematological and immunological abnormalities remains allogeneic bone marrow transplantation, but without impact on skin manifestations or subsequent neurological deterioration. It is all the more effective as it is performed before the onset of an HLH syndrome.
Introduction
Chediak-Higashi syndrome (SCH) is a rare autosomal recessive genetic disorder characterized by oculo-cutaneous albinism, immunodeficiency by cytotoxic activity of T lymphocytes and natural killer cells responsible for recurrent infections, predisposition to bleeding, And late neurological deterioration. According to the International Union of Immunological Societies, the SCH is a primary immunodeficiency by immune dysregulation belonging to familial lymphohistiocytic haematophagocytosis syndromes (HFH) with hypopigmentation [1]. The LYST-CHS1 (Lysosomal Trafficking Regulator Gene) gene was identified on the long arm of chromosome 1 in 1q42-q43 [2,3]. This gene encodes the CHS protein whose exact function remains imprecise. About 500 cases have been reported [4,5]. The diagnosis is oriented by the clinical signs and facilitated by the study of the microscopic aspect of the hair which highlights the presence of pigment aggregates; But the pathognomonic sign of the disease is the presence of giant intracytoplasmic granulations in most cells of the organism [6], especially in peripheral blood or bone marrow. Approximately 85% of patients develop an acceleration phase characterized by a syndrome of lymphohocytic hemophagocytosis (HLH), which occurs during the first decade, rarely present at the onset of the disease [7]; It is fatal in the absence of treatment [8]. Currently, the only effective therapeutic option is bone marrow transplantation, which improves haematological and immune abnormalities, but does not prevent subsequent neurological deterioration. The prognosis remains poor in the absence of a bone marrow transplant, the death often occurring before the age of ten years.
Patients and Methods
They are four children followed in the pediatric department of the CHU Mustapha of Algiers for Chediak-Higashi syndrome between 2014 and 2017. The diagnosis was focused on the clinical manifestations, the presence of giant intra-leukocyte granulations. A genetic study in three children confirmed the diagnosis.
Results
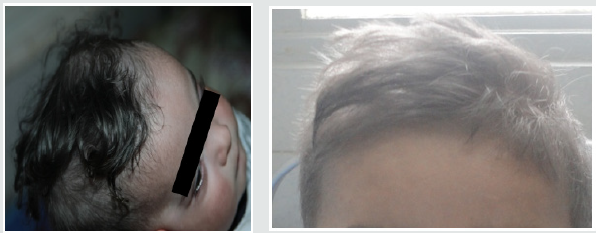
These are three boys and one daughter with an average age of diagnosis of 3.2 years (7 months - 6 years). Consanguinity is found in all cases as well as a family form (2 brothers). Three patients have a history of repeated infections. Children who were vaccinated did not report any particular incidents. Clinically, oculocutaneous albinism is present in 3 children (Figure 1) and melanoderma with a highly pigmented iris in 1 case (Table 1). Silver gray hair is present in all patient (Figure 2). The peripheral blood smear allowed to make the diagnosis by showing the intra-cytoplasmic giant granulations in all the patients. Microscopic study of the hair found deposits of melanin in irregular clods in the hair shaft in favor of Chediak-Higashi syndrome (Figure 3). A syndrome of lymphohistiocyte hemophagocytosis (HLH) (Table 2) is present in 3 cases and then in one case 8 months after the diagnosis.
Figure 1: Hypopigmentation of the skin Clinically, oculocutaneous albinism is present in 3 children hypo pigmentation of the left iris
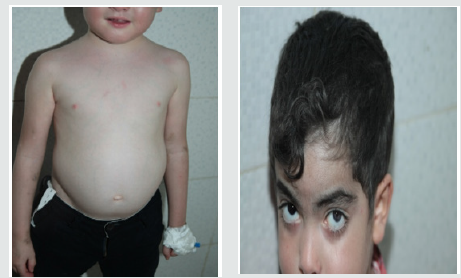
Figure 3: Deposition of melanin in the hair shaft Microscopic study of the hair found deposits of melanin in irregular clods in the hair shaft in favor of Chediak-Higashi syndrome

Melanoderma with a Highly Pigmented Iris in 1 Case All patients with signs of lymphohistiocytic activation were put on HLH 2004 protocol. The evolution is marked by a good tolerance of the treatment. One child received an allogeneic marrow transplant but died six months later. The remaining three patients are still alive and clinically stable.
Discussion
Autosomal recessive genetic disease very rare, Chediak-Higashi syndrome affects all races and all age groups. Less than 500 cases have been reported [4,5]. Most often they are reported cases or small series published. Prevalence is difficult to determine due to reported cases more than once, and other unreported cases. The largest published series (15 cases) was reported in Japan over a period of ten years [9]. Inbreeding in all our patients is reported in 50 to 85% of cases [5]. The LYST / CHS1 gene of the disease encodes the cytosolic CHS protein whose function remains imprecise. It would play a role in the exocytosis of proteins from late multivesicular endosomes. More than sixty mutations have been reported in the literature (false sense, nonsense, deletion, insertions) [10]. Mutations in the LYST / CHS1 gene lead to an abnormal function of the CHS protein with an impairment of intracytoplasmic transport, protein sequestration in giant intracytoplasmic structures and blocking of the secretory function, in particular that of leucocytes and melanocytes. Phenotype / genotype correlations were reported, thus a deletion-type mutation correlated with the early and fulminant onset of the acceleration phase, while a missense-like mutation correlated with a better prognosis with no acceleration phase and neurological deterioration [11]. Clinical signs begin after birth, or before the age of 5 years. Oculocutaneous albinism (AOC) is an important sign of diagnostic orientation, present in three of our patients, characterized by hypopigmentation which generally affects the skin, the hair and the eyes. It is related to pathological aggregation and an unequal distribution of melanosomes. The AOC may be present from birth, and concern the three organs or some of them, total or partial or even absent [12]. Sometimes hyperpigmentation as in one of our patients can exceptionally be seen, delaying the diagnosis [13,14]. Most patients have photosensitivity. Some patients have an atypical phenotype with an attenuated form, or the AOC is subtle or absent and probably unknown [11,15]. Rarely other skin lesions are observed as hyperhydrosis, erythema multiforme. The hypopigmentation of the hair gives them a blonde, gray or white color, often with a silvery or metallic luster. The eyes are blue in color and hypo pigmentation of the iris may be associated with decreased pigmentation of the retina, and ocular manifestations such as photophobia, decreased visual acuity, nystagmus, and strabismus Frequent and recurrent infections are common in childhood. Often severe, they are related to a defect in T cell cytotoxicity, NK function, and a decrease in the chemotactic and bactericidal activity of the granulocytes [16,17]. Pyogenic infections are the most frequent, especially in the skin, upper airways and mucous membranes. The most frequently isolated germs are staphylococcus aureus, β-hemolytic streptococci and pneumococcus. The involvement of the oral cavity manifests in the form of gingivitis, gingival haemorrhage, early falls of the teeth. Mouth ulcers have been described. Periodontitis has been identified as a manifestation of immune dysfunction. The bleeding tendency in these patients is related to a deficiency in the storage pool of dense granules and a defect in platelet aggregation. Haemorrhagic manifestations are usually benign and usually do not require treatment. The accelerated phase of the disease is the most important and dangerous complication of the SCH. It is responsible for a high rate of mortality within a few months [18]. It can occur at any age but especially during the first decade (85%) [19]. Our patients developed this accelerated phase before the age of 6 years. Rarely, it is the first manifestation [7,20]. Its early onset is associated with the existence of a deletion-like genetic mutation [11] and has a collapsed or absent activity in cytotoxic LTs [21]. It is manifested by a syndrome of lymphohystiocytic haematophagocytosis (HLH) whose factors triggering this acceleration are not clear. The role of EBV infection found in one of our patients was raised without this relationship being established [22]. The diagnosis of accelerated phase disease is based on the criteria of the Histiocytic Society 2004 [23]. Neurological manifestations occur in about 50% of cases and may occur at any time in childhood or adulthood, they are variable: peripheral neuropathy, coma, convulsions, ataxia, cognitive disorders, impaired balance, movement abnormalities and mental alterations. The bone marrow transplant does not prevent their subsequent appearance [24]. The diagnosis of SCH should be early, often done at around 6 years of age, but in about 25% of cases the diagnosis is delayed after age 10 [12] for our patients. The average age at diagnosis was 3.2 years. It is suspected on the clinical elements, facilitated by the microscopic study of the hair which shows aggregates of melanin pigments found in all our patients, this aspect allows the differential diagnosis with other types of cutaneous hypopigmentation. But the pathognomonic sign of the disease is the presence of giant intracytoplasmic granules in most of the cells of the organism [6] but often they are identified in peripheral blood as was the case of our patients or in the bone marrow. The diagnosis is confirmed by a genetic test for the mutation LYST. Antenatal diagnosis of the disease is possible in the cells of the chorionic villi, amniotic fluid, leucocytes of the fetal cord [25]. As with Chediak-Higashi disease, other genetic immune deficiencies are accompanied by partial oculocutaneous albinism, such as Griscelli’s disease and Hermansky-Pudlak syndrome. The distinction can be made unambiguously by the different appearance of pigment clusters in the hair sheath, much finer in the case of CHS, and especially by the presence of giant intracytoplasmic granules observed only in the CHS. However, in some cases of myeloid leukemia, one can see giant granulations called pseudo Chediak- Higashi anomaly [26]. The treatment of Chediak-Higashi disease is multidisciplinary and is based on the management of complications of the disease, treatment of the “accelerated phase” or HLH and especially the transplantation of hematopoietic stem cells. Symptomatic treatment of Chediak-Higashi disease is based on effective antibiotic therapy against infections and transfusions of blood derivatives to fight anemia and hemorrhagic complications. Eye disorders should be corrected. The eyes and skin should be protected from UV rays. Vaccinations are generally well tolerated as was the case for our patients and must be done. Hygiene and oral health care are Primordial. The occurrence of neurological symptoms and their progression must be dealt with early enough by a rehabilitation specialist. In the case of an acceleration phase (HLH, a treatment combining corticosteroids, VP16, cyclosporin and intrathecal injections of MTX (HLH 2004) [23] is introduced to achieve remission, which occurs in 75% of cases [27], but relapses are frequent and response to treatment decreases over time. When transplantation is achieved, transplantation is recommended. In SCH patients with HLH by EBV the addition of Rituximab could improve treatment [28]. In the case of refractory HLH, another therapeutic option including a monoclonal anti-CD52 antibody (Alemtuzumab) [29] is possible as a second line treatment before bone marrow transplantation. Allogeneic bone marrow transplantation (BMT) is the only current effective treatment that heals hematologic and immunological abnormalities, but has no effect on oculocutaneous albinism or subsequent neurological deterioration [9,24,30]. The pre-graft conditioning regimen comprises a combination of etoposide, busulfan, cyclophosphamide [31]. Reduced intensity of pre-graft conditioning with fludarabine, melphalan, and alemtuzumab resulted in increased survival in primary or family HLH with lower toxicity [32,33]. Bone marrow transplantation is most effective when performed before the accelerated phase occurs [31]. Patients with a profound decrease in the cytotoxic function of T lymphocytes (CTL) have a high risk of developing lymphocytic syndrome (HLH), so their screening may be an indication for early marrow transplantation [21]. The overall survival rate after marrow transplantation is 60-70% (30-32) The prognosis remains poor in the absence of a bone marrow transplant, death occurs frequently during the first decade by infections or development of an accelerated phase HLH [34] About 10 percent of patients who survive in early childhood will develop severe neurological disorders in adolescence and early adulthood [35].
Conclusion
Chediak-Higashi syndrome is a rare disease, the diagnosis is suspected in a child with oculocutaneous albinism with recurrent infections. The majority of clinical forms are early “infantile” lethal in the absence of treatment. A minority of patients present with an “attenuated” form of the disease will survive after childhood but develop an associated neuro-degerative disease. In all cases early diagnosis should be posed by a simple examination, peripheral blood smear which shows the presence of giant intracytoplasmic granules pathognomonic of this condition. The only current effective treatment of haematological and immunological abnormalities remains allogeneic bone marrow transplantation, but without impact on skin manifestations or subsequent neurological deterioration. It is more effective when it is performed before the onset of an HLH syndrome. In the event of the occurrence of accelerated phase (HLH), a treatment according to the HLH 2004 protocol is instituted in order to obtain a remission before the bone marrow transplant. The prognosis of infant form is poor, with death occurring frequently in the first decade of life through infections or development of HLH. The search for predictive factors for the development of HLH may help in the indication of early bone marrow transplantation.
References
- Picard C, Al-Herz W, Bousfiha A, Casanova JL, Chatila T, et al. (2015) Primary Immunodeficiency Diseases: an Update on the Classification from the International Union of Immunological Societies Expert Committee for Primary Immunodeficiency 2015. J Clin Immunol 35(8): 696-726.
- Barbosa MD, Nguyen QA, Tchernev VT, Ashley JA, Detter JC, et al. (1996) Identification of the homologous beige and Chediak-Higashi syndrome genes. Nature 382: 262-265.
- Nagle DL, Karim MA, Woolf EA, Holmgren L, Bork P, et al. (1996) Identification and mutation analysis of the complete gene for Chediak-Higashi syndrome. Nat Genet 14: 307-311.
- Kaplan J, De Domenico I, Ward DM (2008) Chediak-Higashi syndrome. Curr Opin Hematol 15: 22-29.
- Reddy RR, Babu BM, Venkateshwaramma B, Hymavathi Ch (2011) Silvery hair syndrome in two cousins: Chediak-Higashi syndrome vs Griscelli syndrome, with rare associations. Int J Trichology 3(2): 107-111.
- JC Stinchcombe, LJ Page, GM Griffiths (2000) Secretory lysosome biogenesis in cytotoxic T lymphocytes from normal and Chediak Higashi syndrome patients. Traffic 1(5): 435-444.
- Pooja Jaiswal, Yogesh Kumar Yadav, Nilam Bhasker, Rashmi Kushwaha (2015) Accelerated Phase of Chediak-Higashi Syndrome at Initial Presentation: A Case Report of an Uncommon Occurrence in a Rare Disorder. Journal of Clinical and Diagnostic Research 9(12): 13-14.
- Introne WJ, Westbroek W, Golas GA, Adams D (2009) Chediak-Higashi syndrome. In Gene Reviews™. Edited by Pagon RA, Adam MP, Bird TD, Dolan CR, Fong CT, Stephens K. Seattle (WA): University of Washington, Seattle, USA.
- Nagai K, Ochi F, Terui K, Maeda M, Ohga S, et al. (2013) Clinical characteristics and outcomes of Chediak-Higashi syndrome: a nationwide survey of Japan. Pediatr Blood Cancer 60: 1582-1586.
- Maria L Lozano, Jose Rivera, Isabel Sánchez (2014) Guiu and Vicente Vicente Orphanet Journal of Rare Diseases 9: 132.
- Westbroek W, Adams D, Huizing M, Koshoffer A, Dorward H, et al. (2007) The severity of cellular defects in Chediak-Higashi syndrome correlate with the molecular genotype and clinical phenotype. J Invest Dermatol 127: 2674-2677.
- Barak Y, Nir E (1987) Chédiak-Higashi - Syndrome. Am J Pediatr Hematol Oncol 9(1): 42-55.
- Ho MC, Hsieh YT (2013) Mixed hyperpigmentation and hypopigmentation of iris and choroid in Chediak-Higashi syndrome. J AAPOS 17: 558-560.
- Pujani M, Agarwal K, Bansal S, Ahmad I, Puri V, et al. (2011) ChediakHigashi syndrome - a report of two cases with unusual hyperpigmentation of the face. Turkish J Pathol 27(3): 246-248.
- Karim MA, Suzuki K, Fukai K, Oh J, Nagle DL, et al. (2002) Apparent genotype-phenotype correlation in childhood, adolescent, and adult Chediak Higashi syndrome. Am J Med Genet 108: 16-22.
- Yenan T Bryceson, Daniela Pende, Andrea Maul Pavicic, Kimberly C Gilmour, HeikeUfheil, et al. (2012) A prospective evaluation of degranulation assays in the rapid diagnosis of familial hemophagocytic syndromes Blood 119: 2754-2763.
- MC Dinauer (2014) Disorders of Neutrophil Function: An Overview Methods Mol Biol 1124: 501-515.
- Pullarkat ST (2012) Accelerated phase of Chediak-Higashi syndrome. Blood 119(1): 5.
- Lozano ML, Jose Rivera, Isabel Sanchez Guiu, Vicente V (2014) Towards the targeted management of Chediak-Higashi syndrome Orphanet Journal of Rare Diseases 9: 132.
- Gajendra S, Das RR, Chopra A, Singh A, Seth R (2014) Accelerated Phase at Initial Presentation in Chédiak-Higashi Syndrome: Is It Really Uncommon? Pediatr Hematol Oncol 31: 382-385.
- Birthe Jessen, Andrea Maul Pavicic, Heike Ufheil, Thomas Vraetz, Anselm Enders, et al. (2011) Subtle differences in CTL cytotoxicity determine susceptibility to hemophagocytic lymphohistiocytosis in mice and humans with Chediak-Higashi syndrome Blood 118: 4620-4629.
- Wendy J Introne, Wendy Westbroek, Gretchen A Golas, David Adams (2015) Chediak Higashi Syndrome GeneReviews®.
- Henter JI, Home A, Arico M, Egeler RM, Filipovich AH, et al. (2007) HLH-2004 - Diagnostic and therapeutic guidelines for haemophagocytic lymphohistiocytosis. Paediatr Blood Cancer 48: 124-131.
- Tardieu M, Lacroix C, Neven B, Bordigoni P, de Saint Basile G, et al. (2005) Progressive neurologic dysfunctions 20 years after allogeneic bone marrow transplantation for Chediak-Higashi syndrome Blood 106: 40-42.
- Diukman R, Tanigawa S, Cowan MJ, Golbus MS (1992) Prenatal diagnosis of Chédiak-Higashi Syndrome. Prenat Diagn 12(11): 877-885.
- Chang H, Yi QL (2006) Acute Myeloid Leukemia With Pseudo-Chediak-Higashi Anomaly Exhibits a Specific Immunophenotype With CD2 Expression. Am J Clin Pathol 125: 791-794.
- Filipovich AH (2006) Hemophagocytic lymphohistiocytosis and related disorders. Curr Opin Allergy Clin Immunol 6: 410-415.
- Ogimi C, Tanaka R, Arai T, Kikuchi A, Hanada R, et al. (2011) Rituximab and cyclosporine therapy for accelerated phase Chediak-Higashi syndrome. Pediatr Blood Cancer 57: 677-680.
- Marsh RA, Allen CE, Mcclain KL, Weinstein JL, Kanter J, et al. (2013) Salvage therapy of refractory hemophagocytic lymphohistiocytosis with alemtuzumab. Pediatr Blood Cancer 60: 101-109.
- Eapen M, DeLaat CA, Baker KS, Cairo MS, Cowan MJ, et al. (2007) Hematopoietic cell transplantation for Chediak-Higashi syndrome. Bone Marrow Transplant 39: 411-415.
- Haddad E, Le Deist F, Blanche S, Benkerrou M, Rohrlich P, et al. (1995) Treatment of Chediak-Higashi syndrome by allogenic bone marrow transplantation: report of 10 cases. Blood 85: 3328-3333.
- Marsh RA, Vaughn G, Kim MO, Li D, Jodele S, et al. (2010) Reduced-intensity conditioning (RIC) significantly improves survival of patients with hemophagocytic lymphohistiocytosis undergoing allogeneic hematopoietic cell transplantation. Blood 116: 5824-5831.
- Marsh RA, Jordan MB, Filipovich AH (2011) Reduced-intensity conditioning haematopoietic cell transplantation for haemophagocytic lymphohistiocytosis: an important step forward. Br J Haematol 154: 556-563.
- Cooper N, Rao K, Gilmour K, Hadad L, Adams S et al. (2006) Stem cell transplantation with reduced-intensity conditioning for hemophagocytic lymphohistiocytosis. Blood 107: 1233-1236.
- Laura Dotta, Silvia Parolini, Alberto Prandini, Giovanna Tabellini, Maddalena Antolini, et al. (2013) Clinical, laboratory and molecular signs of immunodeficiency in patients with partial oculo-cutaneous albinism. Orphanet Journal of Rare Diseases 8: 168.

Top Editors
-

Mark E Smith
Bio chemistry
University of Texas Medical Branch, USA -

Lawrence A Presley
Department of Criminal Justice
Liberty University, USA -

Thomas W Miller
Department of Psychiatry
University of Kentucky, USA -

Gjumrakch Aliev
Department of Medicine
Gally International Biomedical Research & Consulting LLC, USA -

Christopher Bryant
Department of Urbanisation and Agricultural
Montreal university, USA -

Robert William Frare
Oral & Maxillofacial Pathology
New York University, USA -

Rudolph Modesto Navari
Gastroenterology and Hepatology
University of Alabama, UK -

Andrew Hague
Department of Medicine
Universities of Bradford, UK -

George Gregory Buttigieg
Maltese College of Obstetrics and Gynaecology, Europe -

Chen-Hsiung Yeh
Oncology
Circulogene Theranostics, England -
.png)
Emilio Bucio-Carrillo
Radiation Chemistry
National University of Mexico, USA -
.jpg)
Casey J Grenier
Analytical Chemistry
Wentworth Institute of Technology, USA -
Hany Atalah
Minimally Invasive Surgery
Mercer University school of Medicine, USA -

Abu-Hussein Muhamad
Pediatric Dentistry
University of Athens , Greece

The annual scholar awards from Lupine Publishers honor a selected number Read More...